UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
Date of report (Date of earliest event reported): March 2, 2026
|
|
|
|
|
(State or other jurisdiction of incorporation)
|
(Commission File Number)
|
(IRS Employer Identification No.)
|
|
|
|
|
|
(Address of principal executive offices)
|
(Zip Code)
|
Registrant’s telephone number, including area code: (858 ) 567-7770
N/A
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the
following provisions (see General Instruction A.2. below):
|
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
|
|
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
|
|
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b))
|
|
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c))
|
Securities registered pursuant to Section 12(b) of the Act:
|
Title of each class
|
Trading Symbol(s)
|
Name of each exchange on which
registered
|
||
|
|
|
|
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter)
or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or
revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
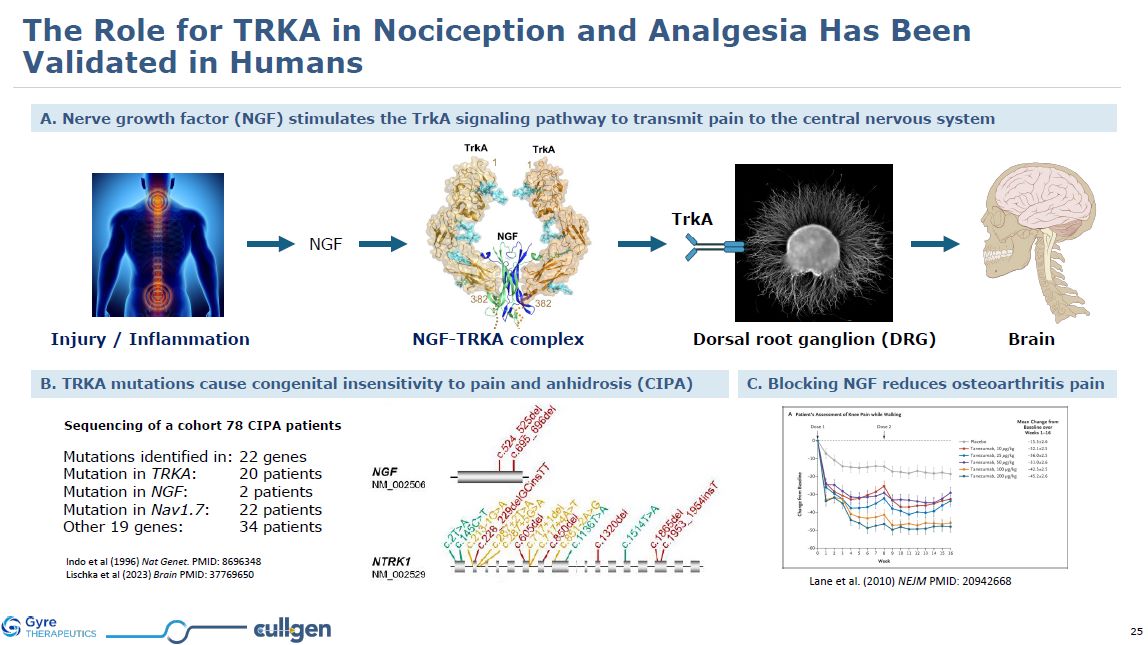
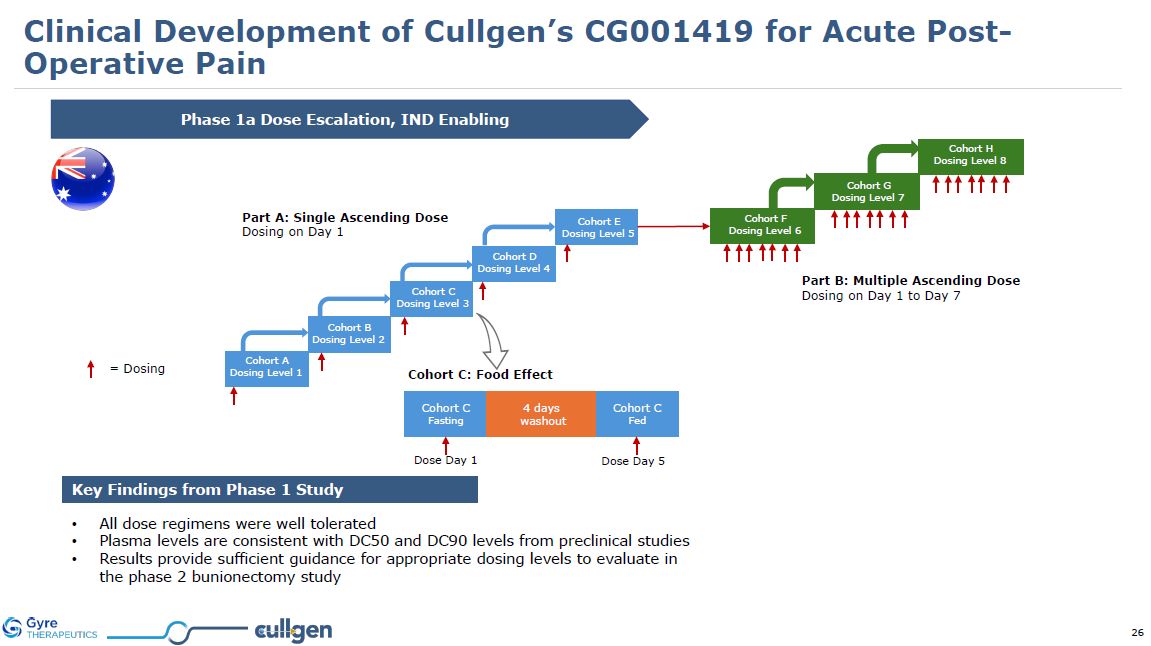
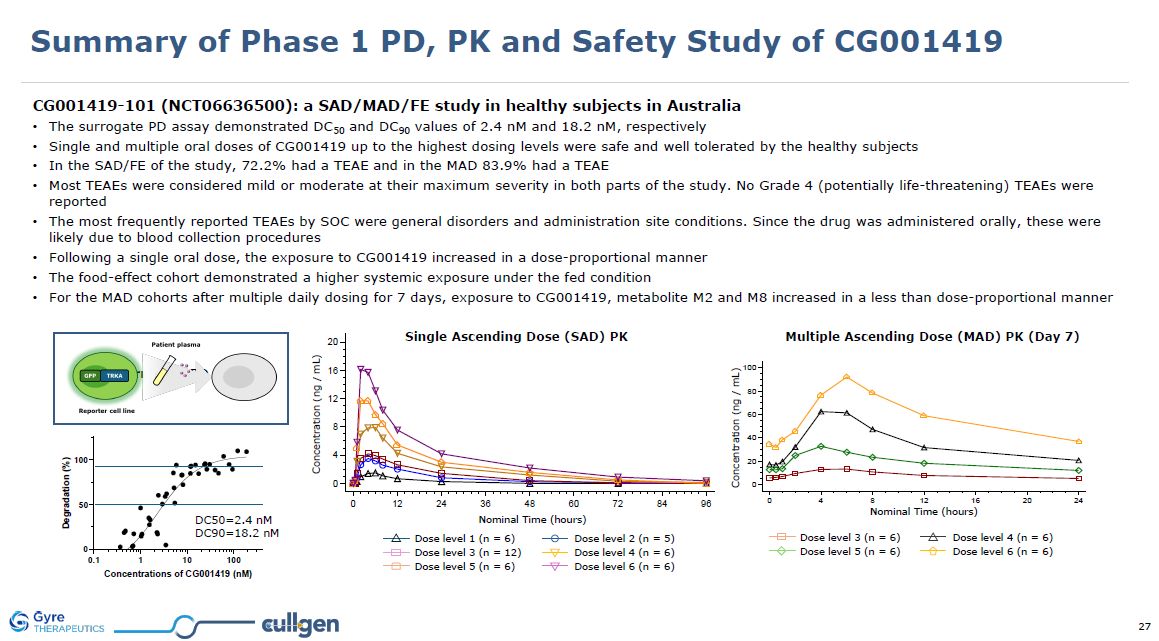
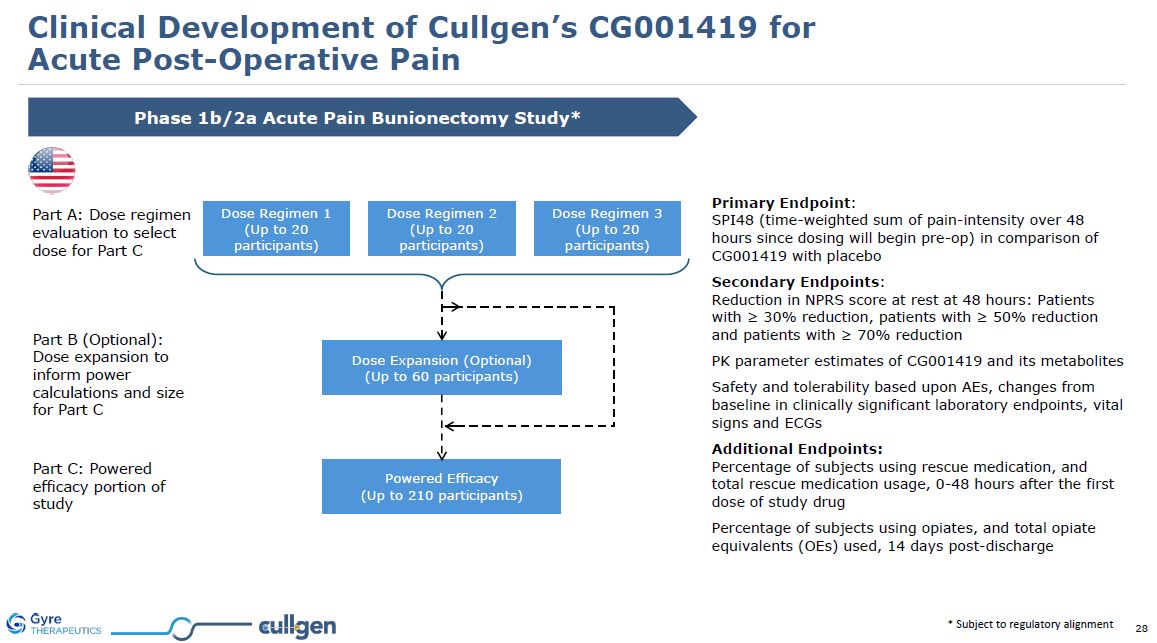
| Item 1.01 |
Entry into a Material Definitive Agreement.
|
Agreement and Plan of Merger and Reorganization
On March 2, 2026, Gyre Therapeutics, Inc., a Delaware corporation (the “Company” or “Gyre”), entered into an Agreement and Plan of Merger and
Reorganization (the “Merger Agreement”) with Cullgen Inc., a Delaware corporation (“Cullgen”), and Helix Merger Sub Corp., a Delaware corporation and wholly owned subsidiary of the Company (“Merger Sub”), pursuant to which, among other matters, and
subject to the satisfaction or waiver of the conditions set forth in the Merger Agreement, Merger Sub will merge with and into Cullgen, with Cullgen continuing as a wholly owned subsidiary of the Company and the surviving corporation of the merger
(the “Merger”). The Merger is intended to qualify for federal income tax purposes as a tax-free reorganization under the provisions of Section 368(a) of the Internal Revenue Code of 1986, as amended.
Under the terms of the Merger Agreement, the Company will acquire Cullgen in an all-stock transaction that values Cullgen at approximately $300
million. At the effective time of the Merger (the “Effective Time”), each then outstanding share of Cullgen capital stock (the “Cullgen Capital Stock”), excluding shares of Cullgen Capital Stock held as treasury stock immediately prior to the
Effective Time and any dissenting shares, will be converted into (1) with respect to shares of Cullgen Capital Stock held by certain designated holders, (i) for each share of Cullgen common stock (“Cullgen Common Stock”) held by such holders, a
number of shares of the Company's Series B Convertible Preferred Stock, par value $0.001 per share (the “Company Preferred Stock”), equal to (x) 0.4753 (the “Exchange Ratio”) divided by five, and (ii) for
each share of Cullgen preferred stock (“Cullgen Preferred Stock”) held by such designated holders, a number of shares of Company Preferred Stock equal to (x) the number of shares of Cullgen Common Stock issuable upon conversion of each share of
Cullgen Preferred Stock, multiplied by the Exchange Ratio, and divided by five, and (2) with respect to shares of Cullgen Capital Stock held by each other holder, (i)
for each share of Cullgen Common Stock held by such holders, a number of shares of Company common stock, par value $0.001 per share (the “Company Common Stock”), equal to the Exchange Ratio divided by five,
and (ii) for each share of Cullgen Preferred Stock held by such designated holders, a number of shares of Company Common Stock equal to the number of shares of Cullgen Common Stock issuable upon conversion of each share of Cullgen Preferred Stock, multiplied by the Exchange Ratio. Each share of Company Preferred Stock received in the Merger is convertible into five shares of Company Common Stock, subject to certain conditions described below with respect to
the Conversion Proposal (as defined below). Notwithstanding anything herein to the contrary, in no event will the Company issue greater than 19.99% of its issued and outstanding Company Common Stock or its voting power prior to the approval of the
Conversion Proposal.
In addition, at the Effective Time (1) each then-outstanding in-the-money option to purchase shares of Cullgen Common Stock that is outstanding and
unexercised immediately prior to the Effective Time, whether vested or unvested, will cease to represent a right to acquire shares of Cullgen Common Stock and will be converted into and become an option to purchase shares of Company Common Stock on
the existing terms and conditions (including with respect to vesting and accelerated vesting), subject to adjustment as set forth in the Merger Agreement, (2) each then-outstanding option to purchase shares of Cullgen Common Stock that is not an
in-the-money option and is outstanding and unexercised immediately prior to the Effective Time will be cancelled at the Effective Time for no consideration, and (3) each Cullgen restricted stock unit will vest and be settled for Cullgen Common Stock
and the holder thereof will be entitled to receive a number of shares of Company Common Stock calculated in accordance with the Merger Agreement.
Reference is made to the discussion of the Company Preferred Stock in Item 5.03 of this Current Report on Form 8-K, which is incorporated into this
Item 1.01 by reference.
Each of the Company and Cullgen has agreed to customary representations, warranties and covenants in the Merger Agreement, including, among others,
covenants relating to (1) using commercially reasonable efforts to obtain the requisite approval of its stockholders, (2) non-solicitation of alternative acquisition proposals, (3) the conduct of their respective businesses during the period between
the date of signing the Merger Agreement and the closing of the Merger, and (4) the Company filing with the U.S. Securities and Exchange Commission (the “SEC”) a proxy statement and other relevant materials relating to the Conversion Proposal (as
defined below). The consummation of the Merger is subject to certain closing conditions, including, among other things, (1) approval by the requisite Cullgen stockholders of the adoption and approval of the Merger Agreement and the transactions
contemplated thereby, and (2) a filing under The Hart-Scott-Rodino (HSR) Antitrust Improvements Act of 1976, as amended. Each party’s obligation to consummate the Merger is also subject to other specified customary conditions, including regarding the
accuracy of the representations and warranties of the other party, subject to the applicable materiality standard, and the performance in all material respects by the other party of its obligations under the Merger Agreement required to be performed
on or prior to the date of the closing of the Merger (the “Merger Closing Date”). The Merger Agreement contains certain termination rights of each of the Company and Cullgen. The Board of Directors of the Company (the “Board”) approved the Merger
Agreement and the related transactions, and the consummation of the Merger does not require the approval of the Company stockholders.
The Merger Agreement has been attached as an exhibit to this Current Report on Form 8-K to provide investors and securityholders with information
regarding its terms. It is not intended to provide any other factual information about the Company or Cullgen or to modify or supplement any factual disclosures about the Company in its public reports filed with the SEC. The Merger Agreement includes
representations, warranties and covenants of the Company, Cullgen and Merger Sub made solely for the purpose of the Merger Agreement and solely for the benefit of the parties thereto in connection with the negotiated terms of the Merger Agreement.
Investors should not rely on the representations, warranties and covenants in the Merger Agreement or any descriptions thereof as characterizations of the actual state of facts or conditions of the Company, Cullgen, Merger Sub or any of their
respective affiliates. Moreover, certain of those representations and warranties may not be accurate or complete as of any specified date, may be subject to a contractual standard of materiality different from those generally applicable to SEC
filings or may have been used for purposes of allocating risk among the parties to the Merger Agreement, rather than establishing matters of fact. Moreover, information concerning
the subject matter of the representations and warranties may change after the date of the Merger Agreement, which subsequent information may or may not be fully reflected in the Company’s public disclosures. For the foregoing reasons, no person
should rely on the representations and warranties as statements of factual information at the time they were made or otherwise.
Pursuant to the Merger Agreement, the Company has agreed to convene a meeting of its stockholders to submit to its stockholders for their
consideration the approval of the conversion of the Company Preferred Stock into shares of Company Common Stock in accordance with certain of the rules of the Nasdaq Stock Market LLC (the “Conversion Proposal”). In connection with these matters, the
Company intends to file with the SEC a proxy statement and other relevant materials.
The foregoing description of the Merger and the Merger Agreement does not purport to be complete and is
qualified in its entirety by reference to the Merger Agreement, which is filed as Exhibit 2.1 to this Current Report on Form 8-K and is incorporated herein by reference.
Support Agreements and Lock-Up Agreement
Concurrently with the execution of the Merger Agreement, (i) certain officers, directors, and stockholders of the Company (solely in their respective
capacities as Company stockholders) have entered into support agreements with the Company and Cullgen to vote all of their shares of Company Common Stock in favor of the Conversion Proposal (the “Company Support Agreement”), and (ii) certain
officers, directors and stockholders of Cullgen (solely in their respective capacities as Cullgen stockholders) have entered into support agreements with the Company and Cullgen to vote all of their shares of Cullgen Capital Stock in favor of the
adoption and approval of the Merger Agreement and the transactions contemplated thereby and against any alternative acquisition proposals (the “Cullgen Support Agreement” and together with the
Company Support Agreement, the “Support Agreements”).
Concurrently with the execution of the Merger Agreement, certain executive officers, directors and stockholders of the Company and Cullgen have
entered into lock-up agreements (the “Lock-Up Agreements”) pursuant to which, subject to specified exceptions, they have agreed not to transfer their shares of Company Common Stock (or shares convertible for Company Common Stock) for (a) with respect
to one-third of the shares of Company Common Stock held by them, the 180-day period following the Merger Closing Date, (b) with respect to one-third of the shares of Company Common Stock held by them, the twelve-month period following the Merger
Closing Date, and (c) with respect to one-third of the shares of Company Common Stock held by them, the eighteen month period following the Merger Closing Date.
The foregoing description of the Support Agreements and the Lock-Up Agreements does not purport to
be complete and is qualified in its entirety by reference to the form of Cullgen Support Agreement, the form of Company Support Agreement and the form of Lock-Up Agreement, which are filed as
Exhibits 10.1, 10.2 and 10.4, respectively, to this Current Report on Form 8-K and which are incorporated herein by reference.
Registration Rights Agreement
In connection with the closing of
the Merger, the Company will enter into a Registration Rights Agreement (the “Registration Rights Agreement”) with Cullgen and certain holders of shares of Cullgen Capital Stock signatory thereto (the
“Company Holders”). Pursuant to the Registration Rights Agreement, the Company is required to prepare and file a resale registration statement with the SEC within 45 calendar days following the Merger
Closing Date with respect to the shares of Company Common Stock (and shares of Company Common Stock issuable upon conversion of the Company Preferred Stock). The Company shall use reasonable best efforts to
cause this registration statement to be declared effective by the SEC within 90 business days of the Merger Closing Date (or, in the event of a “full review” by the SEC, within 120 calendar days of the Merger Closing Date).
The Company has also agreed to, among other things, indemnify the Company Holders and their affiliates, the
officers, directors, members, partners, agents, brokers, investment advisors, and employees of each of them under the registration statement from certain liabilities and pay all fees and expenses (excluding any legal fees of the selling holder(s),
and any underwriting discounts and selling commissions) incident to the Company’s obligations under the Registration Rights Agreement.
The foregoing summary of the Registration Rights Agreement does not purport to be complete and is qualified in its entirety by reference to the form
of Registration Rights Agreement, which is filed as Exhibit 10.3 to this Current Report on Form 8-K.
|
Item 3.02
|
Unregistered Sales of Equity Securities.
|
To the extent required by this Item, the information included in Item 1.01 of this Current Report on Form 8-K is incorporated herein by reference.
|
Item 5.02
|
Departure of Directors or Certain Officers; Election of Directors; Appointment of Certain Officers; Compensatory Arrangements of Certain Officers. |
Resignation of Directors
In accordance with the Merger Agreement, immediately prior to the Effective Time, Thomas Eastling and Songjiang Ma will resign from
the Board and any respective committee of the Board to which they were members, and Mr. Ma will also resign from his position as President of the Company on such date. The resignations are not the result of any disagreements with the Company
relating to the Company’s operations, policies or practices. The size of the Board will be reduced to seven directors.
Appointment of Director and Chief Executive Officer and President
In accordance with the Merger Agreement, effective immediately after the Effective Time, the Company will appoint Ying Luo, Ph.D. as
Chief Executive Officer and President of the Company and to the Board as a Class I director.
Ying Luo, Ph.D., age 60, has served as Chairman of the board of directors and Chief Executive Officer of Cullgen since 2018. He has
also served as a director, representative executive officer, president and chief executive officer of GNI Group Ltd. (“GNI Japan”), a vertically-integrated, multinational bio-pharma company listed on the Tokyo Stock Exchange, since 2007, Chief
Executive Officer of Shanghai Genomics, Inc., a wholly owned subsidiary of GNI Japan, from 2001 to 2021, a member of the board of directors of GNI Hong Kong Limited, a wholly owned subsidiary of GNI Japan, since 2013, a member of the board of
directors and President of GNI USA, a wholly owned subsidiary of GNI Japan (“GNI USA”), since 2015, a director of Berkeley Advanced Biomaterials LLC, a wholly owned subsidiary of GNI USA, since 2017, chairman of the board of directors of Gyre,
from October 2023 to January 2025 and chairman of the board of directors of Gyre Pharmaceuticals Co., Ltd., a majority owned subsidiary of Gyre, from 2011 to January 2025. Dr. Luo previously served as a member of the board of directors of
Catalyst Biosciences, Inc., a Nasdaq-listed biopharmaceutical company (“Catalyst”), from December 2022 until the completion of Catalyst’s business combination in October 2023 to become Gyre. Dr. Luo was a postdoctoral fellow at the University of
California at San Francisco studying HIV gene regulation from 1991 to 1992, a scientist at Aviron Company from 1992 to 1993, a scientist at Clontech Laboratories from 1993 to 1997 and senior scientist/director/senior director of genomics and
target discovery of Rigel Inc. from 1997 to 2000, where he led research in the field of protein-protein interactions in cancer and inflammation signaling pathways. In his career, Dr. Luo has authored more than 37 research publications. Dr. Luo
completed his undergraduate education at Peking Union Medical College (Peking University’s Premedicine) from 1982 to 1986 and received his doctorate in biomedical sciences from the University of Connecticut Health Center in 1991.
There are no arrangements or understandings between Dr. Luo and any other person pursuant to which he will be appointed as a director
of the Company. Dr. Luo is not a party to any transaction required to be disclosed pursuant to Item 404(a) of Regulation S-K.
Indemnification Agreement
In connection with Dr. Luo’s appointment as Chief Executive Officer and President and director, Dr. Luo will enter into the Company’s
standard form of indemnification agreement, a form of which was previously filed as Exhibit 10.3 to the Company’s Current Report on Form 8-K filed on October 30, 2023.
|
Item 5.03
|
Amendments to Articles of Incorporation or Bylaws; Change in Fiscal Year.
|
The Company expects to file with the Secretary of State of the State of Delaware a Certificate of Designation of Preferences, Rights and
Limitations of the Company Preferred Stock (the “Certificate of Designation”) in connection with the Merger referenced in Item 1.01 above. The Certificate
of Designation provides for the issuance of shares of the Company Preferred Stock.
Holders of Company Preferred Stock are entitled to receive dividends on shares of Company Preferred Stock equal to, on an as-if-converted-to-Common-Stock basis, and in the same form as dividends actually paid on shares of Company Common Stock. The Company Preferred Stock has the voting rights set forth in the
Certificate of Designation, including that such shares have one vote on all matters submitted to the stockholders of the Company for approval, subject to certain limitations. In addition, until the Conversion Proposal is approved, the Company will not, without the affirmative vote of the holders of a majority of the then outstanding shares of Company Preferred Stock (a) alter or change adversely the powers, preferences or rights given to
the Company Preferred Stock, (b) alter or amend the Certificate of Designation, (c) amend its certificate of incorporation or other charter documents in any manner that adversely affects any
rights of the holders of Company Preferred Stock, (d) issue further shares of Company Preferred Stock, or increase or decrease (other than by conversion) the number of authorized shares of Company Preferred Stock, (e) consummate a Fundamental
Transaction (as defined in the Certificate of Designation of Company Preferred Stock) or any merger or consolidation of the Company or other business combination in which the stockholders of the Company immediately before such transaction do not
hold at least a majority of the capital stock of the Company immediately after such transaction, or (f) enter into any agreement with respect to any of the foregoing. The Company Preferred Stock does not have a preference upon any liquidation,
dissolution or winding-up of the Company.
Following stockholder approval of the Conversion Proposal, each share of Company Preferred Stock will be convertible, at the option
of the holder, into five shares of Company Common Stock, subject to certain limitations, including that a holder of Company Preferred Stock is prohibited from converting shares of Company Preferred Stock into shares of Company Common Stock if, as a
result of such conversion, such holder, together with its affiliates, would beneficially own more than a specified percentage (to be established by the holder between 0% and 19.99%) of the total number of shares of Company Common Stock issued and
outstanding immediately after giving effect to such conversion.
The foregoing description of the Company Preferred Stock does not purport to be complete and is qualified in their entirety by
reference to the Certificate of Designation, a copy of which is filed as Exhibit 3.1 to this Current Report on Form 8-K and are incorporated herein by reference.
| Item 7.01 |
Regulation FD Disclosure.
|
On March 2, 2026, The Company and Cullgen issued a joint press release announcing the execution of the Merger Agreement. The press release is
furnished as Exhibit 99.1 to this Current Report on Form 8-K and incorporated herein by reference, except that the information contained on the websites referenced in the press release is not incorporated herein by reference.
On March 2, 2026, the Company made available a presentation to be used with investors to discuss the Merger. A copy of the presentation is furnished as Exhibit 99.2
to this Current Report on Form 8-K.
The information in Item 7.01 of this Current Report on Form 8-K, including the information in the press release attached as Exhibit
99.1 and the presentation attached as Exhibit 99.2 to this Current Report on Form 8-K, is furnished pursuant to Item 7.01 of Form 8-K and shall not be deemed “filed” for the purposes of Section 18 of the
Securities Exchange Act of 1934, as amended, or otherwise subject to the liabilities of that section. Furthermore, the information in Item 7.01 of this Current Report on Form 8-K, including the information in the press release attached as Exhibit
99.1 and the presentation attached as Exhibit 99.2 to this Current Report on Form 8-K, shall not be deemed to be incorporated by reference in the filings of the Company under the Securities Act of 1933, as
amended.
Forward-Looking Statements
This Current Report on Form 8-K contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995,
including, but not limited to, statements regarding: stockholder approval of the Conversion Proposal, the filing of a resale registration statement pursuant to the Registration Rights Agreement and the timing thereof; and the closing of the Merger, if at all. The use of words such as, but not limited to, “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “might,” “plan,” “potential,”
“predict,” “project,” “should,” “target,” “will,” or “would” and similar words expressions are intended to identify forward-looking statements. Forward-looking statements are neither historical facts nor assurances of future performance. Instead,
they are based on the Company’s current beliefs, expectations and assumptions regarding the future of its business, future plans and strategies, its clinical results and other future conditions. New risks and uncertainties may emerge from time to
time, and it is not possible to predict all risks and uncertainties. No representations or warranties (expressed or implied) are made about the accuracy of any such forward-looking statements. The Company may not actually achieve the forecasts
disclosed in its forward-looking statements, and you should not place undue reliance on forward-looking statements. Such forward-looking statements are subject to a number of material risks and uncertainties including but not limited to those set
forth under the caption “Risk Factors” in the Company’s most recent Annual Report on Form 10-K filed with the SEC, as supplemented by its Quarterly Reports on Form 10-Q, as well as discussions of potential risks, uncertainties, and other important
factors in the Company’s subsequent filings with the SEC. Any forward-looking statement speaks only as of the date on which it was made. Neither the Company, nor its affiliates, advisors or representatives, undertake any obligation to publicly
update or revise any forward-looking statement, whether as result of new information, future events or otherwise, except as required by law. These forward-looking statements should not be relied upon as representing the Company’s views as of any
date subsequent to the date hereof.
Item 9.01 – Financial Statements and Exhibits.
| (d) |
Exhibits.
|
|
Exhibit
Number
|
Description
|
|
Agreement and Plan of Merger and Reorganization, dated March 2, 2026, by and among Gyre Therapeutics, Inc., Helix Merger Sub Corp., and Cullgen Inc.
|
|
|
Certificate of Designation of Series B Convertible Preferred Stock
|
|
|
Form of Cullgen Support Agreement
|
|
|
Form of Company Support Agreement
|
|
|
Form of Registration Rights Agreement
|
|
|
Form of Lock-Up Agreement
|
|
|
Joint Press Release, issued on March 2, 2026
|
|
|
Investor Presentation, dated March 2, 2026
|
|
|
104
|
Cover Page Interactive Data File (embedded within the Inline
XBRL document)
|
* Exhibits and/or schedules have been omitted pursuant to Item 601(a)(5) of Regulation S-K. The registrant hereby undertakes to furnish supplementally
copies of any of the omitted exhibits and schedules upon request by the SEC; provided, however, that the registrant may request confidential treatment pursuant to Rule 24b-2 under the Exchange Act for any exhibits or schedules so furnished.
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its
behalf by the undersigned hereunto duly authorized.
|
GYRE THERAPEUTICS, INC.
|
||
|
Date: March 2, 2026
|
By:
|
/s/ Ping Zhang
|
|
Name:
|
Ping Zhang
|
|
|
Title:
|
Executive Chairman and Interim Chief Executive Officer
|
|
Exhibit 2.1
Confidential
AGREEMENT AND PLAN OF MERGER AND REORGANIZATION
among:
GYRE THERAPEUTICS, INC.;
HELIX MERGER SUB CORP.;
and
CULLGEN INC.
Dated as of March 2, 2026
Table of Contents
|
Section 1. Definitions and Interpretative Provisions
|
3
|
|
|
1.1 Definitions
|
3
|
|
|
1.2 Other Definitional and Interpretative Provisions
|
14
|
|
|
Section 2. Description of Transaction
|
14
|
|
|
2.1 The Merger
|
14
|
|
|
2.2 Effects of the Merger
|
15
|
|
|
2.3 Closing; Effective Time
|
15
|
|
|
2.4 Organizational Documents; Directors and Officers
|
15
|
|
|
2.5 Conversion of Company; Merger Sub Equity Securities
|
15
|
|
|
2.6 Closing of the Company’s Transfer Books
|
17
|
|
|
2.7 Surrender of Company Capital Stock
|
18
|
|
|
2.8 Reserved
|
19
|
|
|
2.9 Further Action
|
19
|
|
|
2.10 Intended Tax Treatment
|
19
|
|
|
2.11 Withholding
|
19
|
|
|
2.12 Appraisal Rights
|
19
|
|
|
Section 3. Representations and Warranties of the Company
|
20
|
|
|
3.1 Due Organization; Subsidiaries
|
20
|
|
|
3.2 Organizational Documents
|
20
|
|
|
3.3 Authority; Binding Nature of Agreement
|
20
|
|
|
3.4 Vote Required
|
21
|
|
|
3.5 Non-Contravention; Consents
|
21
|
|
|
3.6 Capitalization
|
22
|
|
|
3.7 Financial Statements
|
23
|
|
|
3.8 Absence of Changes
|
24
|
|
|
3.9 Absence of Undisclosed Liabilities
|
24
|
|
|
3.10 Title to Assets
|
24
|
|
|
3.11 Real Property; Leasehold
|
24
|
|
|
3.12 Intellectual Property
|
24
|
|
|
3.13 Agreements, Contracts and Commitments
|
27
|
|
|
3.14 Compliance; Permits; Restrictions
|
29
|
|
|
3.15 Legal Proceedings; Orders
|
31
|
|
|
3.16 Tax Matters
|
31
|
|
|
3.17 Employee and Labor Matters; Benefit Plans
|
33
|
|
|
3.18 Environmental Matters
|
35
|
|
|
3.19 Insurance
|
36
|
|
|
3.20 No Financial Advisors
|
36
|
|
|
3.21 Transactions with Affiliates
|
36
|
|
|
3.22 Privacy and Data Security
|
36
|
|
|
3.23 No Other Representations or Warranties
|
37
|
|
|
Section 4. Representations and Warranties of Parent and Merger Sub
|
37
|
|
|
4.1 Due Organization; Subsidiaries
|
37
|
|
|
4.2 Organizational Documents
|
38
|
|
|
4.3 Authority; Binding Nature of Agreement
|
38
|
|
|
4.4 Vote Required
|
38
|
|
|
4.5 Non-Contravention; Consents
|
38
|
|
A-1
|
4.6 Capitalization
|
39
|
|
|
4.7 SEC Filings; Financial Statements
|
41
|
|
|
4.8 Absence of Changes
|
42
|
|
|
4.9 Absence of Undisclosed Liabilities
|
42
|
|
|
4.10 Title to Assets
|
43
|
|
|
4.11 Real Property; Leasehold
|
43
|
|
|
4.12 Intellectual Property
|
43
|
|
|
4.13 Agreements, Contracts and Commitments
|
45
|
|
|
4.14 Compliance; Permits; Restrictions
|
47
|
|
|
4.15 Legal Proceedings; Orders
|
50
|
|
|
4.16 Tax Matters
|
50
|
|
|
4.17 Employee and Labor Matters; Benefit Plans
|
52
|
|
|
4.18 Environmental Matters
|
54
|
|
|
4.19 Insurance
|
54
|
|
|
4.20 Transactions with Affiliates
|
55
|
|
|
4.21 No Financial Advisors
|
55
|
|
|
4.22 Valid Issuance
|
55
|
|
|
4.23 Privacy and Data Security
|
55
|
|
|
4.24 No Other Representations or Warranties
|
55
|
|
|
Section 5. Certain Covenants of the Parties
|
56
|
|
|
5.1 Operation of Parent’s Business
|
56
|
|
|
5.2 Operation of the Company’s Business
|
57
|
|
|
5.3 Access and Investigation
|
58
|
|
|
5.4 No Solicitation
|
59
|
|
|
5.5 Notification of Certain Matters
|
60
|
|
|
Section 6. Additional Agreements of the Parties
|
60
|
|
|
6.1 Proxy Statement
|
60
|
|
|
6.2 Company Stockholder Written Consent
|
61
|
|
|
6.3 Parent Stockholder Meeting
|
62
|
|
|
6.4 Efforts; Regulatory Approvals
|
63
|
|
|
6.5 Company RSUs; Company Options
|
64
|
|
|
6.6 Employee Benefits
|
65
|
|
|
6.7 Indemnification of Officers and Directors
|
66
|
|
|
6.8 Disclosure
|
67
|
|
|
6.9 Listing
|
67
|
|
|
6.10 Tax Matters
|
67
|
|
|
6.11 Legends
|
68
|
|
|
6.12 Officers and Directors..
|
68
|
|
|
6.13 Termination of Certain Agreements and Rights
|
68
|
|
|
6.14 Section 16 Matters
|
68
|
|
|
6.15 Allocation Information
|
68
|
|
|
6.16 Parent SEC Documents
|
68
|
|
|
6.17 Obligations of Merger Sub
|
69
|
|
|
6.18 Operations of the Combined Company
|
69
|
|
|
6.19 Reservation of Parent Common Stock; Issuance of Shares of Parent Common Stock
|
69
|
|
|
Section 7. Conditions Precedent to Obligations of Each Party
|
69
|
|
|
7.1 Regulatory Approvals
|
69
|
|
|
7.2 Foreign Person Status
|
69
|
|
A-2
|
7.3 No Restraints
|
69
|
|
|
7.4 Lock-Up Agreements
|
69
|
|
|
7.5 Certificate of Designation
|
69
|
|
|
7.6 Registration Rights Agreement
|
69
|
|
|
Section 8. Additional Conditions Precedent to Obligations of Parent and Merger Sub
|
70
|
|
|
8.1 Accuracy of Representations
|
70
|
|
|
8.2 Performance of Covenants
|
70
|
|
|
8.3 Documents
|
70
|
|
|
8.4 No Company Material Adverse Effect
|
70
|
|
|
8.5 Company Stockholder Written Consent
|
71
|
|
|
Section 9. Additional Conditions Precedent to Obligation of the Company.
|
71
|
|
|
9.1 Accuracy of Representations
|
71
|
|
|
9.2 Performance of Covenants
|
71
|
|
|
9.3 Documents
|
71
|
|
|
9.4 No Parent Material Adverse Effect
|
71
|
|
|
Section 10. Termination
|
71
|
|
|
10.1 Termination
|
71
|
|
|
10.2 Effect of Termination
|
73
|
|
|
10.3 Expenses
|
73
|
|
|
Section 11. Miscellaneous Provisions
|
73
|
|
|
11.1 Non-Survival of Representations and Warranties
|
73
|
|
|
11.2 Amendment
|
73
|
|
|
11.3 Waiver
|
73
|
|
|
11.4 Entire Agreement; Counterparts; Exchanges by Electronic Transmission or Facsimile
|
74
|
|
|
11.5 Applicable Law; Jurisdiction
|
74
|
|
|
11.6 Assignability
|
74
|
|
|
11.7 Notices
|
74
|
|
|
11.8 Cooperation
|
75
|
|
|
11.9 Severability
|
75
|
|
|
11.10 Other Remedies; Specific Performance
|
75
|
|
|
11.11 No Third-Party Beneficiaries
|
76
|
|
Exhibits:
|
Exhibit A-1:
|
Form of Parent Stockholder Support Agreement
|
|
Exhibit A-2:
|
Form of Company Stockholder Support Agreement
|
|
Exhibit B:
|
Form of Lock-Up Agreement
|
|
Exhibit C:
|
Registration Rights Agreement
|
|
Exhibit D:
|
Certificate of Merger, incorporated by reference into this Agreement
|
|
Exhibit E:
|
Certificate of Designation
|
A-3
AGREEMENT AND PLAN OF MERGER AND REORGANIZATION
This Agreement and Plan of Merger and Reorganization (this “Agreement”) is made and entered into as of March 2, 2026, by and among Gyre Therapeutics, Inc., a Delaware corporation (“Parent”), Helix Merger Sub Corp., a Delaware
corporation and wholly owned subsidiary of Parent (“Merger Sub”), and Cullgen Inc., a Delaware corporation (the “Company”). Certain capitalized terms used in this
Agreement are defined Section 1.
RECITALS
A. Parent and the Company intend to effect a merger of Merger Sub with and into the Company (the “Merger”) in accordance with this Agreement
and the DGCL. Upon consummation of the Merger, Merger Sub will cease to exist with the Company being the surviving entity and a wholly-owned subsidiary of Parent.
B. The Parties intend that (i) the Merger qualify as a “reorganization” within the meaning of Section 368(a) of the Code, and (ii) this Agreement will constitute, and is hereby
adopted as, a plan of reorganization within the meaning of Treasury Regulations Sections 1.368-2(g) and 1.368-3(a).
C. The Parent Board has established a special committee (the “Special Committee”), consisting solely of members of the
Parent Board who are disinterested and independent of Parent and the Company and their respective Affiliates to, among other things, negotiate the terms of certain transactions, including the Contemplated Transactions, and to make a recommendation
to the Parent Board as to whether the Company should enter into this Agreement;
D. The Special Committee was empowered to approve and recommend to the Parent Board the Contemplated Transactions, and the Parent Board resolved that Parent would not effectuate
the Contemplated Transactions unless, among other things, the Contemplated Transactions were first approved or recommended by the Special Committee;
E. The Special Committee has unanimously (i) determined that the Contemplated Transactions are fair to, advisable and in the best interests of Parent and its stockholders
(including the stockholders who are not affiliated with the Company), (ii) approved and declared advisable this Agreement and the Contemplated Transactions, including the issuance of shares of Parent Capital Stock to the stockholders of the Company
pursuant to the terms of this Agreement, and (iii) determined to recommend, upon the terms and subject to the conditions set forth in this Agreement, that the stockholders of Parent vote to approve the Preferred Stock Conversion Proposal (such
recommendation, the “Special Committee Recommendation”);
F. All disinterested and independent members of the Parent Board, acting upon the unanimous recommendation of the Special Committee at a duly called meeting, have unanimously (i)
determined that the Contemplated Transactions are fair to, advisable and in the best interests of Parent and its stockholders (including the stockholders who are not affiliated with the Company), (ii) approved and declared advisable this Agreement
and the Contemplated Transactions, including the issuance of shares of Parent Capital Stock to the stockholders of the Company pursuant to the terms of this Agreement, and (iii) determined to recommend, upon the terms and subject to the conditions
set forth in this Agreement, that the stockholders of Parent vote to approve the Preferred Stock Conversion Proposal.
G. The Merger Sub Board has (i) determined that the Contemplated Transactions are fair to, advisable, and in the best interests of Merger Sub and its sole stockholder, (ii)
approved and declared advisable this Agreement and the Contemplated Transactions and (iii) determined to recommend, upon the terms and subject to the conditions set forth in this Agreement, that the stockholder of Merger Sub votes to adopt this
Agreement and thereby approve the Contemplated Transactions.
H. The Company Board has (i) determined that the Contemplated Transactions are fair to, advisable and in the best interests of the Company and its stockholders, (ii) approved and
declared advisable this Agreement and the Contemplated Transactions and (iii) determined to recommend, upon the terms and subject to the conditions set forth in this Agreement, that the stockholders of the Company vote to adopt this Agreement and
thereby approve the Contemplated Transactions.
I. Concurrently with the execution and delivery of this Agreement and as a condition and inducement to the Company’s willingness to enter into this Agreement, each of the officers,
directors and stockholders of Parent set forth on Section A of the Parent Disclosure Letter (solely in their capacity as stockholders of Parent) are executing support agreements in favor of the Company in substantially the form
attached hereto as Exhibit A-1, pursuant to which such Persons have, subject to the terms and conditions set forth therein, agreed to vote all of their shares of Parent Capital Stock in favor of the approval of the Preferred Stock Conversion
Proposal.
J. Concurrently with the execution and delivery of this Agreement and as a condition and inducement to Parent’s willingness to enter into this Agreement, each of the officers,
directors and stockholders of the Company listed on Schedule A of the Company Disclosure Letter (solely in their capacity as stockholders of the Company), collectively representing the Required Company Stockholder Vote, are executing
support agreements in favor of Parent in substantially the form attached hereto as Exhibit A-2 (the “Company Stockholder Support Agreement”), pursuant to which such Persons have, subject to the terms
and conditions set forth therein, agreed to vote all of their shares of Company Capital Stock in favor of the adoption of this Agreement and thereby approve the Company Stockholder Written Consents, including the Contemplated Transactions.
K. Concurrently with the execution and delivery of this Agreement and as a condition and inducement to Parent’s and the Company’s willingness to enter into this Agreement, each of
the stockholders of the Company or Parent listed on Schedule B of the Company Disclosure Letter are executing lock-up agreements in substantially the form attached hereto as Exhibit B (the “Lock-Up
Agreement,” and collectively, the “Lock-Up Agreements”).
L. It is expected that as promptly as practicable following the execution and delivery of this Agreement, the holders of shares of Company Capital Stock sufficient to adopt and
approve this Agreement and the Merger as required under the DGCL and the Company’s certificate of incorporation and bylaws will execute and deliver an action by written consent adopting this Agreement, in form and substance reasonably acceptable to
Parent, in order to obtain the Required Company Stockholder Vote.
M. At the Closing, Parent, the Company and certain of the holders of shares of Company Capital Stock shall enter into the registration rights
agreement in substantially the form attached hereto as Exhibit C (the “Registration Rights Agreement”).
2
AGREEMENT
The Parties, intending to be legally bound, agree as follows:
“Acquisition Inquiry” means, with respect to a Party, an inquiry, indication of interest or request for non-public information (other than
an inquiry, indication of interest or request for information made or submitted by the Company, on the one hand, or Parent, on the other hand, to the other Party) that would reasonably be expected to lead to an Acquisition Proposal.
“Acquisition Proposal” means, with respect to a Party, any offer or proposal, whether written or oral (other than an offer or proposal made
or submitted by or on behalf of the Company or any of its Affiliates, on the one hand, or by or on behalf of Parent or any of its Affiliates, on the other hand, to the other Party) contemplating or otherwise relating to any Acquisition Transaction
with such Party.
“Acquisition Transaction” means any transaction or series of related transactions involving:
(a) any merger, consolidation, amalgamation, share exchange, business combination, issuance of securities, acquisition of securities, reorganization, recapitalization, tender
offer, exchange offer or other similar transaction: (i) in which a Person or “group” (as defined in the Exchange Act and the rules promulgated thereunder) of Persons directly or indirectly acquires beneficial or record ownership of securities
representing more than 20% of the outstanding securities of any class of voting securities of a Party or any of its Subsidiaries or (ii) in which a Party or any of its Subsidiaries issues securities representing more than 20% of the outstanding
securities of any class of voting securities of such Party or any of its Subsidiaries, or issues securities convertible into more than 20% of the outstanding securities of any class of voting securities of such Party or any of its Subsidiaries; or
(b) any sale, lease, exchange, transfer, license, acquisition or disposition of any business or businesses or assets that constitute or account for 20% or more of the consolidated
book value or the fair market value of the assets of a Party and its Subsidiaries, taken as a whole.
“Affiliate” shall have the meaning given to such term in Rule 145 under the Securities Act.
“Affordable Care Act” means the Patient Protection and Affordable Care Act.
“Agreed Business Plan” means the business plan of the combined company delivered to the Special Committee prior to the date hereof and
included as Schedule C of the Company Disclosure Letter.
“Business Day” means any day other than a day on which banks in the State of New York are authorized or obligated to be closed.
“Certificate of Designation” means the Certificate of Designation of Preferences, Rights and Limitations of Parent Series B Convertible
Preferred Stock in the form attached hereto as Exhibit E.
“COBRA” means the Consolidated Omnibus Budget Reconciliation Act of 1985, as set forth in Section 4980B of the Code and Section 6 of Title I
of ERISA.
3
“Code” means the Internal Revenue Code of 1986, as amended.
“Company Associate” means any current employee, independent contractor, officer or director of the Company or any of its Subsidiaries.
“Company Board” means the board of directors of the Company.
“Company Capital Stock” means the Company Common Stock and the Company Preferred Stock.
“Company Capitalization Representations” means the representations and warranties of the Company set forth in Sections 3.6(a)
and 3.6(d).
“Company Common Stock” means the common stock, $0.0001 par value per share, of the Company.
“Company Contract” means any Contract: (a) to which the Company or any of its Subsidiaries is a Party, (b) by which the Company or any of
its Subsidiaries is or may become bound or under which the Company or any of its Subsidiaries has, or may become subject to, any obligation or (c) under which the Company or any of its Subsidiaries has or may acquire any right or interest.
“Company Employee Plan” means any Employee Plan that the Company or any of its Subsidiaries (i) sponsors, maintains, administers, or
contributes to, or (ii) provides benefits under or through, or (iii) has any obligation to contribute to or provide benefits under or through, or (iv) may reasonably be expected to have any Liability, or (v) utilizes to provide benefits to or
otherwise cover any current or former employee, officer, director or other service provider of the Company or any of its Subsidiaries (or their spouses, dependents, or beneficiaries).
“Company Fundamental Representations” means the representations and warranties of the Company set forth in Sections 3.1(a),
3.2, 3.3, 3.4, 3.5(a)(i) and 3.20.
“Company IP Rights” means all Intellectual Property rights that are owned or purported to be owned by, assigned to, exclusively licensed to,
or controlled by the Company or its Subsidiaries that are necessary for, or used or held for use in, the operation of the business of the Company and its Subsidiaries as presently conducted.
“Company IP Rights Agreement” means any Contract governing, related to or pertaining to any Company IP Rights other than any confidential
information provided under confidentiality agreements.
“Company Key Employee” means any executive officer of the Company or any of its Subsidiaries.
“Company Material Adverse Effect” means any Effect that, considered together with all other Effects that have occurred prior to the date of
determination of the occurrence of a Company Material Adverse Effect, has or would reasonably be expected to have a material adverse effect on the business, financial condition, assets, liabilities or results of operations of the Company or its
Subsidiaries, taken as a whole; provided, however, that Effects arising or resulting from the following shall not be taken into account in determining whether there has been a Company Material Adverse Effect: (a) the announcement of
this Agreement or the pendency of the Contemplated Transactions, (b) the taking of any action, or the failure to take any action, by the Company that is required to comply with the terms of this Agreement, (c) any natural disaster, calamity or
epidemics, pandemics or other force majeure events, or any act or threat of terrorism or war, any armed hostilities or terrorist activities (including any escalation or general worsening of any of the foregoing) anywhere in the world or any
governmental or other response or reaction to any of the foregoing, (d) any change in GAAP or applicable Law or the interpretation thereof, or (e) general economic or political conditions or conditions generally affecting the industries in which
the Company and its Subsidiaries operate; except in each case with respect to clauses (c), (d) and (e), to the extent disproportionately affecting the Company and its Subsidiaries, taken as a whole, relative to other similarly situated companies in
the industries in which the Company and its Subsidiaries operate.
4
“Company Options” means options or other rights to purchase shares of Company Capital Stock issued by the Company.
“Company Preferred Stock” means the shares of the Company’s capital stock designated as preferred stock, including the Company Series Seed-1
Preferred Stock, Company Series Seed-2 Preferred Stock, Company Series A Preferred Stock, Company Series B Preferred Stock and Company Series C Preferred Stock.
“Company Registered IP” means all Company IP Rights that are owned or exclusively licensed by the Company that are registered, filed or
issued under the authority of, with or by any Governmental Authority, including all patents, registered copyrights and registered trademarks and all applications and registrations for any of the foregoing.
“Company Series A Preferred Stock” means a series of the Company’s preferred stock designated as Series A Preferred Stock, $0.0001 par value
per share.
“Company Series B Preferred Stock” means a series of the Company’s preferred stock designated as Series B Preferred Stock, $0.0001 par value
per share.
“Company Series C Preferred Stock” means a series of the Company’s preferred stock designated as Series C Preferred Stock, $0.0001 par value
per share.
“Company Series Seed-1 Preferred Stock” means a series of the Company’s preferred stock designated as Series Seed-1 Preferred Stock, $0.0001
par value per share.
“Company Series Seed-2 Preferred Stock” means a series of the Company’s preferred stock designated as Series Seed-2 Preferred Stock, $0.0001
par value per share.
“Company Stock Plan” means the Company’s 2018 Equity Incentive Plan.
“Confidentiality Agreement” means the non-disclosure agreement dated as of January 10, 2026, between the Company and Parent.
“Consent” means any approval, consent, ratification, permission, waiver or authorization (including any Governmental Authorization).
“Contemplated Transactions” means the Merger and the other transactions contemplated by this Agreement.
“Continuing Employee” means each employee of the Company or any Subsidiary of the Company as of immediately prior to the Closing who,
following the Closing, remains an employee of the Surviving Entity or such Subsidiary or becomes an employee of Parent or any of its Affiliates.
5
“Contract” means, with respect to any Person, any written agreement, contract, subcontract, lease (whether for real or personal property),
mortgage, license, or other legally binding commitment or undertaking of any nature to which such Person is a party or by which such Person or any of its assets are bound or affected under applicable Law.
“Designated Holder” means each holder of Company Capital Stock listed as a “Designated Holder” on Schedule D of the Company
Disclosure Letter.
“DGCL” means the General Corporation Law of the State of Delaware.
“Effect” means any effect, change, event, circumstance, or development.
“Employee Plan” means (A) an “employee benefit plan” within the meaning of Section 3(3) of ERISA whether or not subject to ERISA; (B) other
plan, program, policy or arrangement providing for stock options, stock purchases, equity-based compensation, bonuses (including any annual bonuses and retention bonuses) or other incentives, severance pay, deferred compensation, employment,
compensation, change in control or transaction bonuses, supplemental, vacation, retirement benefits (including post-retirement health and welfare benefits), pension benefits, profit-sharing benefits, fringe benefits, life insurance benefits,
perquisites, health benefits, medical benefits, dental benefits, vision benefits, and all other employee benefit plans, agreements, and arrangements, not described in (A) above; and (C) all other plans, programs, policies or arrangements providing
compensation to employees, consultants and non-employee directors.
“Encumbrance” means any lien, pledge, hypothecation, charge, mortgage, security interest, lease, exclusive license, option, easement,
reservation, servitude, adverse title, claim, infringement, interference, option, right of first refusal, preemptive right, community property interest or restriction or encumbrance of any nature (including any restriction on the voting of any
security, any restriction on the transfer of any security or other asset, any restriction on the receipt of any income derived from any asset, any restriction on the use of any asset and any restriction on the possession, exercise or transfer of
any other attribute of ownership of any asset).
“Enforceability Exceptions” means the (a) Laws of general application relating to bankruptcy, insolvency and the relief of debtors and (b)
rules of law governing specific performance, injunctive relief and other equitable remedies.
“Entity” means any corporation (including any nonprofit corporation), partnership (including any general partnership, limited partnership or
limited liability partnership), joint venture, estate, trust, company (including any company limited by shares, limited liability company or joint stock company), firm, society or other enterprise, association, organization or entity, and each of
its successors.
“Environmental Law” means any federal, state, local or foreign Law relating to pollution or protection of human health or the environment
(including ambient air, surface water, ground water, land surface or subsurface strata), including any law or regulation relating to emissions, discharges, releases or threatened releases of Hazardous Materials, or otherwise relating to the
manufacture, processing, distribution, use, treatment, storage, disposal, transport or handling of Hazardous Materials.
“ERISA” means the Employee Retirement Income Security Act of 1974, as amended.
“ERISA Affiliate” means, with respect to any Entity, any other Person that would be treated as a single employer with such Entity or part of
the same “controlled group” as such Entity under Sections 414(b),(c),(m) or (o) of the Code.
6
“Exchange Act” means the Securities Exchange Act of 1934, as amended.
“Exchange Ratio” means 0.4753.
“Governmental Authority” means any: (a) nation, state, commonwealth, province, territory, county, municipality, district or other
jurisdiction of any nature, (b) federal, state, local, municipal, foreign, supra-national or other government, (c) governmental or quasi-governmental authority of any nature (including any governmental division, department, agency, commission,
bureau, instrumentality, official, ministry, fund, foundation, center, organization, unit, body or Entity and any court or other tribunal, and for the avoidance of doubt, any taxing authority) or (d) self-regulatory organization (including Nasdaq).
“Governmental Authorization” means any: (a) permit, license, certificate, franchise, permission, variance, exception, order, approval,
clearance, registration, qualification or authorization issued, granted, given or otherwise made available by or under the authority of any Governmental Authority or pursuant to any Law or (b) right under any Contract with any Governmental
Authority.
“Hazardous Materials” means any pollutant, chemical, substance and any toxic, infectious, carcinogenic, reactive, corrosive, ignitable or
flammable chemical, or chemical compound, or hazardous substance, material or waste, whether solid, liquid or gas, that is subject to regulation, control or remediation under any Environmental Law, including without limitation, crude oil or any
fraction thereof, and petroleum products or by-products.
“HSR Act” means the U.S. Hart Scott-Rodino Antitrust Improvements Act of 1976, as amended.
“In-the-Money Company Option” shall mean Company Options with an exercise price equal to or less than the quotient of (i) the Company
Valuation, divided by (ii) the total number of shares of Company Capital Stock outstanding immediately prior to the Effective Time, on a fully diluted and as-converted to Company Common Stock basis.
“Intellectual Property” means: (a) United States, foreign and international patents, patent applications, including all provisionals,
nonprovisionals, substitutions, divisionals, continuations, continuations-in-part, reissues, extensions, supplementary protection certificates, reexaminations, term extensions, certificates of invention and the equivalents of any of the foregoing,
statutory invention registrations, invention disclosures and inventions (collectively, “Patents”), (b) trademarks, service marks, trade names, domain names, corporate names, brand names, URLs, trade dress, logos and other source identifiers,
including registrations and applications for registration thereof and goodwill associated therewith, (c) copyrights, including registrations and applications for registration thereof, (d) software, including all source code, object code and related
documentation, (e) formulae, customer lists, trade secrets, know-how, confidential information and other proprietary rights and intellectual property, whether patentable or not, and (f) all United States and foreign rights arising under or
associated with any of the foregoing.
“IRS” means the United States Internal Revenue Service.
“Knowledge” means, (i) with respect to an individual, that such individual is actually aware of the relevant fact or such individual would
reasonably be expected to know such fact in the ordinary course of the performance of such individual’s employment responsibilities, (ii) with respect to Parent, the Knowledge of the individuals listed on Section A of the Parent Disclosure
Letter as of the date of such knowledge is imputed and (iii) with respect to Company, the Knowledge of the individuals listed on Schedule E of the Company Disclosure Letter as of the date of such knowledge is imputed, and (iv) any Person
that is an Entity (other than Parent and the Company) the Knowledge of any executive officer of such Person as of the date such knowledge is imputed. With respect to any matters relating to Intellectual Property, such awareness or reasonable
expectation to have knowledge does not require any such individual to conduct or have conducted or obtain or have obtained any freedom to operate opinions of counsel or any Intellectual Property rights clearance searches.
7
“Law” means any federal, state, national, supra-national, foreign, local or municipal or other law, statute, constitution, principle of
common law, resolution, ordinance, code, edict, decree, rule, regulation, ruling or requirement issued, enacted, adopted, promulgated, implemented or otherwise put into effect by or under the authority of any Governmental Authority (including under
the authority of Nasdaq or the Financial Industry Regulatory Authority).
“Legal Proceeding” means any action, suit, litigation, arbitration, proceeding (including any civil, criminal, administrative, investigative
or appellate proceeding), hearing, inquiry, audit, examination or investigation commenced, brought, conducted or heard by or before any court or other Governmental Authority or any arbitrator or arbitration panel.
“Merger Sub Board” means the board of directors of Merger Sub.
“Multiemployer Plan” means a “multiemployer plan,” as defined in Section 3(37) or 4001(a)(3) of ERISA.
“Multiple Employer Plan” means a “multiple employer plan” within the meaning of Section 413(c) of the Code or Section 3(40) of ERISA.
“Multiple Employer Welfare Arrangement” means a “multiple employer welfare arrangement” within the meaning of Section 3(40) of ERISA.
“Nasdaq” means The Nasdaq Stock Market LLC.
“Non-Designated Holder” means each holder of Company Capital Stock who is not a Designated Holder.
“Order” means any judgment, order, writ, injunction, ruling, decision or decree of (that is binding on a Party), or any plea agreement,
corporate integrity agreement, resolution agreement or deferred prosecution agreement with, or any settlement under the jurisdiction of, any court or Governmental Authority.
“Ordinary Course of Business” means, in the case of each of the Company and Parent, such actions taken in the ordinary course of its
business and consistent with its past practice or, with respect to the Company, the customary practices of a company at a similar stage of development.
“Organizational Documents” means, with respect to any Person (other than an individual), (a) the certificate or articles of association or
incorporation or organization or limited partnership or limited liability company, and any joint venture, limited liability company, operating or partnership agreement and other similar documents adopted or filed in connection with the creation,
formation or organization of such Person and (b) all bylaws, regulations and similar documents or agreements relating to the organization or governance of such Person, in each case, as amended or supplemented.
“Parent Associate” means any current employee, independent contractor, officer or director of Parent or any of its Subsidiaries.
8
“Parent Balance Sheet” means the audited balance sheet of Parent as of September 30, 2025, included in Parent’s Report on Form 10-Q for the
nine-months ended September 30, 2025, as filed with the SEC.
“Parent Board” means the board of directors of Parent.
“Parent Capital Stock” means the Parent Common Stock and the Parent Preferred Stock.
“Parent Capitalization Representations” means the representations and warranties of Parent and Merger Sub set forth in Sections 4.6(a)
and 4.6(d).
“Parent Common Stock” means the common stock, $0.001 par value per share, of Parent.
“Parent Convertible Preferred Stock” means Parent’s Series B voting convertible preferred stock, par value $0.001 per share, with the
rights, preferences, powers and privileges specified in the Certificate of Designation.
“Parent Contract” means any Contract: (a) to which Parent is a party, (b) by which Parent or any Parent IP Rights or any other asset of
Parent is or may become bound or under which Parent has, or may become subject to, any obligation or (c) under which Parent has or may acquire any right or interest.
“Parent Employee Plan” means any Employee Plan that Parent or any of its Subsidiaries (i) sponsors, maintains, administers, or contributes
to, or (ii) provides benefits under or through, or (iii) has any obligation to contribute to or provide benefits under or through, or (iv) may reasonably be expected to have any Liability, or (v) utilizes to provide benefits to or otherwise cover
any current or former employee, officer, director or other service provider of Parent or any of its Subsidiaries (or their spouses, dependents, or beneficiaries).
“Parent Fundamental Representations” means the representations and warranties of Parent and Merger Sub set forth in Sections 4.1(a),
4.2, 4.3, 4.4, 4.5(a)(i) and 4.21.
“Parent IP Rights” means all Intellectual Property owned, licensed or controlled by Parent that is necessary for, or used or held for use
in, the operation of the business of Parent.
“Parent IP Rights Agreement” means any Contract governing, related or pertaining to any Parent IP Rights.
“Parent Key Employee” means (i) an executive officer of Parent; and (ii) any employee of Parent that reports directly to the Parent Board or
to an executive officer of Parent.
“Parent Material Adverse Effect” means any Effect that, considered together with all other Effects that have occurred prior to the date of
determination of the occurrence of the Parent Material Adverse Effect, has or would reasonably be expected to have a material adverse effect on the business, financial condition, assets, liabilities or results of operations of Parent and its
Subsidiaries, taken as a whole; provided, however, that Effects arising or resulting from the following shall not be taken into account in determining whether there has been a Parent Material Adverse Effect: (a) the announcement of
this Agreement or the pendency of the Contemplated Transactions, (b) any change in the stock price or trading volume of Parent Common Stock (it being understood, however, that any Effect causing or contributing to any change in stock price or
trading volume of Parent Common Stock may be taken into account in determining whether a Parent Material Adverse Effect has occurred, unless such Effects are otherwise excepted from this definition), (c) the taking of any action, or the failure to
take any action, by Parent that is required to comply with the terms of this Agreement, (d) any natural disaster, calamity or epidemics, pandemics or other force majeure events, or any act or threat of terrorism or war, any armed hostilities or
terrorist activities (including any escalation or general worsening of any of the foregoing) anywhere in the world, or any governmental or other response or reaction to any of the foregoing, or (e) any change in GAAP or applicable Law or the
interpretation thereof; except, in each case with respect to clauses (d), (e) and (f), to the extent materially and disproportionately affecting Parent or any of its Subsidiaries, taken as a whole, relative to other similarly situated companies in
the industries in which Parent or any of its Subsidiaries operates.
9
“Parent Options” means options or other rights to purchase shares of Parent Common Stock granted by Parent, including pursuant to any Parent
Stock Plan.
“Parent Preferred Stock” means the shares of Parent’s capital stock designated as preferred stock, par value $0.001 per share of Parent,
including the Parent Convertible Preferred Stock.
“Parent Registered IP” means all Parent IP Rights that are owned or exclusively licensed by Parent that are registered, filed or issued
under the authority of, with or by any Governmental Authority, including all patents, registered copyrights and registered trademarks and all applications for any of the foregoing.
“Party” or “Parties” means the Company, Merger Sub and Parent.
“Permitted Encumbrance” means (a) any statutory liens for current Taxes not yet due and payable or for Taxes that are being contested in
good faith by the appropriate proceedings and for which adequate reserves have been made on the Company Balance Sheet or the Parent Balance Sheet, as applicable, in accordance with GAAP, (b) minor non-monetary liens that have arisen in the Ordinary
Course of Business and that do not (in any case or in the aggregate) materially detract from the value of the assets subject thereto or materially impair the operations of the Company or Parent, as applicable, (c) statutory liens to secure
obligations to landlords, lessors or renters under leases or rental agreements, (d) deposits or pledges made in connection with, or to secure payment of, workers’ compensation, unemployment insurance or similar programs mandated by Law, (e)
statutory liens in favor of carriers, warehousemen, mechanics and materialmen, to secure claims for labor, materials or supplies for amounts that are not yet due and payable and (f) liens arising under applicable securities Law.
“Person” means any individual, Entity or Governmental Authority.
“Personal Information” means any data or information that constitutes “personal information,” “personal data,” “personally identifiable
information,” “protected health information,” or any analogous term under applicable Law, including any such information that identifies, relates to, describes, is linked to, is reasonably capable of being associated with, or could reasonably be
linked, directly or indirectly, with any identified or identifiable individual or household.
“Privacy Laws” mean, collectively, (i) all Laws governing privacy, data protection, data security, trans-border data flow, data loss, data
theft, breach notification, data localization, sending solicited or unsolicited electronic mail or text messages, cookies or other tracking technology, or the collection, handling, use, maintenance, storage, disclosure, transfer, or other
processing of Personal Information, including any such legally binding requirements set forth in regulations and agreements containing consent orders published by regulatory authorities of competent jurisdiction such as the U.S. Federal Trade
Commission, U.S. Federal Communications Commission, and state data protection authorities, including HIPAA, Section 5 of the Federal Trade Commission Act, the Telephone Consumer Protection Act and U.S. state consumer protection and data breach
notification Laws, and (ii) any legally binding requirements of any self-regulatory organizations governing data privacy, data protection, data security, trans-border data flow, data loss, data theft, breach notification, data localization, sending
solicited or unsolicited electronic mail or text messages, cookies or other tracking technology, or the collection, handling, use, maintenance, storage, disclosure, transfer, or other processing of Personal Information.
10
“Representatives” means with respect to a Person, such Person’s directors, officers, employees, agents, attorneys, accountants, investment
bankers, advisors and other representatives; provided, for the avoidance of doubt, that Representatives of the Company expressly do not include GNI Group Ltd. or any of its Affiliates (including without limitation, GNI USA).
“Sarbanes-Oxley Act” means the Sarbanes-Oxley Act of 2002.
“SEC” means the United States Securities and Exchange Commission.
“Securities Act” means the Securities Act of 1933, as amended.
“Subsidiary” means, with respect to an Entity, a Person if such Person directly or indirectly owns or purports to own, beneficially or of
record, (a) an amount of voting securities or other interests in such Entity that is sufficient to enable such Person to elect at least a majority of the members of such entity’s board of directors or other governing body or (b) at least 50% of the
outstanding equity, voting, beneficial or financial interests in such Entity.
“Tax” means any U.S. federal, state, local, foreign or other tax, including any income tax, franchise tax, capital gains tax, gross receipts
tax, value-added tax, surtax, estimated tax, employment tax, unemployment tax, national health insurance tax, environmental tax, excise tax, ad valorem tax, transfer tax, conveyance tax, stamp tax, sales tax, use tax, property tax, business tax,
withholding tax, payroll tax, social security tax, customs duty, licenses tax, alternative or add-on minimum or other tax or similar charge, duty, levy, fee, tariff, impost, obligation or assessment in the nature of a tax (whether imposed directly
or through withholding and whether or not disputed), and including any fine, penalty, addition to tax, interest or additional amount imposed by a Governmental Authority with respect thereto (or attributable to the nonpayment thereof).
“Tax Return” means any return (including any information return), report, statement, declaration, claim or refund, estimate, schedule,
notice, notification, form, election, certificate or other document or information, and any amendment or supplement to any of the foregoing, filed or required to be filed with any Governmental Authority (or provided to a payee) in connection with
the determination, assessment, collection or payment of any Tax or in connection with the administration, implementation or enforcement of or compliance with any Law relating to any Tax.
“Treasury Regulations” means the United States Treasury regulations promulgated under the Code.
(c) Each of the following terms is defined in the Section set forth opposite such term:
|
Terms
|
Section
|
|
Agreement
|
Preamble
|
|
Allocation Certificate
|
6.15
|
|
Assumed Option
|
6.5(b)
|
|
Capitalization Date
|
4.6(a)
|
|
Certificate of Merger
|
2.3
|
11
|
Certifications
|
4.7(a)
|
|
Closing
|
2.3
|
|
Closing Date
|
2.3
|
|
Company
|
Preamble
|
|
Company 409A Plan
|
3.17(i)
|
|
Company Balance Sheet
|
3.7(a)
|
|
Company Board Recommendation
|
6.2(c)
|
|
Company Disclosure Letter
|
3.7(a)
|
|
Company Financials
|
Section 3
|
|
Company Interim Financial Statements
|
6.1(e)
|
|
Company Material Contract
|
3.13(a)
|
|
Company Material Contracts
|
3.13(a)
|
|
Company Permits
|
3.14(b)
|
|
Company Product Candidates
|
3.14(d)
|
|
Company Real Estate Leases
|
3.11
|
|
Company Regulatory Permits
|
3.14(d)
|
|
Company Required Proxy Information
|
6.1(d)
|
|
Company RSU
|
6.5(a)
|
|
Company Stock Certificates
|
2.7(b)
|
|
Company Stockholder Support Agreement
|
Recital
|
|
Company Stockholder Written Consents
|
6.2(a)
|
|
Costs
|
6.7(a)
|
|
D&O Indemnified Parties
|
6.7(a)
|
|
Dissenting Shares
|
2.12(a)
|
|
Drug Regulatory Agency
|
3.14(b)
|
|
Effective Time
|
2.3
|
|
Employment-Related Laws
|
3.17(j)
|
|
End Date
|
10.1(b)
|
|
Exchange Agent
|
2.7(a)
|
|
FDA
|
3.14(b)
|
|
FDCA
|
3.14(c)
|
|
GAAP
|
3.7(a)
|
|
Intended Tax Treatment
|
2.10
|
12
|
Liability
|
3.9
|
|
Lock-Up Agreement
|
Recital
|
|
Lock-Up Agreements
|
Recital
|
|
Merger
|
Recital
|
|
Merger Consideration
|
2.5(a)(ii)
|
|
Merger Sub
|
Preamble
|
|
Ordinary Course Agreement
|
3.16(h)
|
|
Parent
|
Preamble
|
|
Parent 409A Plan
|
4.17(j)
|
|
Parent Board Recommendation
|
6.3(b)
|
|
Parent Disclosure Letter
|
Section 4
|
|
Parent Material Contract
|
4.13(a)
|
|
Parent Material Contracts
|
4.13(a)
|
|
Parent Permits
|
4.14(b)
|
|
Parent Products
|
4.14(d)
|
|
Parent Product Candidates
|
4.14(d)
|
|
Parent Real Estate Leases
|
4.11
|
|
Parent Regulatory Permits
|
4.14(d)
|
|
Parent SEC Documents
|
4.7(a)
|
|
Parent Stock Plan
|
4.6(c)
|
|
Parent Stockholder Meeting
|
6.3(a)
|
|
PHSA
|
3.14(c)
|
|
Pre-Closing Period
|
5.1(a)
|
|
Privacy Policies
|
3.22
|
|
Proxy Statement
|
6.1(a)
|
|
Registration Rights Agreement
|
Recital
|
|
Required Company Stockholder Vote
|
3.4
|
|
SEC Documents
|
6.16
|
|
Service Provider Grants
|
1.1
|
|
Stockholder Notice
|
6.2(b)
|
|
Surviving Entity
|
2.1
|
|
Transaction Litigation
|
6.4(c)
|
|
WARN Act
|
3.17(j)
|
13
1.2 Other Definitional and Interpretative Provisions. The words “hereof,” “herein” and “hereunder” and words of like import used in this
Agreement shall refer to this Agreement as a whole and not to any particular provision of this Agreement. The captions herein are included for convenience of reference only and shall be ignored in the construction or interpretation hereof.
References to Sections, Exhibits and Schedules are to Sections, Exhibits and Schedules of this Agreement unless otherwise specified. Any capitalized terms used in any Exhibit or Schedule but not otherwise defined therein shall have the meaning as
defined in this Agreement. Any singular term in this Agreement shall be deemed to include the plural, and any plural term the singular, the masculine gender shall include the feminine and neuter genders; the feminine gender shall include the
masculine and neuter genders; and the neuter gender shall include masculine and feminine gender. Whenever the words “include,” “includes” or “including” are used in this Agreement, they shall be deemed to be followed by the words “without
limitation,” whether or not they are in fact followed by those words or words of like import. The word “or” is not exclusive. “Writing,” “written” and comparable terms refer to printing, typing and other means of reproducing words (including
electronic media) in a visible form. References to any agreement or Contract (except for references to any agreements or Contracts listed on the Parent Disclosure Letter or Company Disclosure Letter) are to that agreement or Contract as amended,
modified or supplemented from time to time in accordance with the terms hereof and thereof. The Exhibits to this Agreement, the Parent Disclosure Letter and the Company Disclosure Letter are integral parts of the interpretation of this Agreement,
but only Exhibit D is incorporated by reference and made a part hereof for purposes of Section 251 of the DGCL. References to any Person include the successors and permitted assigns of that Person. References to any statute are to that
statute and to the rules and regulations promulgated thereunder, in each case as amended, modified, re-enacted thereof, substituted, from time to time. References to “$” and “dollars” are to the currency of the United States. All accounting terms
used herein will be interpreted, and all accounting determinations hereunder will be made, in accordance with GAAP unless otherwise expressly specified. References from or through any date shall mean, unless otherwise specified, from and including
or through and including, respectively. All references to “days” shall be to calendar days unless otherwise indicated as a “Business Day.” Except as otherwise specifically indicated, for purposes of measuring the beginning and ending of time
periods in this Agreement (including for purposes of “Business Day” and for hours in a day or Business Day), the time at which a thing, occurrence or event shall begin or end shall be deemed to occur in the Eastern time zone of the United States.
The Parties agree that any rule of construction to the effect that ambiguities are to be resolved against the drafting Party shall not be applied in the construction or interpretation of this Agreement. The Parties agree that the Company Disclosure
Letter and the Parent Disclosure Letter shall be arranged in sections and subsections corresponding to the numbered and lettered sections and subsections contained in Section 3 and Section 4, respectively. The disclosures in any
section or subsection of the Company Disclosure Letter or the Parent Disclosure Letter shall qualify other sections and subsections in Section 3 or Section 4, respectively, to the extent it is readily apparent from a reading of the
disclosure that such disclosure is applicable to such other sections and subsections. The words “delivered” or “made available” mean, with respect to any documentation, that prior to 5:00 p.m. (New York City time) on the date that is the day prior
to the date of this Agreement, a copy of such material has been (a) posted to and continuously made available by a Party to the other Party and its Representatives in the electronic data room maintained by such disclosing Party for the purposes of
the Contemplated Transactions or (b) delivered by or on behalf of a Party or its Representatives to the other Party or its Representatives via electronic mail or in hard copy form prior to the execution of this Agreement.
2.1 The Merger. Upon the terms and subject to the conditions set forth in this
Agreement, at the Effective Time, Merger Sub shall be merged with and into the Company, and the separate existence of Merger Sub shall cease. The Company will continue as the surviving entity in the Merger (the “Surviving
Entity”).
14
2.2 Effects of the Merger. The Merger shall have the effects set forth in this Agreement and in the applicable provisions of the DGCL. As a
result of the Merger, the Company will become a wholly owned subsidiary of Parent.
2.3 Closing; Effective Time. Unless this Agreement is earlier terminated pursuant to the provisions of Section 10.1, and subject to the satisfaction or waiver of the
conditions set forth in Section 6, Section 7 and Section 8, the consummation of the Merger (the “Closing”) shall take place remotely, as promptly as practicable (but in no event later
than the second Business Day following the satisfaction or waiver of the last to be satisfied or waived of the conditions set forth in Section 7, Section 8 and Section 9, other than those conditions that by their nature are
to be satisfied at the Closing, but subject to the satisfaction or waiver of each of such conditions), or at such other time, date and place as Parent and the Company may mutually agree in writing. The date on which the Closing actually takes place
is referred to as the “Closing Date.” Immediately prior to the Closing on the Closing Date, Parent shall file the Certificate of Designation with the office of the Secretary of State of the State of Delaware.
At the Closing, (i) the Parties shall cause the Merger to be consummated by executing and filing with the Secretary of State of the State of Delaware a certificate of merger with respect to the Merger, satisfying the applicable requirements of the
DGCL and in form and substance attached hereto as Exhibit D and incorporated herein by reference (the “Certificate of Merger”). The Merger shall become effective at the time of the filing of the
Certificate of Merger with the Secretary of State of the State of Delaware or at such later time as may be specified in the Certificate of Merger with the consent of Parent and the Company (the time as of which the Merger becomes effective being
referred to as the “Effective Time”).
(a) The certificate of incorporation of the Surviving Entity shall be amended and restated in the Merger to read as set forth on Exhibit A to the Certificate of Merger,
until thereafter amended as provided by the DGCL and such certificate of incorporation;
(b) The bylaws of the Surviving Entity shall be identical to the bylaws of the Company as in effect immediately prior to the Effective Time, until thereafter amended as provided
by the DGCL and such bylaws; and
(c) the directors and officers of the Surviving Entity, each to hold office in accordance with the certificate of incorporation and bylaws of the Surviving Entity, shall be such
persons as shall be mutually agreed upon by Parent and the Company.
(a) At the Effective Time, by virtue of the Merger and without any further action on the part of Parent, Merger Sub, the Company or any stockholder of
the Company or Parent:
(i) any shares of Company Capital Stock held as treasury stock immediately prior to the Effective Time shall be cancelled and
retired and shall cease to exist, and no consideration shall be delivered in exchange therefor; and
15
(ii) subject to Section 2.5(c), with respect to shares of Company Capital Stock held by Designated Holders, (A)
each share of Company Common Stock outstanding immediately prior to the Effective Time (excluding shares of Company Capital Stock to be cancelled pursuant to Section 2.5(a)(i) and excluding Dissenting Shares) shall be converted
solely into the right to receive a number of shares of Parent Convertible Preferred Stock equal to (x) the Exchange Ratio divided by (y) five (the “Designated Holder Common Stock Payment Shares”), and (B)
each share of Company Preferred Stock outstanding immediately prior to the Effective Time (excluding shares of Company Capital Stock to be cancelled pursuant to Section 2.5(a)(i) and excluding Dissenting Shares) shall be converted
solely into the right to receive a number of shares of Parent Convertible Preferred Stock equal to (x)(I) the number of shares of Company Common Stock issuable upon conversion of each share of Company Preferred Stock pursuant to the Company’s
certificate of incorporation and as set forth on Schedule 2.5(a)(ii) multiplied by (II) the Exchange Ratio, divided by (y) five (the “Designated Holder Preferred
Stock Payment Shares”);
(iii) subject to Section 2.5(c), with respect to shares of Company Capital Stock held by Non-Designated Holders, (A) each share of Company
Common Stock outstanding immediately prior to the Effective Time (excluding shares of Company Capital Stock to be cancelled pursuant to Section 2.5(a)(i) and excluding Dissenting Shares) shall be converted solely into the right to
receive a number of shares of Parent Common Stock equal to the Exchange Ratio, and (B) each share of Company Preferred Stock outstanding immediately prior to the Effective Time (excluding shares of Company Capital Stock to be cancelled pursuant to
Section 2.5(a)(i) and excluding Dissenting Shares) shall be converted solely into the right to receive a number of shares of Parent Common Stock equal to (x) the number of shares of Company Common Stock issuable upon conversion of
each share of Company Preferred Stock pursuant to the Company’s certificate of incorporation and as set forth on Schedule 2.5(a)(ii) multiplied by (y) the Exchange Ratio; provided, however, that, in
the event the aggregate number of shares of Parent Common Stock issuable to any Non-Designated Holder at Closing (including any shares of Parent Common Stock issuable in accordance with Section 6.5(a)) would result in the issuance of shares
of Parent Common Stock equal to or in excess of 19.9% of the outstanding shares of Parent Common Stock as of immediately before the Effective Time (the “Parent Common Stock Consideration Cap”), Parent shall
issue to any such Non-Designated Holders (x) shares of Parent Common Stock up to the Parent Common Stock Consideration Cap (such shares of Parent Common Stock, the “Non-Designated Holder Common Stock Payment Shares”),
and (y) in lieu of any shares of Parent Common Stock in excess of the Parent Common Stock Consideration Cap (such excess shares, the “Remaining Entitlement”), shares of Parent Convertible Preferred Stock
equal to (I) the Remaining Entitlement, divided by (II) five (the “Non-Designated Holder Preferred Stock Payment Shares,” and together with the Designated Holder Common Stock Payment Shares, the Designated
Holder Preferred Stock Payment Shares and the Non-Designated Holder Common Stock Payment Shares, the “Merger Consideration”), in each case of subclauses (ii) and (iii),
on a pro rata basis among holders of Company Capital Stock in such manner to provide any such holder of Company Capital Stock with the same economic effect as contemplated by this Agreement.
(b) If any shares of Company Capital Stock outstanding immediately prior to the Effective Time are unvested or are subject to a repurchase option or a risk of forfeiture under any
applicable restricted stock purchase agreement or other similar agreement with the Company, then the shares of Parent Capital Stock issued in exchange for such shares of Company Capital Stock will to the same extent be unvested and subject to the
same repurchase option or risk of forfeiture, and such shares of Parent Capital Stock shall accordingly be marked with appropriate legends. The Company shall take all actions that may be necessary to ensure that, from and after the Effective Time,
Parent is entitled to exercise any such repurchase option or other right set forth in any such restricted stock purchase agreement or other agreement.
16
(c) No fractional shares of Parent Capital Stock shall be issued in connection with the Merger, and no certificates or scrip for any such fractional
shares shall be issued. Any holder of Company Capital Stock who would otherwise be entitled to receive a fraction of a share of Parent Common Stock (after aggregating all fractional shares of Parent Common Stock issuable to such holder) shall
receive from Parent, in lieu of such fractional share and upon surrender by such holder of a letter of transmittal in accordance with Section 2.8 and any accompanying documents as required therein: (i) one share of Parent Common
Stock if the aggregate amount of fractional shares of Parent Common Stock such holder of Company Common Stock would otherwise be entitled to is equal to or exceeds 0.50; or (ii) no shares of Parent Common Stock if the aggregate amount of fractional
shares of Parent Common Stock such holder of Company Common Stock would otherwise be entitled to is less than 0.50, with no cash being paid for any fractional share eliminated by such rounding. Any fractional shares of Parent Preferred Stock that a
holder of Company Capital Stock would otherwise be entitled to receive shall be aggregated with all fractional shares of Parent Preferred Stock issuable to such and any remaining fractional shares shall be, in lieu of such fractional share and upon
surrender by such holder of a letter of transmittal in accordance with Section 2.8 and any accompanying documents as required therein, rounded up to the nearest whole share of Parent Preferred Stock.
(d) All Company RSUs outstanding immediately prior to the Effective Time shall be treated in accordance with Section 6.5(a), and all Company Options outstanding
immediately prior to the Effective Time shall be treated in accordance with Section 6.5(b).
(e) Each share of common stock, $0.001 par value per share, of Merger Sub issued and outstanding immediately prior to the Effective Time shall be converted into and exchanged for
one validly issued, fully paid and nonassessable share of common stock, $0.001 par value per share, of the Surviving Entity. Each book entry share of Merger Sub evidencing ownership of any such shares shall, as of the Effective Time, evidence
ownership of such shares of common stock of the Surviving Entity.
(f) If, between the date of this Agreement and the Effective Time, the outstanding Company Capital Stock or Parent Capital Stock shall have been changed into, or exchanged for, a
different number of shares or a different class, by reason of any stock dividend, subdivision, reclassification, recapitalization, split, combination or exchange of shares or other like change, the Exchange Ratio shall, to the extent necessary, be
equitably adjusted to reflect such change to the extent necessary to provide the holders of Company Capital Stock, Company Options and Parent Capital Stock with the same economic effect as contemplated by this Agreement prior to such stock
dividend, subdivision, reclassification, recapitalization, split, combination or exchange of shares or other like change; provided, however, that nothing herein will be construed to permit the Company or Parent to take any action
with respect to Company Capital Stock or Parent Capital Stock, respectively, that is prohibited or not expressly permitted by the terms of this Agreement.
(g) At the Effective Time, by virtue of the Merger and without any action on the part of Parent, the Surviving Entity or their respective stockholders, each share of the Surviving
Entity issued and outstanding immediately prior to the Effective Time shall be cancelled and extinguished without any conversion thereof and no payment or distribution shall be made with respect thereto.
2.6 Closing of the Company’s Transfer Books. At the Effective Time: (a) all Company Capital Stock outstanding immediately prior to the
Effective Time shall be treated in accordance with Section 2.5(a), and all holders of certificates representing Company Capital Stock that were outstanding immediately prior to the Effective Time shall cease to have any rights as
stockholders of the Company and (b) the stock transfer books of the Company shall be closed with respect to all Company Capital Stock outstanding immediately prior to the Effective Time. No further transfer of any such Company Capital Stock shall
be made on such stock transfer books after the Effective Time.
17
(a) On or prior to the Closing Date, Parent and the Company shall jointly select a reputable bank, transfer agent or trust company to act as exchange
agent in the Merger (the “Exchange Agent”). At the Effective Time, Parent shall deposit with the Exchange Agent evidence of book-entry shares representing the shares of Parent Capital Stock issuable pursuant
to Section 2.5(a) in exchange for Company Capital Stock.
(b) Promptly after the Effective Time, the Parties shall cause the Exchange Agent to mail to the Persons who were record holders of shares of Company
Capital Stock that were converted into the right to receive the Merger Consideration: (i) a letter of transmittal in customary form and containing such provisions as Parent may reasonably specify (including a provision confirming that delivery of
physical stock certificates representing shares of Company Capital Stock, (the “Company Stock Certificates”) shall be effected, and risk of loss and title shall pass, only upon delivery of such Company Stock
Certificates to the Exchange Agent) and (ii) instructions for effecting the surrender of Company Stock Certificates, or uncertificated shares of Company Capital Stock, in exchange for book-entry shares of Parent Capital Stock. Upon surrender of a
Company Stock Certificate or other reasonable evidence of the ownership of uncertificated Company Capital Stock to the Exchange Agent for exchange, together with a duly executed letter of transmittal and such other documents as may be reasonably
required by the Exchange Agent or Parent: (A) the holder of such Company Stock Certificate or uncertificated shares of Company Capital Stock shall be entitled to receive in exchange therefor book-entry shares representing the Merger Consideration
(in a number of whole shares of Parent Capital Stock) that such holder has the right to receive pursuant to the provisions of Section 2.5(a) and Section 2.5(c) and (B) the Company Stock Certificate or uncertificated
shares of Company Capital Stock so surrendered shall be cancelled. Until surrendered as contemplated by this Section 2.7(b), each Company Stock Certificate or uncertificated shares of Company Capital Stock shall be deemed, from and
after the Effective Time, to represent only the right to receive book-entry shares of Parent Capital Stock representing the Merger Consideration. If any Company Stock Certificate shall have been lost, stolen or destroyed, Parent may, in its
discretion and as a condition precedent to the delivery of any shares of Parent Capital Stock, require the owner of such lost, stolen or destroyed Company Stock Certificate to provide an applicable affidavit with respect to such Company Stock
Certificate and post a bond indemnifying Parent against any claim suffered by Parent related to the lost, stolen or destroyed Company Stock Certificate or any Parent Capital Stock issued in exchange therefor as Parent may reasonably request.
(c) No dividends or other distributions declared or made with respect to Parent Capital Stock with a record date after the Effective Time shall be paid to the holder of any
unsurrendered Company Stock Certificate with respect to the shares of Parent Capital Stock that such holder has the right to receive in the Merger until such holder surrenders such Company Stock Certificate or uncertificated shares of Company
Capital Stock or provides an affidavit of loss or destruction in lieu thereof in accordance with this Section 2.7 (at which time such holder shall be entitled, subject to the effect of applicable abandoned property, escheat or
similar Laws, to receive all such dividends and distributions, without interest).
(d) Any shares of Parent Capital Stock deposited with the Exchange Agent that remain undistributed to holders of Company Stock Certificates as of the date that is 180 days after
the Closing Date shall be delivered to Parent upon demand, and any holders of Company Stock Certificates who have not theretofore surrendered their Company Stock Certificates or uncertificated shares of Company Capital Stock in accordance with this
Section 2.7 shall thereafter look only to Parent for satisfaction of their claims for Parent Capital Stock and any dividends or distributions with respect to shares of Parent Capital Stock.
(e) No Person shall be liable to any holder of any Company Stock Certificate or uncertificated shares of Company Capital Stock or to any other Person with respect to any shares of
Parent Capital Stock (or dividends or distributions with respect thereto) or for any cash amounts delivered to any public official pursuant to any applicable abandoned property Law, escheat Law or similar Law.
18
2.9 Further Action. If, at any time after the Effective Time, any further action is determined by the Surviving Entity to be necessary or
desirable to carry out the purposes of this Agreement or to vest the Surviving Entity with full right, title and possession of and to all rights and property of the Company, then the officers and directors of the Surviving Entity shall be fully
authorized, and shall use their and its commercially reasonable efforts (in the name of the Company, in the name of Merger Sub, in the name of the Surviving Entity and otherwise) to take such action.
2.10 Intended Tax Treatment. The Parties acknowledge and agree that, for U.S. federal (and applicable state and
local) income Tax purposes, the Merger is intended to qualify as a reorganization pursuant to Section 368(a) of the Code (the “Intended Tax Treatment”). The Parties adopt this Agreement as a “plan of
reorganization” within the meaning of Treasury Regulations Sections 1.368-2(g) and 1.368-3.
2.11 Withholding. Each of the Exchange Agent, Parent and the Surviving Entity shall be entitled to deduct and withhold from any consideration
deliverable pursuant to this Agreement to any Person such amounts as are required to be deducted or withheld from such consideration under applicable Law; provided that the Exchange Agent, Parent and the Surviving Entity shall use
commercially reasonable efforts to promptly notify such Persons of any intention to withhold any portion of such consideration and cooperate with such Persons to reduce or eliminate any such withholding to the extent permitted by applicable Law. To
the extent such amounts are so deducted or withheld and remitted to the appropriate Governmental Authority, such amounts shall be treated for all purposes under this Agreement as having been paid to the Person to whom such amounts would otherwise
have been paid. All payments made under this agreement that constitute compensation to employees for services for Tax purposes shall be made through the payroll of the Surviving Entity or Parent, as applicable.
(a) Notwithstanding any provision of this Agreement to the contrary, shares of Company Capital Stock that are outstanding immediately prior to the
Effective Time and which are held by stockholders or owned by beneficial owners who have exercised and perfected appraisal rights for such shares of Company Capital Stock in accordance with the DGCL (collectively, the “Dissenting Shares”) shall not be converted into or represent the right to receive the Merger Consideration described in Section 2.5 attributable to such Dissenting Shares. Such stockholders or beneficial owners shall
be entitled to receive payment of the fair value of such shares of Company Capital Stock held by them in accordance with the DGCL, unless and until such stockholders or beneficial owners fail to perfect or effectively withdraw or otherwise lose
their appraisal rights under the DGCL. All Dissenting Shares held by stockholders or owned by beneficial owners who shall have failed to perfect or shall have effectively withdrawn or lost their right to appraisal of such shares of Company Capital
Stock under the DGCL (whether occurring before, at or after the Effective Time) shall thereupon be deemed to be converted into and to have become exchangeable for, as of the Effective Time, the right to receive the Merger Consideration, without
interest, attributable to such Dissenting Shares upon their surrender in the manner provided in Sections 2.5 and 2.7.
(b) The Company shall give Parent prompt written notice of any demands by dissenting stockholders or beneficial owners received by the Company, withdrawals of such demands and any
other instruments served on the Company and any material correspondence received by the Company in connection with such demands, and Parent shall have the right to participate in all negotiations and proceedings with respect to such demands. The
Company shall not, except with Parent’s prior written consent, not to be unreasonably withheld, delayed or conditioned, make any payment with respect to, or settle or offer to settle, any such demands, or approve any withdrawal of any such demands
or agree to do any of the foregoing.
19
Except as set forth in the written disclosure document delivered by the Company to Parent (the “Company Disclosure Letter”) concurrently
with the execution of this Agreement, the Company represents and warrants to Parent and Merger Sub as follows:
(a) The Company and each of its Subsidiaries is a corporation or other legal entity duly incorporated or otherwise organized, validly existing and in
good standing under the Laws of the jurisdiction of its incorporation or organization and has all necessary power and authority: (i) to conduct its business in the manner in which its business is currently being conducted, (ii) to own or lease and
use its property and assets in the manner in which its property and assets are currently owned or leased and used and (iii) to perform its obligations under all Contracts by which it is bound.
(b) The Company and each of its Subsidiaries is duly licensed and qualified to do business, and is in good standing (to the extent applicable in such jurisdiction), under the Laws
of all jurisdictions where the nature of its business in the manner in which its business is currently being conducted requires such licensing or qualification other than in jurisdictions where the failure to be so qualified individually or in the
aggregate would not be reasonably expected to have a Company Material Adverse Effect.
(c) The Company has no Subsidiaries except as set forth on Section 3.1(c) of the Company Disclosure Letter The Company is not and has
never otherwise been, directly or indirectly, a party to, member of or participant in any partnership, joint venture or similar business entity. The Company has not agreed or is obligated to make, or is bound by any Contract under which it may
become obligated to make, any future investment in or capital contribution to any other Entity. The Company has not, at any time, been a general partner of, or has otherwise been liable for any of the debts or other obligations of, any general
partnership, limited partnership or other Entity.
3.2 Organizational Documents. The Company has delivered to Parent accurate and complete copies of the
Organizational Documents of the Company. The Company is not in breach or violation of its Organizational Documents in any material respect.
3.3 Authority; Binding Nature of Agreement. The Company has all necessary corporate power and authority to enter
into and to perform its obligations under this Agreement and to consummate the Contemplated Transactions. The Company Board has (i) determined that the Contemplated Transactions are fair to, advisable and in the best interests of the Company and
its stockholders, (ii) approved and declared advisable this Agreement and the Contemplated Transactions and (iii) determined to recommend, upon the terms and subject to the conditions set forth in this Agreement, that the stockholders of the
Company vote to adopt this Agreement and thereby approve the Contemplated Transactions. This Agreement has been duly executed and delivered by the Company and assuming the due authorization, execution and delivery by Parent and Merger Sub,
constitutes the legal, valid and binding obligation of the Company, enforceable against the Company in accordance with its terms, subject to the Enforceability Exceptions.
20
3.4 Vote Required. The affirmative vote (or written
consent) of (i) the holders of at least a majority of the outstanding shares of Company Series C Preferred Stock, as a class, (ii) the holders of at least sixty-seven percent (67%) of the outstanding shares of Company Series B Preferred Stock, as a
class, (iii) the holders of at least a majority of the outstanding shares of Company Series A Preferred Stock, as a class, and (iv) a majority of the outstanding Company Capital Stock entitled to vote thereon, as a class (collectively, the “Required Company Stockholder Vote”) is the only vote of the holders of any class or series of Company Capital Stock necessary to adopt and approve this Agreement and approve the Contemplated Transactions.
(a) Subject to obtaining the Required Company Stockholder Vote, compliance with any applicable requirements of the HSR Act, and the filing of the Certificate of Merger required by
the DGCL, neither (x) the execution, delivery or performance of this Agreement by the Company, nor (y) the consummation of the Contemplated Transactions, will directly or indirectly (with or without notice or lapse of time):
(i) contravene, conflict with or result in a violation of any of the provisions of the Company’s Organizational Documents;
(ii) contravene, conflict with or result in a material violation of, or give any Governmental Authority or other Person the right to challenge the Contemplated
Transactions or to exercise any remedy or obtain any relief under, any Law or any Order by which the Company, or any of the assets owned or used by the Company, is subject;
(iii) contravene, conflict with or result in a material violation of any of the terms or requirements of, or give any Governmental Authority the right to
revoke, withdraw, suspend, cancel, terminate or modify, any Governmental Authorization that is held by the Company or that otherwise relates to the business of the Company, or any of the assets owned, leased or used by the Company;
(iv) contravene, conflict with or result in a violation or breach of, or result in a default under, any provision of any Company Material Contract, or give any
Person the right to: (A) declare a default or exercise any remedy under any Company Material Contract, (B) any material payment, rebate, chargeback, penalty or change in delivery schedule under any Company Material Contract, (C) accelerate the
maturity or performance of any Company Material Contract or (D) cancel, terminate or modify any term of any Company Material Contract, except in the case of any nonmaterial breach, default, penalty or modification; or
(v) result in the imposition or creation of any Encumbrance upon or with respect to any asset owned or used by the Company (except for Permitted Encumbrances).
(b) Except for (i) the Required Company Stockholder Vote, (ii) the filing of the Certificate of Merger with the Secretary of State of the State of Delaware pursuant to the DGCL,
(iii) compliance with any applicable requirements of the HSR Act, and (iv) such consents, waivers, approvals, orders, authorizations, registrations, declarations and filings as may be required under applicable federal and state securities laws, the
Company was not, is not, nor will be required to make any filing with or give any notice to, or to obtain any Consent from, any Person in connection with (x) the execution, delivery or performance of this Agreement or (y) the consummation of the
Contemplated Transactions.
21
(c) No state takeover statute or similar Law applies or purports to apply to the Merger, this Agreement, the Company Stockholder Support Agreements or any of the Contemplated
Transactions.
(a) The authorized capital stock of the Company consists of (i) 143,329,269 shares of Company Common Stock of which 10,023,615 shares have been issued
and are outstanding as of the date hereof and (ii) 57,821,355 shares of Company Preferred Stock, of which, as of the date hereof, 10,000,000 shares have been designated Series Seed-1 Preferred Stock, all of which have been issued and are
outstanding, 5,000,000 shares have been designated Series Seed-2 Preferred Stock, all of which have been issued and are outstanding, 9,411,765 shares have been designated Series A Preferred Stock, all of which have been issued and are outstanding,
20,080,321 shares have been designated Series B Preferred Stock, all of which have been issued and are outstanding, and 13,329,269 shares have been designated Series C Preferred Stock, all of which have been issued and are outstanding. The Company
does not hold any shares of its capital stock in its treasury. As of the date of this Agreement, the Company’s capital stock is held by the Persons and in the amounts set forth in Section 3.6(a) of the Company Disclosure Letter,
which further sets forth for each such Person (i) the name of such Person and the number of shares held, (ii) the class and series of such shares, (iii) the number of the applicable book-entry positions representing such shares or the number of the
certificate representing such shares, (iv) whether such Person is or has ever been an employee, and (v) the state of residence of such Person. Each share of Company Preferred Stock is convertible into one share of Company Common Stock. There are no
declared or accrued but unpaid dividends with respect to any shares of the Company’s capital stock and the Company has never declared or paid any dividend or other distribution.
(b) All of the outstanding Company Capital Stock as set out in Section 3.6(a) of the Company Disclosure Letter have been duly
authorized and validly issued, and are fully paid and nonassessable and are free of any Encumbrances other than Encumbrances set forth in the Organizational Documents or under applicable securities Laws. None of the outstanding Company Capital
Stock is entitled or subject to any preemptive right, right of participation, right of maintenance or any similar right and none of the outstanding Company Capital Stock is subject to any right of first refusal in favor of the Company. Except as
contemplated herein, there is no Company Contract relating to the voting or registration of, or restricting any Person from purchasing, selling, pledging or otherwise disposing of (or granting any option or similar right with respect to), any
Company Capital Stock. The Company is not under any obligation, nor is it bound by any Contract pursuant to which it may become obligated, to repurchase, redeem or otherwise acquire any outstanding Company Capital Stock or other securities. Section
3.6(b) of the Company Disclosure Letter accurately and completely describes all repurchase rights held by the Company with respect to Company Capital Stock (including shares issued pursuant to the exercise of stock options) and specifies
which of those repurchase rights are currently exercisable.
(c) Except for the Company Stock Plans and except as set forth on Section 3.6(c) of the Company Disclosure Letter, the Company does
not have any stock option plan or any other plan, program, agreement or arrangement providing for any equity-based compensation for any Person. Section 3.6(c) of the Company Disclosure Letter sets forth the following information
with respect to each Company Option outstanding as of the date hereof: (i) the name of the holder, (ii) the number of shares of Company Common Stock subject to such Company Option as of the date hereof, (iii) the exercise price of such Company
Option, (iv) the date on which such Company Option was granted, (v) the applicable vesting schedule, including any acceleration provisions, (vi) the date on which such Company Option expires, and (vii) whether such Company Option is intended to be
an “incentive stock option” (as defined in the Code) or a nonqualified stock option. The Company has made available to Parent accurate and complete copies of equity incentive plans pursuant to which the Company has equity-based awards, the forms of
all award agreements evidencing such equity-based awards and evidence of board and stockholder approval of the Company Stock Plan and any amendments thereto.
22
(d) Except for the outstanding Company Options or Company Options and any other equity awards issued under the Company Stock Plan (including any
shares of Company Common Stock issuable upon the exercise of such Company Options or other equity awards) to directors, employees, consultants or other service providers following the date hereof but prior to the Closing (collectively, the “Service Provider Grants”), there is no: (i) outstanding subscription, option, call, warrant or right (whether or not currently exercisable) to acquire any Company Capital Stock or other securities of the Company,
(ii) outstanding security, instrument or obligation that is or may become convertible into or exchangeable for any shares of the capital stock or other securities of the Company, (iii) stockholder rights plan (or similar plan commonly referred to
as a “poison pill”) or Contract under which the Company is or may become obligated to sell or otherwise issue any Company Capital Stock or any other securities or (iv) condition or circumstance that may give rise to or provide a basis for the
assertion of a claim by any Person to the effect that such Person is entitled to acquire or receive any shares of capital stock or other securities of the Company. There are no outstanding or authorized stock appreciation, phantom stock, profit
participation or other similar rights with respect to the Company.
(e) All outstanding Company Capital Stock, Company Options and other securities of the Company have been issued and granted in compliance in all material respects with (i) all
applicable securities laws and other applicable Law and (ii) all requirements set forth in applicable Contracts.
(f) The Company Capital Stock are uncertificated.
(a) Section 3.7(a) of the Company Disclosure Letter includes true and complete copies of the Company’s unaudited balance sheets at
December 31, 2024 (the “Company Balance Sheet”), together with related unaudited statements of operations, changes in stockholders’ equity and cash flows, and notes thereto, of the Company for the fiscal year
then ended (collectively, the “Company Financials”). The Company Financials (i) were prepared in accordance with United States generally accepted accounting principles (“GAAP”)
(except that the Company Financials may not have notes thereto and other presentation items that may be required by GAAP and are subject to normal and recurring year-end adjustments that are not reasonably expected to be material in amount) applied
on a consistent basis unless otherwise noted therein throughout the periods indicated and (ii) fairly present, in all material respects, the financial position and operating results of the Company as of the dates and for the periods indicated
therein.
(b) The Company maintains a system of internal accounting controls designed to provide reasonable assurance that: (i) transactions are executed in accordance with management’s
general or specific authorizations, (ii) transactions are recorded as necessary to permit preparation of the financial statements of the Company in conformity with GAAP and to maintain accountability of the Company’s assets, (iii) access to the
Company’s assets is permitted only in accordance with management’s general or specific authorization and (iv) the recorded accountability for the Company’s assets is compared with the existing assets at regular intervals and appropriate action is
taken with respect to any differences. The Company maintains internal controls consistent with the practices of similarly situated private companies over financial reporting that provides reasonable assurance regarding the reliability of financial
reporting and the preparation of financial statements for external purposes.
(c) Section 3.7(c) of the Company Disclosure Letter lists, and the Company has delivered to Parent accurate and complete copies of the
documentation creating or governing, all securitization transactions and “off-balance sheet arrangements” (as defined in Item 303(c) of Regulation S-K under the Exchange Act) effected by the Company.
23
(d) There have been no formal internal investigations regarding financial reporting or accounting policies and practices discussed with, reviewed by or initiated at the direction
of the chief executive officer or chief financial officer of the Company, the Company Board or any committee thereof. Neither the Company nor its independent auditors have identified (i) any significant deficiency or material weakness in the design
or operation of the system of internal accounting controls utilized by the Company, (ii) any fraud, whether or not material, that involves the Company, the Company’s management or other employees who have a role in the preparation of financial
statements or the internal accounting controls utilized by the Company or (iii) any claim or allegation regarding any of the foregoing.
3.8 Absence of Changes. Except as set forth on Section 3.8 of the Company Disclosure Letter,
between December 31, 2024 and the date of this Agreement, the Company and its Subsidiaries has conducted its business only in the Ordinary Course of Business (except for the execution and performance of this Agreement and the discussions,
negotiations and transactions related thereto) and there has not been any (a) Company Material Adverse Effect or (b) action, event or occurrence that would have required consent of Parent pursuant to Section 5.2(b) of this Agreement
had such action, event or occurrence taken place after the execution and delivery of this Agreement.
3.9 Absence of Undisclosed Liabilities. Since December 31, 2024, neither the Company
nor any of its Subsidiaries has any liability, indebtedness, obligation, expense, claim, deficiency, guaranty or endorsement of any kind, whether accrued, absolute, contingent, matured, unmatured or otherwise (each a “Liability”), except for: (a) Liabilities disclosed, reflected or reserved against in the Company Financials, (b) normal and recurring current Liabilities that have been incurred by the Company since the date of the Company Balance
Sheet in the Ordinary Course of Business (none of which relates to any breach of contract, breach of warranty, tort, infringement or violation of Law), (c) Liabilities for performance of obligations of the Company under Company Contracts, (d)
Liabilities incurred in connection with the Contemplated Transactions, and (e) those Liabilities that are not material to the Company.
3.10 Title to Assets. The Company owns and has good and valid title to, or, in the case of leased properties and assets, valid leasehold
interests in, all tangible properties or tangible assets and equipment used or held for use in its business or operations or purported to be owned by it, including: (a) all tangible assets reflected on the Company Balance Sheet and (b) all other
tangible assets reflected in the books and records of the Company as being owned by the Company. All of such assets are owned or, in the case of leased assets, leased by the Company free and clear of any Encumbrances, other than Permitted
Encumbrances.
3.11 Real Property; Leasehold. The Company does not own and has never owned any real property, nor is the Company
party to any agreement to purchase or sell any real property. The Company has made available to Parent (a) an accurate and complete list of all real properties with respect to which the Company directly or indirectly holds a valid leasehold
interest as well as any other real estate that is in the possession of or leased by the Company and (b) copies of all leases under which any such real property is possessed (the “Company Real Estate Leases”),
each of which is in full force and effect, with no existing material default thereunder by the Company or to the Company’s Knowledge, the other party thereto.
(a) Section 3.12(a) of the Company Disclosure Letter is an accurate, true and complete listing of all Company Registered IP.
24
(b) Section 3.12(b) of the Company Disclosure Letter accurately identifies (i) all Company Contracts pursuant to which any Company IP
Rights are licensed to the Company (other than (A) any non-customized software that (1) is so licensed solely in executable or object code form pursuant to a nonexclusive, internal use software license and other Intellectual Property associated
with such software and (2) is not incorporated into, or material to the development, manufacturing or distribution of, any of the Company’s products or services, (B) any Intellectual Property licensed on a nonexclusive basis ancillary to the
purchase or use of services, equipment, reagents or other materials, (C) any confidential information provided under confidentiality agreements and (D) agreements between Company and its employees in Company’s standard form thereof) and (ii)
whether the license or licenses granted to the Company are exclusive or nonexclusive.
(c) Section 3.12(c) of the Company Disclosure Letter accurately identifies each Company Contract pursuant to which any Person has been
granted any license or covenant not to sue under, or otherwise has received or acquired any right (whether or not currently exercisable) or interest in, any Company IP Rights (other than (i) any confidential information provided under
confidentiality agreements and (ii) any Company IP Rights nonexclusively licensed to academic collaborators, suppliers or service providers for the sole purpose of enabling such academic collaborator, supplier or service providers to provide
services for the Company’s benefit).
(d) The Company is not bound by, and no Company IP Rights are subject to, any Contract containing any covenant or other provision that in any way limits or restricts the ability
of the Company to use, exploit, assert or enforce any Company IP Rights anywhere in the world.
(e) The Company exclusively owns all right, title and interest to and in Company IP Rights (other than (i) Company IP Rights licensed to the Company,
or co-owned rights each as identified in Section 3.12(e) of the Company Disclosure Letter, (ii) any non-customized software that (A) is licensed to the Company solely in executable or object code form pursuant to a nonexclusive,
internal use software license and other Intellectual Property associated with such software and (B) is not incorporated into, or material to the development, manufacturing or distribution of, any of the Company’s products or services and (iii) any
Intellectual Property licensed on a nonexclusive basis ancillary to the purchase or use of equipment, reagents or other materials), in each case, free and clear of any Encumbrances (other than Permitted Encumbrances). Without limiting the
generality of the foregoing:
(i) All documents and instruments necessary to register or apply for or renew registration of Company Registered IP have been validly executed, delivered and
filed in a timely manner with the appropriate Governmental Authority.
(ii) Each Person who is or was an employee or contractor of the Company and who is or was involved in the creation or development of any Intellectual Property
for the Company has signed a valid, enforceable agreement containing a present assignment of such Intellectual Property to the Company and confidentiality provisions protecting trade secrets and confidential information of the Company.
(iii) To the Knowledge of the Company, no current or former stockholder, officer, director or employee of the Company has any claim, right (whether currently
exercisable, or exercisable in the future) or interest to or in any Company IP Rights purported to be owned by the Company. To the Knowledge of the Company, no employee of the Company is (a) bound by or otherwise subject to any Contract restricting
him or her from performing his or her duties for the Company or (b) in breach of any Contract with any former employer or other Person concerning Company IP Rights purported to be owned by the Company or confidentiality provisions protecting trade
secrets and confidential information comprising Company IP Rights purported to be owned by the Company.
25
(iv) No funding, facilities or personnel of any Governmental Authority or any university, college, research institute or other educational institution were
used, directly or indirectly, to develop or create, in whole or in part, any Company IP Rights in which the Company has an ownership interest, except for any such funding or use of facilities or personnel that does not result in such Governmental
Authority or institution owning such Company IP Rights or the right to receive royalties or other remuneration for the practice of such Company IP Rights as of the date of this Agreement.
(v) The Company has taken reasonable steps to maintain the confidentiality of and otherwise protect and enforce its rights in all proprietary information that
the Company holds, or purports to hold, as confidential or a trade secret.
(vi) The Company has not assigned or otherwise transferred ownership of, or agreed to assign or otherwise transfer ownership of, any Company IP Rights to any
other Person.
(f) The Company has delivered or made available to Parent, a complete and accurate copy of all Company IP Rights Agreements. With respect to each of the Company IP Rights
Agreements: (i) each such agreement is valid and binding on the Company and in full force and effect, (ii) the Company has not received any written notice of termination or cancellation under such agreement, or received any written notice of breach
or default under such agreement, which breach has not been cured or waived and (iii) the Company, and to the Knowledge of the Company, no other party to any such agreement, is not in breach or default thereof in any material respect.
(g) The manufacture, marketing, offering for sale, sale, importation, use or intended use or other disposal of any product as currently sold or under development by the Company
does not violate any license or agreement between the Company and any other third party, and, to the Knowledge of the Company, does not infringe or misappropriate any valid and issued Patent right or other Intellectual Property of any other Person,
which infringement or misappropriation would reasonably be expected to have a Company Material Adverse Effect. To the Knowledge of the Company, no third party is infringing upon any Patents owned by Company within the Company IP Rights, or
otherwise violating any Company IP Rights Agreement.
(h) As of the date of this Agreement, Company is not a party to any Legal Proceeding (including, but not limited to, opposition, interference or other proceeding in any patent or
other government office) contesting the validity, enforceability, claim construction, ownership or right to use, sell, offer for sale, license or dispose of any Company IP Rights. The Company has not received any written notice asserting that any
Company IP Rights or the proposed use, sale, offer for sale, license or disposition of products, methods or processes claimed or covered thereunder infringes or misappropriates or violates the rights of any other Person or that the Company has
otherwise infringed, misappropriated or otherwise violated any Intellectual Property of any Person. None of the Company IP Rights is subject to any outstanding order of, judgment of, decree of or agreement with any Governmental Authority that
limits the ability of the Company to exploit any Company IP Rights.
(i) Each item of Company Registered IP is and at all times has been filed and maintained in compliance in all material respects with all applicable Law and all filings, payments
and other actions required to be made or taken to maintain such item of Company Registered IP in full force and effect have been made by the applicable deadline. To the Knowledge of the Company, all Company Registered IP that is issued or granted
is valid and enforceable.
26
(j) To the Knowledge of the Company, no trademark (whether registered or unregistered) or trade name owned, used or applied for by the Company conflicts or interferes with any
trademark (whether registered or unregistered) or trade name owned, used or applied for by any other Person. None of the goodwill associated with or inherent in any trademark (whether registered or unregistered) in which the Company has or purports
to have an ownership interest has been impaired as determined by the Company in accordance with GAAP.
(k) Except as set forth in Sections 3.12(b), 3.12(c) or 3.12(k) of the Company Disclosure Letter or as contained in
“off-the-shelf” license agreements entered into in the Ordinary Course of Business by the Company, (i) the Company is not bound by any Contract to indemnify, defend, hold harmless or reimburse any other Person with respect to any Intellectual
Property infringement, misappropriation, or similar claim which is material to the Company, taken as a whole and (ii) the Company has never assumed, or agreed to discharge or otherwise take responsibility for, any existing or potential liability of
another Person for infringement, misappropriation, or violation of any Intellectual Property right, which assumption, agreement or responsibility remains in force as of the date of this Agreement.
(l) The Company is not party to any Contract that, as a result of such execution, delivery and performance of this Agreement, will cause the grant of any license or other right to
any Company IP Rights, result in breach of, default under or termination of such Contract with respect to any Company IP Rights, or impair the right of the Company or the Surviving Entity and its Subsidiaries to use, sell or license or enforce any
Company IP Rights or portion thereof, except for the occurrence of any such grant or impairment that would not individually or in the aggregate, reasonably be expected to result in a Company Material Adverse Effect.
(a) Section 3.13(a) of the Company Disclosure Letter lists the following Company Contracts in effect as of the date of this Agreement
(each, a “Company Material Contract” and collectively, the “Company Material Contracts”):
(i) each Company Contract relating to any agreement of indemnification or guaranty not entered into in the Ordinary Course of Business;
(ii) each Company Contract containing (A) any covenant limiting the freedom of the Company or the Surviving Entity to engage in any line of business or compete
with any Person, or limiting the development, manufacture or distribution of the Company’s products or services (B) any most-favored pricing arrangement, (C) any exclusivity provision or (D) any non-solicitation provision;
(iii) each Company Contract (A) pursuant to which any Person granted the Company an exclusive license under any Intellectual Property, or (B) pursuant to which
the Company granted any Person an exclusive license under any Company IP Rights;
(iv) each Company Contract relating to capital expenditures and requiring payments after the date of this Agreement in excess of $100,000 pursuant to its
express terms and not cancelable without penalty;
(v) each Company Contract containing any royalty, dividend or similar arrangement based on the revenues or profits of the Company, any of its Subsidiaries, or
of a product;
27
(vi) each Company Contract relating to the disposition or acquisition of material assets or any ownership interest in any Entity, in each case, involving
payments in excess of $100,000 after the date of this Agreement;
(vii) each Company Contract relating to any mortgages, indentures, loans, notes or credit agreements, security agreements or other agreements or instruments
relating to the borrowing of money or extension of credit in excess of $100,000 or creating any material Encumbrances with respect to any assets of the Company or any loans or debt obligations with officers or directors of the Company;
(viii) each Company Contract requiring payment by or to the Company after the date of this Agreement in excess of $100,000 pursuant to its express terms
relating to: (A) any distribution agreement (identifying any that contain exclusivity provisions), (B) any agreement involving provision of services or products with respect to any pre-clinical or clinical development activities of the Company, (C)
any dealer, distributor, joint marketing, alliance, joint venture, cooperation, development or other agreement currently in force under which the Company has continuing obligations to develop or market any product, technology or service, or any
agreement pursuant to which the Company has continuing obligations to develop any Intellectual Property that will not be owned, in whole or in part, by the Company or (D) any Contract to license any patent, trademark registration, service mark
registration, trade name or copyright registration to or from any third party to manufacture or produce any product, service or technology of the Company or any Contract to sell, distribute or commercialize any products or service of the Company,
in each case, except for Company Contracts entered into in the Ordinary Course of Business;
(ix) each Company Contract with any Person, including any financial advisor, broker, finder, investment banker or other Person, providing advisory services to
the Company in connection with the Contemplated Transactions and requiring payments by Company after the date in this Agreement in excess of $100,000 pursuant to its express terms;
(x) each Company Contract to which the Company is a party or by which any of its assets and properties is currently bound, which involves annual obligations of
payment by, or annual payments to, the Company in excess of $100,000;
(xi) each Company Contract entered into in settlement of any Legal Proceeding or other dispute pursuant to which the Company or any of its Subsidiaries has
outstanding obligations to pay consideration in excess of $100,000;
(xii) any other Company Contract that is not terminable at will (with no penalty or payment) by the Company, and (A) which involves payment or receipt by the
Company after the date of this Agreement under any such agreement, contract or commitment of more than $100,000 in the aggregate, or obligations after the date of this Agreement in excess of $100,000 in the aggregate or (B) that is material to the
business or operations of the Company taken as a whole; or
(xiii) Company Real Estate Leases.
28
(b) The Company has delivered or made available to Parent accurate and complete copies of all Company Material Contracts, including all amendments thereto. There are no Company
Material Contracts that are not in written form. The Company has not, nor to the Company’s Knowledge, as of the date of this Agreement has any other party to a Company Material Contract, breached, violated or defaulted under, or received notice
that it breached, violated or defaulted under, any of the terms or conditions of any Company Material Contract in such a manner, and, if such Company Material Contract provides for a cure period, the Company or such other party fails to have cured
such breach, violation or default, so that any other party or the Company, as the case may be, is permitted to modify, cancel or terminate any such Company Material Contract, or would permit any other party to seek damages which would reasonably be
expected to have a Company Material Adverse Effect. As to the Company, as of the date of this Agreement, each Company Material Contract is valid, binding, enforceable and in full force and effect, subject to the Enforceability Exceptions. No Person
is renegotiating, or has a right pursuant to the terms of any Company Material Contract to change, any material amount paid or payable to the Company under any Company Material Contract or any other material term or provision of any Company
Material Contract.
(a) The Company is, and has been in material compliance with all applicable Laws. No investigation, claim, suit, proceeding, audit, Order or other Legal Proceeding or action by
any Governmental Authority is pending or, to the Knowledge of the Company, threatened against the Company. There is no agreement or Order binding upon the Company which (i) has or would reasonably be expected to have the effect of prohibiting or
materially impairing any business practice of the Company, any acquisition of material property by the Company or the conduct of business by the Company as currently conducted, (ii) is reasonably likely to have an adverse effect on the Company’s
ability to comply with or perform any covenant or obligation under this Agreement or (iii) is reasonably likely to have the effect of preventing, delaying, making illegal or otherwise interfering with the Contemplated Transactions.
(b) Except for matters regarding the U.S. Food and Drug Administration (or any successor agency thereto) (“FDA”)
or other comparable Governmental Authority responsible for regulation of the development, testing, manufacturing, processing, storage, labelling, sale, marketing, advertising, distribution and importation or exportation of drug products (“Drug Regulatory Agency”), the Company holds all required Governmental Authorizations for the operation of the business of the Company as currently conducted (the “Company Permits”).
Section 3.14(b) of the Company Disclosure Letter identifies each Company Permit. The Company is in material compliance with the terms of the Company Permits. No Legal Proceeding is pending or, to the Knowledge of the Company,
threatened, which seeks to revoke, substantially limit, suspend or materially modify any Company Permit. The rights and benefits of each Company Permit will be available to the Surviving Entity or its Subsidiaries, as applicable, immediately after
the Effective Time on terms substantially identical to those enjoyed by the Company as of the date of this Agreement and immediately prior to the Effective Time.
(c) There are no Legal Proceedings pending or, to the Knowledge of the Company, threatened with respect to an alleged violation by the Company of the
Federal Food, Drug, and Cosmetic Act (“FDCA”), the Public Health Service Act (“PHSA”), FDA regulations adopted thereunder, the Controlled Substances Act or any other
similar Law promulgated by a Drug Regulatory Agency.
29
(d) The Company holds all required Governmental Authorizations issuable by any Drug Regulatory Agency necessary for the conduct of the business of the
Company as currently conducted (the “Company Product Candidates”) (collectively, the “Company Regulatory Permits”) and no such Company Regulatory Permit has been (i)
revoked, withdrawn, suspended, cancelled or terminated or (ii) modified in any adverse manner, other than immaterial adverse modifications. Section 3.14(d) of the Company Disclosure Letter identifies each material Company Regulatory
Permit. The Company has timely maintained and is in compliance in all material respects with the Company Regulatory Permits and has not received any written notice or correspondence or, to the Knowledge of the Company, other communication from any
Drug Regulatory Agency regarding (A) any material violation of or failure to comply materially with any term or requirement of any Company Regulatory Permit or (B) any revocation, withdrawal, suspension, cancellation, termination or material
modification of any Company Regulatory Permit. The Company has made available to Parent all information requested by Parent in the Company’s possession or control relating to material Company Product Candidates, including but not limited to
complete copies of the following (to the extent there are any): (x) adverse event reports; preclinical, clinical and other study reports and material study data; inspection reports, notices of adverse findings, untitled letters, warning letters,
filings and letters and other written correspondence to and from any Drug Regulatory Agency; and meeting minutes with any Drug Regulatory Agency and (y) similar reports, material study data, notices, letters, filings, correspondence and meeting
minutes with any other Governmental Authority. All such information is accurate and complete in all material respects.
(e) All clinical, preclinical and other studies and tests conducted by or on behalf of, or sponsored by, the Company, or in which the Company or its current products or product
candidates, including the Company Product Candidates, have participated, were, and, if still pending, are being conducted in accordance in all material respects with standard medical and scientific research procedures, in accordance in all material
respects with the applicable protocols and in compliance in all material respects with the applicable regulations of the Drug/Device Regulatory Agencies and other applicable Law, including 21 C.F.R. Parts 11, 50, 54, 56, 58, 312 and 812. The
Company has not received any written notices, correspondence or other communications from any Drug Regulatory Agency, Governmental Authority, institutional review board, ethics committee or safety monitoring committee requiring, or to the Knowledge
of the Company threatening to initiate, any action to place a clinical hold order on, or otherwise terminate, delay or suspend any clinical studies conducted by or on behalf of, or sponsored by, the Company or in which the Company or its current
products or product candidates, including the Company Product Candidates, have participated. Further, no clinical investigator, researcher or clinical staff participating in any clinical study conducted by or, to the Knowledge of the Company, on
behalf of the Company has been disqualified from participating in studies involving the Company Product Candidates, and to the Knowledge of the Company, no such administrative action to disqualify such clinical investigators, researchers or
clinical staff has been threatened or is pending.
(f) The Company is not, and to the Knowledge of the Company, no contract manufacturer with respect to any Company Product Candidate, is the subject of any pending or, to the
Knowledge of the Company, threatened investigation in respect of its business or products, including Company Product Candidates, by the FDA pursuant to its “Fraud, Untrue Statements of Material Facts, Bribery, and Illegal Gratuities” Final Policy
set forth in 56 Fed. Reg. 46191 (September 10, 1991) and any amendments thereto or by any other Drug Regulatory Agency under a comparable policy. Neither the Company, nor any of its officers, directors, employees or agents have been debarred or
excluded from participation in any federal healthcare programs. The Company has not, and to the Knowledge of the Company, no contract manufacturer, nor their respective officers, employees or agents, with respect to any Company Product Candidate
has committed any acts, made any statement or failed to make any statement, in each case in respect of its business or products that would violate the FDA’s “Fraud, Untrue Statements of Material Facts, Bribery, and Illegal Gratuities” Final Policy,
and any amendments thereto or a comparable policy of any other Drug Regulatory Agency. None of the Company, and to the Knowledge of the Company, any contract manufacturer with respect to any Company Product Candidate, or any of their respective
officers, employees or agents is currently or has been debarred, convicted of any crime or is engaging or has engaged in any conduct that could result in a debarment or exclusion under (i) 21 U.S.C. Section 335a or (ii) any similar applicable Law.
To the Knowledge of the Company, no debarment or exclusionary claims, actions, proceedings or investigations in respect of their business or any Company Product Candidates are pending or threatened against the Company, or to the Knowledge of the
Company, any contract manufacturer with respect to any Company Product Candidate, or any of their respective officers, employees or agents.
30
(g) All manufacturing operations conducted by, or to the Knowledge of the Company, for the benefit of the Company in connection with any Company Product Candidate have been and are being conducted in
compliance in all material respects with applicable Laws, including the applicable FDA requirements for current good manufacturing practices, and any similar Laws enforced by Governmental Authorities in countries outside the United States.
(h) Neither the Company nor, to the Knowledge of the Company, any manufacturing site of a contract manufacturer or laboratory, with respect to any Company Product Candidate, (i) is subject to a Drug
Regulatory Agency shutdown or import or export prohibition or (ii) has received any Form FDA 483, notice of violation, warning letter, untitled letter or similar correspondence or notice from the FDA or other Drug/Device Regulatory Agency alleging or
asserting noncompliance with any applicable Law, in each case, that have not been complied with or closed to the satisfaction of the relevant Drug Regulatory Agency, and, to the Knowledge of the Company, neither the FDA nor any other Drug Regulatory
Agency is considering such action.
3.15 Legal Proceedings; Orders.
(a) There is no pending Legal Proceeding and, to the Knowledge of the Company, no Person has threatened to commence any Legal Proceeding: (i) that involves the Company or any of its Subsidiaries or
any Company Associate (in his or her capacity as such) or any of the material assets owned or used by the Company or any of its Subsidiaries or (ii) that challenges, or that may have the effect of preventing, delaying, making illegal or otherwise
interfering with, the Contemplated Transactions.
(b) There is no Order to which the Company or any of its Subsidiaries, or any of the material assets owned or used by the Company or any of its Subsidiaries, is subject. To the Knowledge of the
Company, no officer or Company Key Employee is subject to any Order that prohibits such officer or Company Key Employee from engaging in or continuing in any conduct, activity or practice relating to the Company or any of its Subsidiaries or any
material assets owned or used by the Company or any of its Subsidiaries.
3.16 Tax Matters.
(a) All income Tax Returns and all other material Tax Returns required to be filed by or with respect to the Company under applicable Law have been duly and timely filed (taking into account any
applicable extensions). All such Tax Returns were true, correct and complete in all material respects. Subject to exceptions as would not be material, no written claim has been made by a Governmental Authority in a jurisdiction where the Company does
not file Tax Returns that the Company is subject to taxation by that jurisdiction.
(b) All material amounts of Taxes due and owing by the Company (whether or not shown on any Tax Return) have been timely paid (taking into account any applicable extensions).
(c) The Company has withheld and paid to the appropriate Governmental Authority all material Taxes required to have been withheld and paid in connection with any amounts paid or owing to any employee,
independent contractor, creditor, stockholder or other third party.
(d) There are no Encumbrances for a material amount of Taxes (other Encumbrances described in clause (a) of the definition of “Permitted Encumbrances”) upon any of the assets of the Company.
31
(e) No deficiencies for a material amount of Taxes with respect to the Company have been claimed, proposed or assessed by any Governmental Authority that have not been timely paid in full. There are
no pending (or, based on written notice, threatened) material audits, assessments, examinations or other actions for or relating to any liability in respect of Taxes of the Company. The Company has not granted a waiver of any statute of limitations in
respect of a material amount of Taxes or an extension of time with respect to a material Tax assessment or deficiency that, in each case, is currently in effect.
(f) The Company has not entered into a “closing agreement” as described in section 7121 of the Code (or any comparable provisions of state, local or non-U.S. Law) with any Governmental Authority.
(g) The Company has not been a United States real property holding corporation within the meaning of Section 897(c)(2) of the Code in the last five (5) years.
(h) The Company is not a party to any Tax allocation, Tax sharing or similar agreement (including indemnity arrangements), other than customary commercial Contracts entered into in the Ordinary Course
of Business the primary purpose of which does not relate to Tax (an “Ordinary Course Agreement”).
(i) The Company has not been a member of an affiliated group filing a consolidated U.S. federal income Tax Return (other than a group the common parent of which is the Company). The Company has no
Liability for the Taxes of any Person under Treasury Regulations Section 1.1502-6 (or any similar provision of state, local, or foreign law), as a transferee or successor, or by Contract (other than an Ordinary Course Agreement).
(j) The Company will not be required to include any material item of income in, or exclude any material item of deduction from, taxable income, or pay any Taxes, in any taxable period (or portion
thereof) ending after the Closing Date to the extent such item is economically attributable to a taxable period (or portion thereof) ending on or before the Closing Date, including as a result of: (i) any change in, or use of an improper, method of
accounting prior to the Closing, (ii) any “closing agreement” as described in Section 7121 of the Code (or any similar or analogous provision of state, local or non-U.S. Law) executed on or prior to the Closing Date, (iii) instalment sale or open
transaction disposition made on or prior to the Closing, (iv) any prepaid amount or advance payment received or deferred revenue accrued on or prior to the Closing Date, or (v) intercompany transactions occurring on or prior to the Closing Date or any
excess loss account in existence on the Closing Date described in Treasury Regulations under Section 1502 of the Code (or any similar or analogous provision of state, local or non-U.S. Law).
(k) The Company is not subject to, nor has the Company applied for, any private letter ruling of the Internal Revenue Service, request for administrative relief, request for technical advice, request
for a change of any method of Tax accounting, or comparable rulings concerning Tax matters of any Governmental Authority. The Company has not granted to any Person any power of attorney that will remain in force after the Closing with respect to any
Tax matter.
(l) The Company has not distributed stock of another Person, or has had its stock distributed by another Person, in a transaction that was purported or intended to be governed in whole or in part by
Section 355 of the Code or Section 361 of the Code.
(m) The Company has not entered into any “reportable transaction” within the meaning of Section 6707A of the Code or Treasury Regulations Sections 1.6011-4(b).
32
(n) The Company is currently classified as a corporation and has never elected to be classified as an S corporation for U.S. federal income Tax purposes.
(o) Any related party transactions conducted by the Company have been on an arms-length basis in accordance with Section 482 of the Code or, to the extent applicable, any analogous, corresponding or
similar provision of applicable Law in all material respects.
(p) The Company is not aware of any facts or circumstances, and has not taken or agreed to take any action, in each case, that would reasonably be expected to prevent or impede the Intended Tax
Treatment.
3.17 Employee and Labor Matters; Benefit Plans.
(a) The Company has made available to Parent a list (on an anonymized basis) setting forth, for each Company Associate who is an employee of the Company or any of its Subsidiaries, whether full- or
part-time, such employee’s annual salary (or if hourly, hourly rate), most recent annual bonus received, and current annual bonus opportunity. No Company Key Employee has indicated to the Company, or any of its Subsidiaries, that he or she intends to
resign or retire as a result of the transactions contemplated by this Agreement or otherwise. The Company has made available to Parent a list (on an anonymized basis) setting forth, for each Company Associate who is an individual independent contractor
engaged by the Company, such contractor’s rate of compensation.
(b) The employment of the Company’s and each of its Subsidiaries’ employees is terminable by the Company and/or its applicable Subsidiary at will. The Company has made available to Parent accurate and
complete copies of all employee manuals and handbooks, to the extent currently effective and material.
(c) Neither the Company nor any of its Subsidiaries is a party to, bound by the terms of, and does not have a duty to bargain under, any collective bargaining agreement or other Contract with a labor
organization representing its employees, and there are no labor organizations representing or, to the Knowledge of the Company, purporting to represent or seeking to represent any employees of the Company.
(d) Section 3.17(d) of the Company Disclosure Letter lists all Company Employee Plans (other than employment arrangements which are terminable “at will” without any contractual
obligation on the part of the Company or any of its Subsidiaries to make any severance, termination, change in control or similar payment and that are substantively identical to the employment arrangements made available to Parent).
(e) Each Company Employee Plan that is intended to be qualified under Section 401(a) of the Code has received a favorable determination or opinion letter with respect to such qualified status from the
IRS. To the Knowledge of the Company, nothing has occurred that would reasonably be expected to adversely affect the qualified status of any such Company Employee Plan or the exempt status of any related trust.
(f) Each Company Employee Plan has been established, maintained and operated in compliance, in all material respects, with its terms all applicable Law, including, without limitation, the Code, ERISA
and the Affordable Care Act. No Legal Proceeding (other than those relating to routine claims for benefits) is pending or, to the Knowledge of the Company, threatened with respect to any Company Employee Plan. All payments and/or contributions required
to have been made with respect to all Company Employee Plans either have been made or have been accrued in accordance with the terms of the applicable Company Employee Plan and applicable Law.
33
(g) Neither the Company nor any of its ERISA Affiliates maintains, contributes to or is required to contribute to, or has, in the past six (6) years, maintained, contributed to or been required to
contribute to (i) any defined benefit pension plan as defined under Section 3(35) of ERISA that is or was subject to Title IV or Section 302 of ERISA or Section 412 of the Code, (ii) a Multiemployer Plan, (iii) any funded welfare benefit plan within
the meaning of Section 419 of the Code, (iv) any Multiple Employer Plan, or (v) any Multiple Employer Welfare Arrangement. Neither the Company nor any of its ERISA Affiliates has incurred any liability under Title IV of ERISA in the past six (6) years.
(h) No Company Employee Plan provides for, and the Company has not promised to provide any, medical or other welfare benefits to any service provider beyond termination of service or retirement, other
than (i) pursuant to COBRA or an analogous state law requirement or (ii) continuation coverage through the end of the month in which such termination or retirement occurs. The Company does not sponsor or maintain any self-funded medical or long-term
disability benefit plan.
(i) Each Company Employee Plan that constitutes in any part a “nonqualified deferred compensation plan” (as such term is defined under Section 409A(d)(1) of the Code and the guidance thereunder)
(each, a “Company 409A Plan”) has been operated and maintained in all material respects in operational and documentary compliance with the requirements of Section 409A of the Code and the applicable guidance
thereunder.
(j) The Company and each of its Subsidiaries is, and has been, in material compliance with all applicable federal, state and local laws, rules and regulations respecting employment, employment
practices, terms and conditions of employment, worker classification, tax withholding, prohibited discrimination, retaliation and harassment, equal employment, fair employment practices, meal and rest periods, immigration status, employee and workplace
safety and health, wages (including overtime wages), compensation, hours of work, “plant closings” and “mass layoffs” within the meaning of the Worker Adjustment and Retraining Act of 1988 or similar state or local law (the “WARN Act”), labor practices or disputes, restrictive covenants, employment agreements, workers’ compensation and long-term disability policies, leaves of absence and worker privacy (collectively, “Employment-Related
Laws”), and in each case, with respect to employees of the Company and any of its Subsidiaries: (i) has withheld and reported all material amounts required by law or by agreement to be withheld and reported with respect to wages, salaries and
other payments to employees, (ii) is not liable for any material amounts of arrears of wages, severance pay or any Taxes or any penalty for failure to comply with any of the foregoing and (iii) is not liable for any material payment to any trust or
other fund governed by or maintained by or on behalf of any Governmental Authority, with respect to unemployment compensation benefits, social security or other benefits or obligations for employees (other than routine payments to be made in the
Ordinary Course of Business). There are no material Legal Proceedings, claims, labor disputes or organizing activities, or grievances pending or, to the Knowledge of the Company, threatened or reasonably anticipated against or involving the Company or
any of its Subsidiaries or any trustee of the Company or any of its Subsidiaries relating to any employee, contingent worker, director, employment agreement or Employee Plan (other than routine claims for benefits) or Employment-Related Laws. To the
Knowledge of the Company, there are no material pending or threatened or reasonably anticipated claims or actions against the Company, any trustee or any trustee of any Subsidiary of the Company under any workers’ compensation policy or long-term
disability policy. The Company is not a party to a conciliation agreement, consent decree or other agreement or Order with any federal, state or local agency or Governmental Authority with respect to employment practices.
34
(k) Neither the Company nor any of its Subsidiaries has any material liability with respect to any misclassification within the last three (3) years of: (i) any Person as an independent contractor
rather than as an employee, (ii) any employee leased from another employer or (iii) any employee currently or formerly classified as exempt from overtime wages.
(l) To the Company’s Knowledge, in the last three (3) years, there has never been, nor has there been any threat of, any material strike, slowdown, work stoppage, lockout, job action, union,
organizing activity, question concerning representation or any similar activity or dispute, by or with respect to any Company Associates. No event has occurred within the past six months, and no condition or circumstance exists, that, to the Company’s
Knowledge, might directly or indirectly be likely to give rise to or provide a basis for the commencement of any such material strike, slowdown, work stoppage, lockout, job action, union organizing activity, question concerning representation or any
similar activity or dispute.
(m) Neither the Company nor any of its Subsidiaries is, nor has the Company nor any of its Subsidiaries been, engaged in any material unfair labor practice within the meaning of the National Labor
Relations Act. There is no material Legal Proceeding, claim, labor dispute or grievance pending or, to the Knowledge of the Company, threatened or reasonably anticipated relating to any employment contract, privacy right, labor dispute, wages and
hours, leave of absence, plant closing notification, workers’ compensation policy, long-term disability policy, harassment, retaliation, immigration, employment statute or regulation, safety or discrimination matter involving any current or former
employee of the Company or any of its Subsidiaries including charges of unfair labor practices or discrimination complaints.
(n) There is no contract, agreement, plan or arrangement to which the Company or any of its Subsidiaries is a party or by which it is bound to compensate any of its employees or other service
providers for any income or excise taxes paid pursuant to Section 4999 or Section 409A of the Code.
(o) Neither the Company nor any of its Subsidiaries is a party to any Contract that as a result of the execution and delivery of this Agreement, the stockholder approval of this Agreement, nor the
consummation of the transactions contemplated hereby, could (either alone or in conjunction with any other event) result in, or cause the accelerated vesting, payment, funding or delivery of any payment or benefit to any employee, officer, director or
other service provider of the Company or any of its Subsidiaries.
3.18 Environmental Matters. The Company has complied with all applicable Environmental Laws, which compliance includes the possession by the Company of all permits and other Governmental
Authorizations required under applicable Environmental Laws and compliance with the terms and conditions thereof, except for any failure to be in compliance that, individually or in the aggregate, would not result in a Company Material Adverse Effect.
The Company has not received any written notice or other communication (in writing or otherwise), whether from a Governmental Authority, citizens group, employee or otherwise, that alleges that the Company is not in compliance with any Environmental
Law and, to the Knowledge of the Company, there are no circumstances that may prevent or interfere with the Company’s compliance with any Environmental Law in the future, except where such failure to comply would not reasonably be expected to have a
Company Material Adverse Effect. To the Knowledge of the Company: (i) no current or prior owner of any property leased or controlled by the Company has received any written notice or other communication relating to property owned or leased at any time
by the Company, whether from a Governmental Authority, citizens group, employee or otherwise, that alleges that such current or prior owner or the Company is not in compliance with or violated any Environmental Law relating to such property and (ii)
the Company has no material liability under any Environmental Law. The Company has made available all environmental site assessments, environmental audits and other material environmental documents in the Company’s possession or control relating to the
Company, including the Company’s business and current or former facilities.
35
3.19 Insurance. The Company has delivered to Parent accurate and complete copies of all material insurance policies and all material self-insurance programs and arrangements relating to the
business, assets, liabilities and operations of the Company. Each of such insurance policies is in full force and effect and the Company is in compliance in all material respects with the terms thereof. Other than customary end of policy notifications
from insurance carriers, the Company has not received any notice or other communication regarding any actual or possible: (i) cancellation or invalidation of any insurance policy or (ii) refusal or denial of any coverage, reservation of rights or
rejection of any material claim under any insurance policy. The Company has provided timely written notice to the appropriate insurance carrier(s) of each Legal Proceeding pending against the Company, and no such carrier has issued a denial of coverage
or a reservation of rights with respect to any such Legal Proceeding, or informed the Company of its intent to do so.
3.20 No Financial Advisors. No broker, finder or investment banker is entitled to any brokerage fee, finder’s fee, opinion fee, success fee, transaction fee or other fee or commission in
connection with the Contemplated Transactions based upon arrangements made by or on behalf of the Company.
3.21 Transactions with Affiliates. Section 3.21 of the Company Disclosure Letter describes any material transactions or relationships between, on one hand, the Company and, on
the other hand, any (a) executive officer or director of the Company or any of such executive officer’s or director’s immediate family members, (b) owner of more than 5% of the voting power of the outstanding Company Capital Stock or (c) to the
Knowledge of the Company, any “related person” (within the meaning of Item 404 of Regulation S-K under the Securities Act) of any such officer, director or owner (other than the Company) in the case of each of (a), (b) or (c) that is of the type that
would be required to be disclosed under Item 404 of Regulation S-K under the Securities Act.
3.22 Privacy and Data Security. The Company is and has at all times been in compliance with all applicable Privacy Laws and the applicable terms of any Company Contracts governing privacy, data
protection, data security, trans-border data flow, data loss, data theft, or breach notification, data localization, sending solicited or unsolicited electronic mail or text messages, cookies or other tracking technology, with respect to, or the
collection, handling, use, maintenance, storage, disclosure, transfer, or other processing of, Personal Information (including any such information of individuals, clinical trial participants, patients, patient family members, caregivers or advocates,
physicians and other health care professionals, clinical trial investigators, researchers, pharmacists that interact with the Company in connection with the operation of the Company’s business), except, in each case, for such noncompliance as has not
had, and would not reasonably be expected to have, individually or in the aggregate, a Company Material Adverse Effect. To the Knowledge of the Company, the Company (i) has implemented and maintains reasonable written policies and procedures that
materially comply with applicable Privacy Laws and are designed to protect the privacy and security of Personal Information (the “Privacy Policies”) and (ii) has complied with such Privacy Policies, except for such noncompliance as has not had, and
would not reasonably be expected to have, individually or in the aggregate, a Company Material Adverse Effect. To the Knowledge of the Company, no Legal Proceeding has been asserted or threatened against the Company by any Person alleging a violation
of Privacy Laws, Privacy Policies, or the applicable terms of any Company Contracts governing privacy, data protection, data security, trans-border data flow, data loss, data theft, or breach notification, data localization, sending solicited or
unsolicited electronic mail or text messages, cookies or other tracking technology, with respect to, or the collection, handling, use, maintenance, storage, disclosure, transfer, or other processing of, Personal Information. To the Knowledge of the
Company, there have been no data security incidents or data breaches or other adverse events or incidents that have resulted in any unauthorized access to, or collection, use, disclosure, modification or destruction of, Personal Information or other
data in the possession or control of the Company or any service provider acting on behalf of the Company, in each case, where such incident, breach or event resulted in a notification obligation to any Person under applicable Law or pursuant to the
terms of any Company Contract.
36
3.23 No Other Representations or Warranties. The Company hereby acknowledges and agrees that, except for the representations and warranties contained in this Agreement, neither Parent nor any
other person on behalf of Parent makes any express or implied representation or warranty with respect to Parent or with respect to any other information provided to the Company, any of its stockholders or any of their respective Affiliates in
connection with the Contemplated Transactions, and (subject to the express representations and warranties of Parent set forth in Section 4 (in each case as qualified and limited by the Parent Disclosure Letter)) none of the Company, or any of
its Representatives or stockholders, has relied on any such information (including the accuracy or completeness thereof).
Section 4. Representations and Warranties of Parent and Merger Sub
Except (i) as set forth in the written disclosure document delivered by Parent to the Company (the “Parent Disclosure Letter”) concurrently with the execution of
this Agreement or (ii) as disclosed in the Parent SEC Documents filed with the SEC prior to the date hereof and publicly available on the SEC’s Electronic Data Gathering Analysis and Retrieval system (but (A) without giving effect to any amendment
thereof filed with, or furnished to the SEC on or after the date hereof and (B) excluding any disclosures contained under the heading “Risk Factors” and any disclosure of risks included in any “forward-looking statements” disclaimer or in any other
section to the extent they are forward-looking statements or cautionary, predictive or forward-looking in nature), it being understood that any matter disclosed in the Parent SEC Documents shall be deemed to be disclosed in a section of the Parent
Disclosure Letter only to the extent that is readily apparent from a reading of such Parent SEC Documents that is applicable to such section or subsection of the Parent Disclosure Letter, Parent and Merger Sub represent and warrant to the Company as
follows:
4.1 Due Organization; Subsidiaries
(a) Each of Parent and Merger Sub is a corporation duly incorporated or formed, as applicable, validly existing and in good standing under the Laws of the jurisdiction of its incorporation or
formation, as applicable, and has all necessary corporate power and authority: (i) to conduct its business in the manner in which its business is currently being conducted, (ii) to own or lease and use its property and assets in the manner in which its
property and assets are currently owned or leased and used and (iii) to perform its obligations under all Contracts by which it is bound. Since the date of its incorporation, Merger Sub has not engaged in any activities other than in connection with or
as contemplated by this Agreement.
(b) Each of Parent and its Subsidiaries is licensed and qualified to do business, and is in good standing (to the extent applicable in such jurisdiction), under the Laws of all jurisdictions where the
nature of its business in the manner in which its business is currently being conducted requires such licensing or qualification other than in jurisdictions where the failure to be so qualified individually or in the aggregate would not be reasonably
expected to have a Parent Material Adverse Effect.
37
(c) Parent has no Subsidiaries other than Merger Sub and except as set forth on Section 4.1(c) of the Parent Disclosure Letter, Parent does not own any capital stock of, or any equity
ownership or profit sharing interest of any nature in, or control directly or indirectly, any other Entity other than Merger Sub. Except as set forth on Section 4.1(c) of the Parent Disclosure Letter, Parent is not and has not otherwise
been, directly or indirectly, a party to, member of or participant in any partnership, joint venture or similar business entity. Parent has not agreed and is not obligated to make, nor is Parent bound by any Contract under which it may become obligated
to make, any future investment in or capital contribution to any other Entity. Parent has not, at any time, been a general partner of, and has not otherwise been liable for any of the debts or other obligations of, any general partnership, limited
partnership or other Entity.
4.2 Organizational Documents. Parent has delivered to the Company accurate and complete copies of Parent’s Organizational Documents. Parent is not in breach or violation of its Organizational
Documents in any material respect.
4.3 Authority; Binding Nature of Agreement. Each of Parent and Merger Sub has all necessary corporate power and authority to enter into and to perform its obligations under this Agreement and
to consummate the Contemplated Transactions. The Special Committee and the Parent Board have: (a) determined that the Contemplated Transactions are fair to, advisable and in the best interests of Parent and its stockholders (including the stockholders
who are not affiliated with the Company), (b) approved and declared advisable this Agreement and the Contemplated Transactions, including the issuance of shares of Parent Capital Stock to the stockholders of the Company pursuant to the terms of this
Agreement and (c) determined to recommend, upon the terms and subject to the conditions set forth in this Agreement, that the stockholders of Parent vote to approve the Preferred Stock Conversion Proposal. The Merger Sub Board (by unanimous written
consent) has: (x) determined that the Contemplated Transactions are fair to, advisable and in the best interests of Merger Sub and its sole stockholder, (y) deemed advisable and approved this Agreement and the Contemplated Transactions and (z)
determined to recommend, upon the terms and subject to the conditions set forth in this Agreement, that the stockholder of Merger Sub vote to adopt this Agreement and thereby approve the Contemplated Transactions. This Agreement has been duly executed
and delivered by Parent and Merger Sub and, assuming the due authorization, execution and delivery by the Company, constitutes the legal, valid and binding obligation of Parent and Merger Sub, enforceable against each of Parent and Merger Sub in
accordance with its terms, subject to the Enforceability Exceptions.
4.4 Vote Required. The affirmative vote (or written consent) of a majority of the issued and outstanding shares of Parent Common Stock is the only vote of the holders of any class or series of
Parent Capital Stock necessary to adopt and approve the Preferred Stock Conversion Proposal.
4.5 Non-Contravention; Consents.
(a) Subject to compliance with any applicable requirements of the HSR Act, and the filing of the Certificate of Merger required by the DGCL, and neither (x) the execution, delivery or performance of
this Agreement by Parent or Merger Sub, nor (y) the consummation of the Contemplated Transactions, will directly or indirectly (with or without notice or lapse of time):
(i) contravene, conflict with or result in a violation of any of the provisions of the Organizational Documents of Parent or its Subsidiaries;
(ii) contravene, conflict with or result in a material violation of, or give any Governmental Authority or other Person the right to challenge the Contemplated Transactions or to
exercise any remedy or obtain any relief under, any Law or any Order to which Parent or its Subsidiaries or any of the assets owned or used by Parent or its Subsidiaries, is subject;
38
(iii) contravene, conflict with or result in a material violation of any of the terms or requirements of, or give any Governmental Authority the right to revoke, withdraw, suspend,
cancel, terminate or modify, any Governmental Authorization that is held by Parent or its Subsidiaries or that otherwise relates to the business of Parent, or any of the assets owned, leased or used by Parent;
(iv) contravene, conflict with or result in a violation or breach of, or result in a default under, any provision of any Parent Material Contract, or give any Person the right to:
(A) declare a default or exercise any remedy under any Parent Material Contract, (B) any material payment, rebate, chargeback, penalty or change in delivery schedule under any such Parent Material Contract, (C) accelerate the maturity or performance of
any Parent Material Contract or (D) cancel, terminate or modify any term of any Parent Material Contract, except in the case of any nonmaterial breach, default, penalty or modification; or
(v) result in the imposition or creation of any Encumbrance upon or with respect to any asset owned or used by Parent or its Subsidiaries (except for Permitted Encumbrances).
(b) Except for (i) any Consent set forth on Section 4.5(a) of the Parent Disclosure Letter under any Parent Contract, (ii) the filing of the Certificate of Merger with the Secretary of
State of the State of Delaware pursuant to the DGCL, (iii) compliance with any applicable requirements of the HSR Act, and (iv) such consents, waivers, approvals, orders, authorizations, registrations, declarations and filings as may be required under
applicable federal and state securities laws, and neither Parent nor any of its Subsidiaries was, is or will be required to make any filing with or give any notice to, or to obtain any Consent from, any Person in connection with (x) the execution,
delivery or performance of this Agreement or (y) the consummation of the Contemplated Transactions.
(c) The Parent Board and the Merger Sub Board have taken and will take all actions necessary to ensure that the restrictions applicable to business combinations contained in Section 203 of the DGCL
are, and will be, inapplicable to the execution, delivery and performance of this Agreement and to the consummation of the Contemplated Transactions. No other state takeover statute or similar Law applies or purports to apply to the Merger, this
Agreement or any of the other Contemplated Transactions.
4.6 Capitalization.
(a) The authorized capital stock of Parent consists of (i) 400,000,000 shares of Parent Common Stock of which 90,890,381 shares have been issued and are outstanding as of September 30, 2024 (the “Capitalization Date”) and (ii) 5,000,000 shares of Parent Preferred Stock, of which no shares are currently designated. No shares of Parent Preferred Stock have been issued and are outstanding as of the
Capitalization Date. Parent does not hold any shares of its capital stock in its treasury.
(b) All of the outstanding shares of Parent Common Stock have been duly authorized and validly issued, and are fully paid and nonassessable and are free of any Encumbrances other than Encumbrances set
forth in the Organizational Documents or under applicable securities Laws. None of the outstanding shares of Parent Common Stock is entitled or subject to any preemptive right, right of participation, right of maintenance or any similar right and none
of the outstanding shares of Parent Common Stock is subject to any right of first refusal in favor of Parent. Except as contemplated herein, there is no Parent Contract relating to the voting or registration of, or restricting any Person from
purchasing, selling, pledging or otherwise disposing of (or granting any option or similar right with respect to), any shares of Parent Common Stock. Parent is not under any obligation, nor is Parent bound by any Contract pursuant to which it may
become obligated, to repurchase, redeem or otherwise acquire any outstanding shares of Parent Common Stock or other securities. Section 4.6(b) of the Parent Disclosure Letter accurately and completely describes all repurchase rights
held by Parent with respect to shares of Parent Common Stock (including shares issued pursuant to the exercise of stock options) and specifies which of those repurchase rights are currently exercisable.
39
(c) Except for the Parent 2023 Omnibus Incentive Plan, (as may be amended from time to time, the “Parent Stock Plan”) and except as set forth on Section 4.6(c)(i)
of the Parent Disclosure Letter, Parent does not have any stock option plan or any other plan, program, agreement or arrangement providing for any equity-based compensation for any Person. Parent does not have any employee stock purchase plan or
similar program. Section 4.6(c)(ii) of the Parent Disclosure Letter sets forth the following information with respect to each Parent Option outstanding as of the Capitalization Date, as applicable: (i) the name of the holder, (ii) the
number of shares of Parent Common Stock subject to such Parent Option as of the Capitalization Date, (iii) the exercise price of such Parent Option, (iv) the date on which such Parent Option was granted, (v) the applicable vesting schedule, including
any acceleration provisions, (vi) the date on which such Parent Option expires, (vii) whether such Parent Option is intended to be an “incentive stock option” (as defined in the Code) or a nonqualified stock option and (viii) in the case of a Parent
Option, the plan pursuant to which such Parent Option was granted. Parent has made available to the Company accurate and complete copies of equity incentive plans pursuant to which Parent has equity-based awards, the forms of all award agreements
evidencing such equity-based awards and evidence of board and stockholder approval of the Parent Stock Plan and any amendments thereto.
(d) Except for the outstanding Parent Options or as set forth on Section 4.6(d) of the Parent Disclosure Letter, there is no: (i) outstanding subscription, option, call, warrant or
right (whether or not currently exercisable) to acquire any shares of the capital stock or other securities of Parent, (ii) outstanding security, instrument or obligation that is or may become convertible into or exchangeable for any shares of the
capital stock or other securities of Parent, (iii) stockholder rights plan (or similar plan commonly referred to as a “poison pill”) or Contract under which Parent is or may become obligated to sell or otherwise issue any shares of its capital stock or
any other securities or (iv) condition or circumstance that may give rise to or provide a basis for the assertion of a claim by any Person to the effect that such Person is entitled to acquire or receive any shares of capital stock or other securities
of Parent. There are no outstanding or authorized stock appreciation, phantom stock, profit participation or other similar rights with respect to Parent.
(e) All outstanding shares of Parent Common Stock, Parent Options and other securities of Parent have been issued and granted in compliance in all material respects with (i) all applicable securities
laws and other applicable Law and (ii) all requirements set forth in applicable Contracts.
(f) With respect to Parent Options granted pursuant to the Parent Stock Plan, (i) each grant of a Parent Option was duly authorized no later than the date on which the grant of such Parent Option was
by its terms to be effective (the “Parent Grant Date”) by all necessary corporate action, including, as applicable, approval by the Parent Board (or a duly constituted and authorized committee thereof) or duly
authorized officer and any required stockholder approval by the necessary number of votes or written consents, (ii) each Parent Option grant was made in accordance with the terms of the Parent Stock Plan pursuant to which it was granted and all other
applicable Law and regulatory rules or requirements, and (iii) the per share exercise price of each Parent Option was not less than the fair market value of a share of Parent Common Stock on the applicable Parent Grant Date.
40
4.7 SEC Filings; Financial Statements.
(a) Parent has filed or furnished, as applicable, on a timely basis all forms, statements, certifications, reports and documents required to be filed or furnished by it with the SEC under the Exchange
Act or the Securities Act (the “Parent SEC Documents”). As of the time it was filed with the SEC (or, if amended or superseded by a filing prior to the date of this Agreement, then on the date of such filing),
each of the Parent SEC Documents complied in all material respects with the applicable requirements of the Securities Act or the Exchange Act (as the case may be) and as of the time they were filed, none of the Parent SEC Documents (excluding
information in such Parent SEC Documents that is “furnished” instead of “filed” under Items 2.02 or 7.01 in the Parent’s Current Reports on Form 8-K) contained any untrue statement of a material fact or omitted to state a material fact required to be
stated therein or necessary in order to make the statements therein, in light of the circumstances under which they were made, not misleading. The certifications and statements required by (i) Rule 13a-14 under the Exchange Act and (ii) 18 U.S.C. §1350
(Section 906 of the Sarbanes-Oxley Act) relating to the Parent SEC Documents (collectively, the “Certifications”) are accurate and complete and comply as to form and content with all applicable Laws. As used in
this Section 4.7, the term “file” and variations thereof shall be broadly construed to include any manner in which a document or information is furnished, supplied or otherwise made available to the SEC.
(b) The financial statements (including any related notes) contained or incorporated by reference in the Parent SEC Documents: (i) complied as to form in all material respects with the Securities Act
and the Exchange Act, as applicable, and the published rules and regulations of the SEC applicable thereto, (ii) were prepared in accordance with GAAP (except as may be indicated in the notes to such financial statements or, in the case of unaudited
financial statements, as permitted by Form 10-Q of the SEC, and except that the unaudited financial statements may not contain footnotes and are subject to normal and recurring year-end adjustments that are not reasonably expected to be material in
amount) applied on a consistent basis unless otherwise noted therein throughout the periods indicated and (iii) fairly present, in all material respects, the financial position of Parent as of the respective dates thereof and the results of operations
and cash flows of Parent for the periods covered thereby. Other than as expressly disclosed in the Parent SEC Documents filed prior to the date hereof, there has been no material change in Parent’s accounting methods or principles that would be
required to be disclosed in Parent’s financial statements in accordance with GAAP. The books of account and other financial records of Parent and each of its Subsidiaries are true and complete in all material respects.
(c) Parent’s auditor has at all times since the date of enactment of the Sarbanes-Oxley Act been: (i) a registered public accounting firm (as defined in Section 2(a)(12) of the Sarbanes-Oxley Act),
(ii) to the Knowledge of Parent, “independent” with respect to Parent within the meaning of Regulation S-X under the Exchange Act and (iii) to the Knowledge of Parent, in compliance with subsections (g) through (l) of Section 10A of the Exchange Act
and the rules and regulations promulgated by the SEC and the Public Company Accounting Oversight Board thereunder.
(d) Parent has not received any comment letter from the SEC or the staff thereof or any correspondence from Nasdaq or the staff thereof relating to the delisting or maintenance of listing of the
Parent Common Stock on Nasdaq. Parent has not disclosed any unresolved comments in the Parent SEC Documents.
(e) There have been no formal internal investigations regarding financial reporting or accounting policies and practices discussed with, reviewed by or initiated at the direction of the chief
executive officer, chief financial officer or general counsel of Parent, the Parent Board or any committee thereof, other than ordinary course audits or reviews of accounting policies and practices or internal controls required by the Sarbanes-Oxley
Act.
(f) Parent is in compliance in all material respects with the applicable provisions of the Sarbanes-Oxley Act, the Exchange Act and the applicable listing and governance rules and regulations of
Nasdaq.
41
(g) Parent maintains a system of internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) of the Exchange Act) that is sufficient to provide reasonable assurance
regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with GAAP, including policies and procedures sufficient to provide reasonable assurance (i) that Parent maintains records
that in reasonable detail accurately and fairly reflect Parent’s transactions and dispositions of assets, (ii) that transactions are recorded as necessary to permit preparation of financial statements in accordance with GAAP, (iii) that receipts and
expenditures are made only in accordance with the authorization policy and (iv) regarding prevention or timely detection of the unauthorized acquisition, use or disposition of Parent’s assets that could have a material effect on Parent’s financial
statements. Parent has evaluated the effectiveness of Parent’s internal control over financial reporting and, to the extent required by applicable Law, presented in any applicable Parent SEC Document that is a report on Form 10-K or Form 10-Q (or any
amendment thereto) its conclusions about the effectiveness of the internal control over financial reporting as of the end of the period covered by such report or amendment based on such evaluation. Parent has disclosed to Parent’s auditors and the
Audit Committee of the Parent Board (and made available to the Company a summary of the significant aspects of such disclosure) (A) all significant deficiencies and material weaknesses in the design or operation of internal control over financial
reporting that are reasonably likely to adversely affect Parent’s ability to record, process, summarize and report financial information and (B) any fraud, whether or not material, that involves management or other employees who have a significant role
in Parent’s or its Subsidiaries’ internal control over financial reporting. Except as disclosed in the Parent SEC Documents filed prior to the date hereof, Parent’s internal control over financial reporting is effective at the reasonable assurance
level and Parent has not identified any material weaknesses in the design or operation of Parent’s internal control over financial reporting.
(h) Parent’s “disclosure controls and procedures” (as defined in Rules 13a-15(e) and 15d-15(e) of the Exchange Act) are designed to ensure that all information (both financial and nonfinancial)
required to be disclosed by Parent in the reports that it files or submits under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the rules and forms of the SEC, and that all such information is
accumulated and communicated to Parent’s principal executive officer and principal financial officer as appropriate to allow timely decisions regarding required disclosure and to make the Certifications and such disclosure controls and procedures are
effective. Parent has carried out evaluation of the effectiveness of its disclosure controls and procedures as required by Rule 13a-15 of the Exchange Act.
4.8 Absence of Changes. Except as set forth on Section 4.8 of the Parent Disclosure Letter, between December 31, 2024 and the date of this Agreement, Parent has conducted its
business only in the Ordinary Course of Business (except for the execution and performance of this Agreement and the discussions, negotiations and transactions related thereto) and there has not been any Parent Material Adverse Effect.
4.9 Absence of Undisclosed Liabilities. Since December 31, 2024, neither Parent nor any of its Subsidiaries has any Liability of a type required to be reflected or reserved for on a balance
sheet prepared in accordance with GAAP except for: (a) Liabilities disclosed, reflected or reserved against in the Parent Balance Sheet, (b) normal and recurring current Liabilities that have been incurred by Parent or its Subsidiaries since the date
of the Parent Balance Sheet in the Ordinary Course of Business (none of which relates to any breach of contract, breach of warranty, tort, infringement or violation of Law), (c) Liabilities for performance of obligations of Parent or any of its
Subsidiaries under Parent Contracts, (d) Liabilities incurred in connection with the Contemplated Transactions, (e) Liabilities described in Section 4.9 of the Parent Disclosure Letter and (f) those Liabilities that are not material to
Parent.
42
4.10 Title to Assets. Each of Parent and its Subsidiaries owns, and has good and valid title to, or, in the case of leased properties and assets, valid leasehold interests in, all tangible
properties or tangible assets and equipment used or held for use in its business or operations or purported to be owned by it, including: (a) all tangible assets reflected on the Parent Balance Sheet and (b) all other tangible assets reflected in the
books and records of Parent as being owned by Parent. All of such assets are owned or, in the case of leased assets, leased by Parent or any of its Subsidiaries free and clear of any Encumbrances, other than Permitted Encumbrances.
4.11 Real Property; Leasehold. Neither Parent nor any of its Subsidiaries owns or has ever owned any real property, nor is Parent party to any agreement to purchase or sell any real property.
Parent has made available to the Company (a) an accurate and complete list of all real properties with respect to which Parent directly or indirectly holds a valid leasehold interest as well as any other real estate that is in the possession of or
leased by Parent or any of its Subsidiaries and (b) copies of all leases under which any such real property is possessed (the “Parent Real Estate Leases”), each of which is in full force and effect, with no
existing material default thereunder by Parent or its Subsidiaries or, to Parent’s Knowledge, the other party thereto.
4.12 Intellectual Property.
(a) Section 4.12(a) of the Parent Disclosure Letter is an accurate, true and complete listing of all Parent Registered IP.
(b) Section 4.12(b) of the Parent Disclosure Letter accurately identifies (i) all Parent Contracts pursuant to which any Parent IP Rights are licensed to Parent (other than (A) any
non-customized software that (1) is so licensed solely in executable or object code form pursuant to a nonexclusive, internal use software license and other Intellectual Property associated with such software and (2) is not incorporated into, or
material to the development, manufacturing, or distribution of, any of Parent products or services, (B) any Intellectual Property licensed on a nonexclusive basis ancillary to the purchase or use of services, equipment, reagents or other materials, (C)
any confidential information provided under confidentiality agreements and (D) agreements between Parent and its employees in Parent’s standard form thereof) and (ii) whether the license or licenses granted to Parent are exclusive or nonexclusive.
(c) Section 4.12(c) of the Parent Disclosure Letter accurately identifies each Parent Contract pursuant to which any Person has been granted any license or covenant not to sue under,
or otherwise has received or acquired any right (whether or not currently exercisable) or interest in, any Parent IP Rights (other than (i) any confidential information provided under confidentiality agreements and (ii) any Parent IP Rights
nonexclusively licensed to academic collaborators, suppliers or service providers for the sole purpose of enabling such academic collaborator, supplier or service providers to provide services for Parent’s benefit).
(d) Neither Parent nor any of its Subsidiaries is bound by, and no Parent IP Rights are subject to, any Contract containing any covenant or other provision that in any way limits or restricts the
ability of Parent or any of its Subsidiaries to use, exploit, assert, or enforce any Parent IP Rights anywhere in the world.
(e) Parent or one of its Subsidiaries exclusively owns all right, title, and interest to and in the Parent IP Rights (other than (i) Parent IP Rights licensed to Parent, or co-owned rights each as
identified in Section 4.12(e) of the Parent Disclosure Letter, (ii) any non-customized software that (A) is licensed to Parent solely in executable or object code form pursuant to a nonexclusive, internal use software license and other
Intellectual Property associated with such software and (B) is not incorporated into, or material to the development, manufacturing or distribution of, any of Parent or its Subsidiaries’ products or services and (iii) any Intellectual Property licensed
on a nonexclusive basis ancillary to the purchase or use of equipment, reagents or other materials), in each case, free and clear of any Encumbrances (other than Permitted Encumbrances). Without limiting the generality of the foregoing:
43
(i) All documents and instruments necessary to register or apply for or renew registration of Parent Registered IP have been validly executed, delivered, and filed in a timely
manner with the appropriate Governmental Authority.
(ii) Each Person who is or was an employee or contractor of Parent or any of its Subsidiaries and who is or was involved in the creation or development of any Intellectual Property
for Parent or any of its Subsidiaries has signed a valid, enforceable agreement containing a present assignment of such Intellectual Property to Parent or such Subsidiary and confidentiality provisions protecting trade secrets and confidential
information of Parent and its Subsidiaries.
(iii) To the Knowledge of Parent, no current or former stockholder, officer, director or employee of Parent or any of its Subsidiaries has any claim, right (whether currently
exercisable, or exercisable in the future), or interest to or in any Parent IP Rights purported to be owned by Parent. To the Knowledge of Parent, no employee of Parent or any of its Subsidiaries is (a) bound by or otherwise subject to any Contract
restricting him or her from performing his or her duties for Parent or such Subsidiary or (b) in breach of any Contract with any former employer or other Person concerning Parent IP Rights purported to be owned by Parent or such Subsidiary or
confidentiality provisions protecting trade secrets and confidential information comprising Parent IP Rights purported to be owned by Parent or such Subsidiary.
(iv) No funding, facilities or personnel of any Governmental Authority were used, directly or indirectly, to develop or create, in whole or in part, any Parent IP Rights in which
Parent or any of its Subsidiaries has an ownership interest.
(v) Parent and each of its Subsidiaries has taken reasonable steps to maintain the confidentiality of and otherwise protect and enforce its rights in all proprietary information
that Parent or such Subsidiary holds, or purports to hold, as confidential or a trade secret.
(vi) Parent or any of its Subsidiaries has not assigned or otherwise transferred ownership of, or agreed to assign or otherwise transfer ownership of, any Parent IP Rights to any
other Person.
(f) Parent has delivered, or made available to the Company, a complete and accurate copy of all material Parent IP Rights Agreements.
(g) The manufacture, marketing, offering for sale, sale, importation, use or intended use or other disposal of any product as currently sold or under development by Parent does not violate any license
or agreement between Parent or its Subsidiaries and any third party in any material respect, and, to the Knowledge of Parent, does not infringe or misappropriate any valid and issued Patent right or other Intellectual Property of any other Person,
which infringement or misappropriation would reasonably be expected to have a Parent Material Adverse Effect. To the Knowledge of Parent, no third party is infringing upon any Patents owned by Parent within the Parent IP Rights, or violating any Parent
IP Rights Agreement.
44
(h) As of the date of this Agreement, Parent is not a party to any Legal Proceeding (including, but not limited to, opposition, interference or other proceeding in any patent or other government
office) contesting the validity, ownership or right to use, sell, offer for sale, license or dispose of any Parent IP Rights. Parent has not received any written notice asserting that any Parent Registered IP or the proposed use, sale, offer for sale,
license or disposition of any products, methods or processes claimed or covered thereunder infringes or misappropriates or violates the rights of any other Person or that Parent or any of its Subsidiaries have otherwise infringed, misappropriated or
otherwise violated any Intellectual Property of any Person.
(i) To the Knowledge of Parent, no trademark (whether registered or unregistered) or trade name owned, used or applied for by Parent conflicts or interferes with any trademark (whether registered or
unregistered) or trade name owned, used or applied for by any other Person except as would not have a Parent Material Adverse Effect. None of the goodwill associated with or inherent in any trademark (whether registered or unregistered) in which Parent
has or purports to have an ownership interest has been impaired as determined by Parent in accordance with GAAP.
(j) Except as may be set forth in the Contracts listed on Section 4.12(b), 4.12(c) or 4.12(k) of the Parent Disclosure Letter or as contained in “off-the-shelf” license
agreements entered into in the Ordinary Course of Business by Parent, (i) Parent is not bound by any Contract to indemnify, defend, hold harmless or reimburse any other Person with respect to any Intellectual Property infringement, misappropriation or
similar claim which is material to Parent taken as a whole and (ii) Parent has never assumed, or agreed to discharge or otherwise take responsibility for, any existing or potential liability of another Person for infringement, misappropriation or
violation of any Intellectual Property right, which assumption, agreement or responsibility remains in force as of the date of this Agreement.
(k) Neither Parent nor any of its Subsidiaries is party to any Contract that, as a result of such execution, delivery and performance of this Agreement, will cause the grant of any license or other
right to any Parent IP Rights, result in breach of, default under or termination of such Contract with respect to any Parent IP Rights, or impair the right of Parent or the Surviving Entity and its Subsidiaries to use, sell or license or enforce any
Parent IP Rights or portion thereof, except for the occurrence of any such grant or impairment that would not individually or in the aggregate, reasonably be expected to result in a Parent Material Adverse Effect.
4.13 Agreements, Contracts and Commitments.
(a) Section 4.13(a) of the Parent Disclosure Letter identifies each Parent Contract that is in effect as of the date of this Agreement (each, an “Parent
Material Contract” and collectively, the “Parent Material Contracts”):
(i) each Parent Contract relating to any material bonus, deferred compensation, severance, incentive compensation, pension, profit-sharing or retirement plans, or any other employee
benefit plans or arrangements;
(ii) each Parent Contract requiring payments by Parent after the date of this Agreement in excess of $100,000 pursuant to its express terms relating to the employment of, or the
performance of employment-related services by, any Parent Associate providing employment related, consulting or independent contractor services, not terminable by Parent on thirty (30) calendar days or less notice without liability;
45
(iii) each Parent Contract relating to any agreement or plan, including any option plan, stock appreciation right plan or stock purchase plan, any of the benefits of which will be
increased or the vesting of benefits of which will be accelerated, by the occurrence of any of the Contemplated Transactions (either alone or in conjunction with any other event, such as termination of employment), or the value of any of the benefits
of which will be calculated on the basis of any of the Contemplated Transactions;
(iv) each Parent Contract relating to any agreement of indemnification or guaranty not entered into in the Ordinary Course of Business;
(v) each Parent Contract containing (A) any covenant limiting the freedom of Parent or any of its Subsidiaries to engage in any line of business or compete with any Person, or
limiting the development, manufacture or distribution of the Parent’s products or services (B) any most-favored pricing arrangement, (C) any exclusivity provision or (D) any non-solicitation provision;
(vi) each Parent Contract (A) pursuant to which any Person granted Parent an exclusive license under any Intellectual Property, or (B) pursuant to which Parent granted any Person an
exclusive license under any Parent IP Rights;
(vii) each Parent Contract containing any royalty, dividend or similar arrangement based on the revenues or profits of Parent, any of its Subsidiaries, or of a product;
(viii) each Parent Contract relating to capital expenditures and requiring payments after the date of this Agreement in excess of $100,000 pursuant to its express terms and not
cancelable without penalty;
(ix) each Parent Contract relating to the disposition or acquisition of material assets or any ownership interest in any Entity, in each case, involving payments in excess of
$100,000 after the date of this Agreement;
(x) each Parent Contract entered into in settlement of any Legal Proceeding or other dispute pursuant to which Parent or any of its Subsidiaries has outstanding obligations to pay
consideration in excess of $100,000;
(xi) each Parent Contract relating to any mortgages, indentures, loans, notes or credit agreements, security agreements or other agreements or instruments relating to the borrowing
of money or extension of credit in excess of $100,000 or creating any material Encumbrances with respect to any assets of Parent or any loans or debt obligations with officers or directors of Parent;
(xii) each Parent Contract requiring payment by or to Parent after the date of this Agreement in excess of $100,000 pursuant to its express terms relating to: (A) any distribution
agreement (identifying any that contain exclusivity provisions), (B) any agreement involving provision of services or products with respect to any pre-clinical or clinical development activities of Parent, (C) any dealer, distributor, joint marketing,
alliance, joint venture, cooperation, development or other agreement currently in force under which Parent or any of its Subsidiaries has continuing obligations to develop or market any product, technology or service, or any agreement pursuant to which
Parent or any of its Subsidiaries has continuing obligations to develop any Intellectual Property that will not be owned, in whole or in part, by Parent or such Subsidiary or (D) any Contract to license any patent, trademark registration, service mark
registration, trade name or copyright registration to or from any third party to manufacture or produce any product, service or technology of Parent or any of its Subsidiaries or any Contract to sell, distribute or commercialize any products or service
of Parent or any of its Subsidiaries, in each case, except for Parent Contracts entered into in the Ordinary Course of Business;
46
(xiii) each Parent Contract with any Person, including any financial advisor, broker, finder, investment banker or other Person, providing advisory services to Parent in connection
with the Contemplated Transactions and requiring payments by Parent after the date in this Agreement in excess of $100,000 pursuant to its express terms;
(xiv) each Parent Contract to which Parent or any of its Subsidiaries is a party or by which any of their assets and properties is currently bound (other than Parent Real Estate
Leases), which involves annual obligations of payment by, or annual payments to, Parent or such Subsidiary in excess of $100,000;
(xv) any Parent Real Estate Lease;
(xvi) a Contract disclosed in or required to be disclosed in Section 4.12(b) of the Parent Disclosure Letter; or
(xvii) any other Parent Contract (other than Parent Real Estate Leases) that is not terminable at will (with no penalty or payment) by Parent or any of its Subsidiaries, and (A)
which involves payment or receipt by Parent or such Subsidiary after the date of this Agreement under any such agreement, contract or commitment of more than $100,000 in the aggregate, or obligations after the date of this Agreement in excess of
$100,000 in the aggregate or (B) that is material to the business or operations of Parent and its Subsidiaries taken as a whole.
(b) Parent has delivered or made available to the Company accurate and complete copies of all Parent Material Contracts, including all amendments thereto. There are no Parent Material Contracts that
are not in written form. Parent has not nor, to Parent’s Knowledge as of the date of this Agreement, has any other party to a Parent Material Contract, breached, violated or defaulted under, or received notice that it breached, violated or defaulted
under, any of the terms or conditions of any Parent Material Contract in such a manner, and, if such Parent Material Contract provides for a cure period, Parent or such other party fails to have cured such breach, violation or default, so that any
other party or Parent, as the case may be, is permitted to modify, cancel or terminate any such Parent Material Contract, or would permit any other party to seek damages which would reasonably be expected to have a Parent Material Adverse Effect. As to
Parent and its Subsidiaries, as of the date of this Agreement, each Parent Material Contract is valid, binding, enforceable and in full force and effect, subject to the Enforceability Exceptions. No Person is renegotiating, or has a right pursuant to
the terms of any Parent Material Contract to change, any material amount paid or payable to Parent under any Parent Material Contract or any other material term or provision of any Parent Material Contract.
4.14 Compliance; Permits; Restrictions.
(a) Parent and each of its Subsidiaries is, and since January 1, 2023, has been in material compliance with all applicable Laws. No investigation, claim, suit, proceeding, audit, Order or other action
by any Governmental Authority is pending or, to the Knowledge of Parent, threatened against Parent or any of its Subsidiaries. There is no agreement or Order binding upon Parent or any of its Subsidiaries which (i) has or would reasonably be expected
to have the effect of prohibiting or materially impairing any business practice of Parent or any of its Subsidiaries, any acquisition of material property by Parent or any of its Subsidiaries or the conduct of business by Parent or any of its
Subsidiaries as currently conducted, (ii) is reasonably likely to have an adverse effect on Parent’s ability to comply with or perform any covenant or obligation under this Agreement or (iii) is reasonably likely to have the effect of preventing,
delaying, making illegal or otherwise interfering with the Contemplated Transactions.
47
(b) Except for matters regarding the FDA or other Drug Regulatory Agency, each of Parent and its Subsidiaries holds all required Governmental Authorizations that are material to the operation of the
business of Parent and Merger Sub as currently conducted (collectively, the “Parent Permits”). Section 4.14(b) of the Parent Disclosure Letter identifies each Parent Permit. Each of Parent and its
Subsidiaries is in material compliance with the terms of the Parent Permits. No Legal Proceeding is pending or, to the Knowledge of Parent, threatened, which seeks to revoke, substantially limit, suspend or materially modify any Parent Permit. The
rights and benefits of each Parent Permit, if any, will be available to Parent and Surviving Entity immediately after the Effective Time on terms substantially identical to those enjoyed by Parent and its Subsidiaries as of the date of this Agreement
and immediately prior to the Effective Time.
(c) There are no Legal Proceedings pending or, to the Knowledge of Parent, threatened with respect to an alleged violation by Parent or any of its Subsidiaries of the FDCA, PHSA, FDA regulations
adopted thereunder, the Controlled Substances Act or any other similar Law promulgated by a Drug Regulatory Agency.
(d) Each of Parent and its Subsidiaries holds all required Governmental Authorizations issuable by any Drug Regulatory Agency necessary for the conduct of the business of Parent and Merger Sub as
currently conducted, and, as applicable, the development, testing, manufacturing, processing, storage, labelling, sale, marketing, advertising, distribution and importation or exportation, as currently conducted, of any of its products (“Parent Products”) or candidates (the “Parent Product Candidates”) (the “Parent Regulatory Permits”) and no such Parent Regulatory Permit
has been (i) revoked, withdrawn, suspended, cancelled or terminated or (ii) modified in any adverse manner other than immaterial adverse modifications. Section 4.14(d) of the Parent Disclosure Letter identifies each material Parent
Regulatory Permit. Parent has timely maintained and is in compliance in all material respects with the Parent Regulatory Permits and neither Parent nor or any of its Subsidiaries has, since January 1, 2023, received any written notice or correspondence
or, to the Knowledge of Parent, other communication from any Drug Regulatory Agency regarding (A) any material violation of or failure to comply materially with any term or requirement of any Parent Regulatory Permit or (B) any revocation, withdrawal,
suspension, cancellation, termination or material modification of any Parent Regulatory Permit. Parent has made available to the Company all information requested by the Company in Parent’s or its Subsidiaries’ possession or control relating to Parent
Products or material Parent Product Candidates and the development, testing, manufacturing, processing, storage, labeling, sale, marketing, advertising, distribution and importation or exportation of the Parent Products or Parent Product Candidates,
including, but not limited to, complete copies of the following (to the extent there are any): (x) adverse event reports; pre-clinical, clinical and other study reports and material study data; inspection reports, notices of adverse findings, untitled
letters, warning letters, filings and letters and other written correspondence to and from any Drug Regulatory Agency; and meeting minutes with any Drug Regulatory Agency and (y) similar reports, material study data, notices, letters, filings,
correspondence and meeting minutes with any other Governmental Authority. All such information are accurate and complete in all material respects.
48
(e) All clinical, pre-clinical and other studies and tests conducted by or on behalf of, or sponsored by, Parent or its Subsidiaries, in which Parent or its Subsidiaries or their respective products,
including the Parent Products, or product candidates, including the Parent Product Candidates, have participated were, since January 1, 2023, and, if still pending, are being conducted in accordance in all material respects with applicable medical and
scientific research standards, and in compliance in all material respects with the applicable regulations of the Drug Regulatory Agencies and other applicable Law, including 21 C.F.R. Parts 11, 50, 54, 56, 58, and 312, as applicable. Since January 1,
2023, neither Parent nor any of its Subsidiaries has received any written notices, correspondence, or other communications from any Drug Regulatory Agency requiring or, to the Knowledge of Parent, any action to place a clinical hold order on, or
otherwise terminate, delay or suspend any clinical studies conducted by or on behalf of, or sponsored by, Parent or any of its Subsidiaries or in which Parent or any of its Subsidiaries or its products, including the Parent Products, or current product
candidates, including the Parent Product Candidates, have participated. Further, no clinical investigator, researcher or clinical staff participating in any clinical study conducted by or, to the Knowledge of Parent, on behalf of Parent or any of its
Subsidiaries has been disqualified from participating in studies involving the Parent Products or Parent Product Candidates, and to the Knowledge of Parent, no such administrative action to disqualify such clinical investigators, researchers or
clinical staff has been threatened or is pending.
(f) Neither Parent nor any of its Subsidiaries and, to the Knowledge of Parent, any contract manufacturer with respect to any Parent Product or Parent Product Candidate is the subject of any pending
or, to the Knowledge of Parent, threatened investigation in respect of its business or products by the FDA pursuant to its “Fraud, Untrue Statements of Material Facts, Bribery, and Illegal Gratuities” Final Policy set forth in 56 Fed. Reg. 46191
(September 10, 1991) and any amendments thereto or by any other Drug Regulatory Agency under a comparable policy. Neither the Company, nor any of its officers, directors, employees or agents have been debarred or excluded from participation in any
federal healthcare programs. Neither Parent nor any of its Subsidiaries and, to the Knowledge of Parent, any contract manufacturer, nor their respective officers, employees or agents, with respect to any Parent Product or Parent Product Candidate has
committed any acts, made any statement or failed to make any statement, in each case in respect of its business or products that would violate FDA’s “Fraud, Untrue Statements of Material Facts, Bribery, and Illegal Gratuities” Final Policy, and any
amendments thereto. None of Parent, any of its Subsidiaries, and to the Knowledge of Parent, any contract manufacturer with respect to any Parent Product or Parent Product Candidate, or any of their respective officers, employees or agents is currently
or has been debarred, convicted of any crime or is engaging or has engaged in any conduct that could result in a material debarment or exclusion under (i) 21 U.S.C. Section 335a or (ii) any similar applicable Law. To the Knowledge of Parent, no
material debarment or exclusionary claims, actions, proceedings or investigations in respect of their business or any Parent Product or Parent Product Candidate are pending or threatened against Parent, any of its Subsidiaries, and to the Knowledge of
the Parent, any contract manufacturer with respect to any Parent Product or Parent Product Candidate, or any of its officers, employees or agents.
(g) All manufacturing operations conducted by, or to the Knowledge of Parent, for the benefit of, Parent or its Subsidiaries in connection with any Parent Product or Parent Product Candidate, since
January 1, 2023, have been and are being conducted in compliance in all material respects with applicable Laws, including the FDA’s standards for current good manufacturing practices, including applicable requirements contained in 21 C.F.R. Parts 210
and 211, and the respective counterparts thereof promulgated by Governmental Authorities in countries outside the United States.
(h) None of Parent, any of its Subsidiaries, and to the Knowledge of Parent, any manufacturing site of a contract manufacturer or laboratory, with respect to any Parent Product or Parent Product
Candidate, (i) is subject to a Drug Regulatory Agency shutdown or import or export prohibition or (ii) has received any Form FDA 483, notice of violation, warning letter, untitled letter or similar correspondence or notice from the FDA or other Drug
Regulatory Agency alleging or asserting noncompliance with any applicable Law, in each case, that have not been complied with or closed to the satisfaction of the relevant Drug Regulatory Agency, and, to the Knowledge of Parent, neither the FDA nor any
other Drug Regulatory Agency is considering such action.
49
4.15 Legal Proceedings; Orders.
(a) There is no pending Legal Proceeding and, to the Knowledge of Parent, no Person has threatened to commence any Legal Proceeding: (i) that involves Parent or any of its Subsidiaries or any Parent
Associate (in his or her capacity as such) or any of the material assets owned or used by Parent or any of its Subsidiaries or (ii) that challenges, or that may have the effect of preventing, delaying, making illegal or otherwise interfering with, the
Contemplated Transactions.
(b) There is no Order to which Parent or any of its Subsidiaries, or any of the material assets owned or used by Parent or any of its Subsidiaries is subject. To the Knowledge of Parent, no officer or
other Parent Key Employee or any of its Subsidiaries is subject to any Order that prohibits such officer or employee from engaging in or continuing in any conduct, activity or practice relating to the business of Parent or any of its Subsidiaries or
any material assets owned or used by Parent or any of its Subsidiaries.
4.16 Tax Matters.
(a) Each of Parent and each of its Subsidiaries has timely filed (or caused to be timely filed) all income Tax Returns and all other material Tax Returns required to be filed by it under applicable
Law (taking into account any applicable extensions). All such Tax Returns were true, correct and complete in all material respects. Subject to exceptions as would not be material, no written claim has been made by a Governmental Authority in a
jurisdiction where Parent or any of its Subsidiaries does not file Tax Returns that Parent or any of its Subsidiaries is subject to taxation by that jurisdiction.
(b) All material amounts of Taxes due and owing by Parent or any of its Subsidiaries (whether or not shown on any Tax Return) have been timely paid (taking into account any applicable extensions).
(c) Each of Parent and each of its Subsidiaries has withheld and paid to the appropriate Governmental Authority all material Taxes required to have been withheld and paid in connection with any
amounts paid or owing to any employee, independent contractor, creditor, stockholder or other third party.
(d) There are no Encumbrances for a material amount of Taxes (other Encumbrances described in clause (a) of the definition of “Permitted Encumbrances”) upon any of the assets of Parent or any of its
Subsidiaries.
(e) No deficiencies for a material amount of Taxes with respect to Parent or any of its Subsidiaries have been claimed, proposed or assessed by any Governmental Authority that have not been timely
paid in full. There are no pending (or, based on written notice, threatened) material audits, assessments, examinations or other actions for or relating to any liability in respect of Taxes of Parent or any of its Subsidiaries. Neither Parent nor any
of its Subsidiaries has granted a waiver of any statute of limitations in respect of a material amount of Taxes or an extension of time with respect to a material Tax assessment or deficiency that, in each case, is currently in effect.
(f) Neither Parent nor any of its Subsidiaries has entered into a “closing agreement” as described in section 7121 of the Code (or any comparable provisions of state, local or non-U.S. Law) with any
Governmental Authority.
(g) Neither Parent nor any of its Subsidiaries has been a United States real property holding corporation within the meaning of Section 897(c)(2) of the Code in the last five (5) years.
50
(h) Neither Parent nor any of its Subsidiaries is a party to any Tax allocation, Tax sharing or similar agreement (including indemnity arrangements), other than Ordinary Course Agreements.
(i) Neither Parent nor any of its Subsidiaries has been a member of an affiliated group filing a consolidated U.S. federal income Tax Return (other than a group the common parent of which is Parent).
Neither Parent nor any of its Subsidiaries has any material Liability for the Taxes of any Person (other than Parent or its Subsidiaries) under Treasury Regulations Section 1.1502-6 (or any similar provision of state, local, or foreign law), as a
transferee or successor, or by Contract (other than an Ordinary Course Agreement).
(j) Neither Parent nor any of its Subsidiaries will be required to include any material item of income in, or exclude any material item of deduction from, taxable income, or pay any Taxes, in any
taxable period (or portion thereof) ending after the Closing Date to the extent such item is economically attributable to a taxable period (or portion thereof) ending on or before the Closing Date, including as a result of: (i) any change in, or use of
an improper, method of accounting prior to the Closing, (ii) any “closing agreement” as described in Section 7121 of the Code (or any similar or analogous provision of state, local or non-U.S. Law) executed on or prior to the Closing Date, (iii)
instalment sale or open transaction disposition made on or prior to the Closing, (iv) any prepaid amount or advance payment received or deferred revenue accrued on or prior to the Closing Date, or (v) intercompany transactions occurring on or prior to
the Closing Date or any excess loss account in existence on the Closing Date described in Treasury Regulations under Section 1502 of the Code (or any similar or analogous provision of state, local or non-U.S. Law).
(k) Neither Parent nor any of its Subsidiaries is subject to, nor have Parent or any of its Subsidiaries applied for, any private letter ruling of the Internal Revenue Service, request for
administrative relief, request for technical advice, request for a change of any method of Tax accounting, or comparable rulings concerning Tax matters of any Governmental Authority. Neither Parent nor any of its Subsidiaries have granted to any
Person any power of attorney that will remain in force after the Closing with respect to any Tax matter.
(l) Neither Parent nor any of its Subsidiaries has distributed stock of another Person, or has had its stock distributed by another Person, in a transaction that was purported or intended to be
governed in whole or in part by Section 355 of the Code or Section 361 of the Code.
(m) Neither Parent nor any of its Subsidiaries has entered into any “reportable transaction” within the meaning of Section 6707A of the Code or Treasury Regulations Sections 1.6011-4(b).
(n) Schedule 4.16(n) sets forth the U.S. federal income tax classifications of each of Parent and its Subsidiaries.
(o) Any related party transactions conducted by Parent and each of its Subsidiaries have been on an arms-length basis in accordance with Section 482 of the Code or, to the extent applicable, any
analogous, corresponding or similar provision of applicable Law in all material respects.
(p) Neither Parent nor any of its Subsidiaries is aware of any facts or circumstances or has taken or agreed to take any action, in each case, that would reasonably be expected to prevent or impede
the Intended Tax Treatment.
51
4.17 Employee and Labor Matters; Benefit Plans.
(a) The Parent has made available to Company a list setting forth, for each Parent Associate who is an employee of Parent or any of its Subsidiaries, such employee’s name, employer, title, hire date,
location, whether full- or part-time, whether active or on leave (and, if on leave, the expected return), whether exempt from the Fair Labor Standards Act and applicable state law, annual salary (or if hourly, hourly rate), most recent annual bonus
received and current annual bonus opportunity. The Parent has made available to Company a list setting forth, for each Parent Associate who is an individual independent contractor engaged by Parent or any of its Subsidiaries, such contractor’s name,
duties and rate of compensation.
(b) The employment of Parent’s employees is terminable by Parent at will. Parent has made available to the Company accurate and complete copies of all employee manuals and handbooks, to the extent
currently effective and material.
(c) Parent is not a party to, bound by the terms of, and does not have a duty to bargain under, any collective bargaining agreement or other Contract with a labor organization representing any of its
employees, and there are no labor organizations representing or, to the Knowledge of Parent, purporting to represent or seeking to represent any employees of Parent.
(d) Section 4.17(d) of the Parent Disclosure Letter lists all Parent Employee Plans (other than employment arrangements which are terminable “at will” without any contractual
obligation on the part of Parent or any of its Subsidiaries to make any severance, termination, change in control or similar payment and that are substantively identical to the employment arrangements made available to the Company).
(e) Each Parent Employee Plan that is intended to be qualified under Section 401(a) of the Code has received a favorable determination or opinion letter with respect to such qualified status from the
IRS. To the Knowledge of Parent, nothing has occurred that would reasonably be expected to adversely affect the qualified status of any such Parent Employee Plan or the exempt status of any related trust.
(f) Each Parent Employee Plan has been established, maintained and operated in compliance, in all material respects, with its terms all applicable Law, including, without limitation, the Code, ERISA
and the Affordable Care Act. No Legal Proceeding (other than those relating to routine claims for benefits) is pending or, to the Knowledge of Parent, threatened in writing with respect to any Parent Employee Plan. All payments and/or contributions
required to have been made with respect to all Parent Employee Plans either have been made or have been accrued in accordance with the terms of the applicable Parent Employee Plan and applicable Law.
(g) Neither Parent nor any of its ERISA Affiliates maintains, contributes to or is required to contribute to, or has, in the past six (6) years, maintained, contributed to or been required to
contribute to (i) any defined benefit pension plan under Section 3(35) of ERISA that is or was subject to Title IV or Section 302 of ERISA or Section 412 of the Code, (ii) a Multiemployer Plan, (iii) any funded welfare benefit plan within the meaning
of Section 419 of the Code, (iv) any Multiple Employer Plan, or (v) any Multiple Employer Welfare Arrangement. Neither Parent nor any of its ERISA Affiliates has incurred any liability under Title IV of ERISA in the past six years.
(h) No Parent Employee Plan provides for medical or other welfare benefits to any service provider beyond termination of service or retirement, other than (1) pursuant to COBRA or an analogous state
law requirement or (2) continuation coverage through the end of the month in which such termination or retirement occurs. Parent does not sponsor or maintain any self-funded medical or long-term disability benefit plan.
52
(i) No Parent Employee Plan is subject to any law of a foreign jurisdiction outside of the United States.
(j) Each Parent Employee Plan that constitutes in any part a “nonqualified deferred compensation plan” (as such term is defined under Section 409A(d)(1) of the Code and the guidance thereunder)
(each, a “Parent 409A Plan”) has been operated and maintained in all material respects in operational and documentary compliance with the requirements of Section 409A of the Code and the applicable guidance
thereunder. No payment to be made under any Parent 409A Plan is or, when made in accordance with the terms of the Parent 409A Plan, will be subject to the penalties of Section 409A(a)(1) of the Code.
(k) Parent is in material compliance with all Employment-Related Laws and in each case, with respect to the employees of Parent: (i) has withheld and reported all material amounts required by law
or by agreement to be withheld and reported with respect to wages, salaries and other payments to employees, (ii) is not liable for any material amounts of arrears of wages, severance pay or any Taxes or any penalty for failure to comply with any
of the foregoing and (iii) is not liable for any material payment to any trust or other fund governed by or maintained by or on behalf of any Governmental Authority, with respect to unemployment compensation benefits, social security or other
benefits or obligations for employees (other than routine payments to be made in the Ordinary Course of Business). There are no material Legal Proceedings, claims, labor disputes or organizing activities, or grievances pending or, to the Knowledge
of Parent, threatened or reasonably anticipated against or involving Parent or any trustee of Parent relating to any employee, contingent worker, director, employment agreement or Parent Employee Plan (other than routine claims for benefits) or
Employment-Related Laws. To the Knowledge of Parent, there are no material pending or threatened or reasonably anticipated claims or actions against Parent, any Parent trustee or any trustee of any Subsidiary of Parent under any workers’
compensation policy or long-term disability policy. Parent is not a party to a conciliation agreement, consent decree or other agreement or Order with any federal, state or local agency or Governmental Authority with respect to employment
practices.
(l) Parent has no material liability with respect to any misclassification within the past three (3) years of: (i) any Person as an independent contractor rather than as an employee, (ii) any
employee leased from another employer or (iii) any employee currently or formerly classified as exempt from overtime wages. Parent has not taken any action which would constitute a “plant closing” or “mass layoff” within the meaning of the WARN
Act, issued any notification of a plant closing or mass layoff required by the WARN Act (nor has Parent been under any requirement or obligation to issue any such notification), or incurred any liability or obligation under the WARN Act that
remains unsatisfied.
(m) To the Knowledge of Parent, there has never been, nor has there been any threat of, any strike, slowdown, work stoppage, lockout, job action, union, organizing activity, question concerning
representation or any similar activity or dispute, with respect to any Parent Associate. No event has occurred within the past six months, and no condition or circumstance exists, that, to the Knowledge of Parent, might directly or indirectly be
likely to give rise to or provide a basis for the commencement of any such strike, slowdown, work stoppage, lockout, job action, union organizing activity, question concerning representation or any similar activity or dispute.
(n) Parent is not, nor has Parent been, engaged in any material unfair labor practice within the meaning of the National Labor Relations Act. There is no material Legal Proceeding, claim, labor
dispute or grievance pending or, to the Knowledge of Parent, threatened or reasonably anticipated relating to any employment contract, privacy right, labor dispute, wages and hours, leave of absence, plant closing notification, workers’
compensation policy, long-term disability policy, harassment, retaliation, immigration, employment statute or regulation, safety or discrimination matter involving any current or former employee of Parent, including charges of unfair labor
practices or discrimination complaints.
53
(o) There is no contract, agreement, plan or arrangement to which Parent or any of its Subsidiaries is a party or by which it is bound to compensate any of its employees or other service providers
for any income or excise taxes paid pursuant to the Code, including, but not limited to, Section 4999 or Section 409A of the Code.
(p) Neither Parent nor any of its Subsidiaries is a party to any Contract that as a result of the execution and delivery of this Agreement, the stockholder approval of this Agreement, nor the
consummation of the transactions contemplated hereby, could (either alone or in conjunction with any other event) (i) result in the payment of any “parachute payment” within the meaning of Section 280G of the Code or (ii) result in, or cause the
accelerated vesting, payment, funding or delivery of, or increase the amount or value of, any payment or benefit to any employee, officer, director or other service provider of Parent or any of its Subsidiaries.
4.18 Environmental Matters. Since January 1, 2023, Parent and each of its Subsidiaries has complied with all applicable Environmental Laws, which compliance includes the possession by
Parent of all permits and other Governmental Authorizations required under applicable Environmental Laws and compliance with the terms and conditions thereof, except for any failure to be in compliance that, individually or in the aggregate, would
not result in a Parent Material Adverse Effect. Neither Parent nor any of its Subsidiaries has received since January 1, 2023, any written notice or other communication (in writing or otherwise), whether from a Governmental Authority, citizens
group, employee or otherwise, that alleges that Parent or any of its Subsidiaries is not in compliance with any Environmental Law, and, to the Knowledge of Parent, there are no circumstances that may prevent or interfere with Parent’s or any of its
Subsidiaries’ compliance with any Environmental Law in the future, except where such failure to comply would not reasonably be expected to have a Parent Material Adverse Effect. To the Knowledge of Parent: (i) no current or prior owner of any
property leased or controlled by Parent or any of its Subsidiaries has received since January 1, 2023, any written notice or other communication relating to property owned or leased at any time by Parent or any of its Subsidiaries, whether from a
Governmental Authority, citizens group, employee or otherwise, that alleges that such current or prior owner or Parent or any of its Subsidiaries is not in compliance with or violated any Environmental Law relating to such property and (ii) neither
Parent nor any of its Subsidiaries has any material liability under any Environmental Law. Parent has made available all environmental site assessments, environmental audits and other material environmental documents in the Parent’s possession or
control relating to the Parent and its Subsidiaries, including the Parent’s and its Subsidiaries’ business and current or former facilities.
4.19 Insurance. Parent has delivered to the Company accurate and complete copies of all material insurance policies and all material self-insurance programs and arrangements relating to the
business, assets, liabilities and operations of Parent and its Subsidiaries (including Merger Sub). Each of such insurance policies is in full force and effect and Parent and its Subsidiaries (including Merger Sub) are in compliance in all material
respects with the terms thereof. Other than customary end of policy notifications from insurance carriers, since January 1, 2023, neither Parent nor any of its Subsidiaries has received any notice or other communication regarding any actual or
possible: (i) cancellation or invalidation of any insurance policy or (ii) refusal or denial of any coverage, reservation of rights or rejection of any material claim under any insurance policy. Each of Parent and its Subsidiaries (including Merger
Sub) has provided timely written notice to the appropriate insurance carrier(s) of each Legal Proceeding pending against Parent or such Subsidiary for which Parent or such Subsidiary has insurance coverage, and no such carrier has issued a denial
of coverage or a reservation of rights with respect to any such Legal Proceeding, or informed Parent or any of its Subsidiaries of its intent to do so.
54
4.20 Transactions with Affiliates. Except as set forth in the Parent SEC Documents filed prior to the date of this Agreement, since the date of Parent’s last proxy statement filed with the
SEC, no event has occurred that would be required to be reported by Parent pursuant to Item 404 of Regulation S-K promulgated by the SEC. Section 4.20 of the Parent Disclosure Letter identifies each Person who is (or who may be
deemed to be) an Affiliate of Parent as of the date of this Agreement.
4.21 No Financial Advisors. Except as set forth on Section 4.21 of the Parent Disclosure Letter, no broker, finder or investment banker is entitled to any brokerage fee,
finder’s fee, opinion fee, success fee, transaction fee or other fee or commission in connection with the Contemplated Transactions based upon arrangements made by or on behalf of Parent.
4.22 Valid Issuance. The Parent Capital Stock to be issued in the Merger will, when issued in accordance with the provisions of this Agreement, be validly issued, fully paid and
nonassessable.
4.23 Privacy and Data Security. Parent and its Subsidiaries are and since January 1, 2023, have been in compliance with all applicable Privacy Laws and the applicable terms of any Parent
Contracts governing privacy, data protection, data security, trans-border data flow, data loss, data theft, or breach notification, data localization, sending solicited or unsolicited electronic mail or text messages, cookies or other tracking
technology, with respect to, or the collection, handling, use, maintenance, storage, disclosure, transfer, or other processing of, Personal Information (including any such information of individuals, clinical trial participants, patients, patient
family members, caregivers or advocates, physicians and other health care professionals, clinical trial investigators, researchers, pharmacists that interact with Parent or any of its Subsidiaries in connection with the operation of Parent’s and
its Subsidiaries’ business), except, in each case, for such noncompliance as has not had, and would not reasonably be expected to have, individually or in the aggregate, a Parent Material Adverse Effect. To the Knowledge of Parent, Parent (i) has
implemented and maintains reasonable Privacy Policies that materially comply with applicable Privacy Laws and are designed to protect the privacy and security of Personal Information and (ii) has complied with such Privacy Policies, except for such
noncompliance as has not had, and would not reasonably be expected to have, individually or in the aggregate, a Parent Material Adverse Effect. To the Knowledge of Parent, no Legal Proceeding has been asserted or threatened against Parent by any
Person alleging a violation of Privacy Laws, Privacy Policies, or the applicable terms of any Parent Contracts governing privacy, data protection, data security, trans-border data flow, data loss, data theft, or breach notification, data
localization, sending solicited or unsolicited electronic mail or text messages, cookies or other tracking technology, with respect to, or the collection, handling, use, maintenance, storage, disclosure, transfer, or other processing of, Personal
Information. To the Knowledge of Parent, there have been no data security incidents or data breaches, or other adverse events or incidents that have resulted in any unauthorized access to, or collection, use, disclosure, modification or destruction
of, Personal Information or other data in the possession or control of Parent or any service provider acting on behalf of Parent, in each case, where such incident, breach, or event has resulted in a notification obligation to any Person under
applicable Law or pursuant to the terms of any Parent Contract.
4.24 No Other Representations or Warranties. Parent hereby acknowledges and agrees that, except for the representations and warranties contained in this Agreement, neither the Company nor
any of its Subsidiaries nor any other person on behalf of the Company or its Subsidiaries makes any express or implied representation or warranty with respect to the Company or its Subsidiaries or with respect to any other information provided to
Parent, Merger Sub or stockholders or any of their respective Affiliates in connection with the Contemplated Transactions, and (subject to the express representations and warranties of the Company set forth in Section 3 (in each case as
qualified and limited by the Company Disclosure Letter)) none of Parent, Merger Sub nor any of their respective Representatives or stockholders, has relied on any such information (including the accuracy or completeness thereof).
55
Section 5. Certain Covenants of the Parties.
5.1 Operation of Parent’s Business.
(a) Except (i) as expressly contemplated or permitted by this Agreement, (ii) as set forth in Section 5.1(a) of the Parent Disclosure Letter, (iii) as required by applicable Law,
or (iv) unless the Company shall otherwise consent in writing (which consent shall not be unreasonably withheld, delayed or conditioned), during the period commencing on the date of this Agreement and continuing until the earlier to occur of the
termination of this Agreement pursuant to Section 10 and the Effective Time (the “Pre-Closing Period”), Parent shall, and shall cause its Subsidiaries to, use commercially reasonable efforts to (x)
conduct its business and operations in the Ordinary Course of Business and in material compliance with all applicable Law and the requirements of all Contracts that constitute Parent Material Contracts and (y) continue to pay material outstanding
accounts payable and other material current Liabilities (including payroll) when due and payable.
(b) Except (i) as expressly contemplated or permitted by this Agreement, (ii) as set forth in Section 5.1(b) of the Parent Disclosure Letter, (iii) as required by applicable Law,
or (iv) with the prior written consent of the Company (which consent shall not be unreasonably withheld, delayed or conditioned), at all times during the Pre-Closing Period, Parent shall not, nor shall it cause or permit any of Subsidiaries to, do
any of the following:
(i) declare, accrue, set aside or pay any dividend or make any other distribution in respect of any shares of its capital stock or repurchase, redeem or otherwise reacquire any
shares of its capital stock or other securities, (except for shares of Parent Common Stock from terminated employees, directors or consultants of Parent);
(ii) except as required to give effect to anything in contemplation of the Closing, amend any of its Organizational Documents, or effect or be a party to any merger,
consolidation, share exchange, business combination, recapitalization, reclassification of shares, stock split, reverse stock split or similar transaction except, for the avoidance of doubt, the Contemplated Transactions;
(iii) sell, issue, grant or authorize the issuance of: (A) any capital stock or other security (except for Parent Common Stock issued upon the valid exercise or settlement of
outstanding Parent Options), (B) any option, warrant or right to acquire any capital stock or any other security or (C) any instrument convertible into or exchangeable for any capital stock or other security;
(iv) acquire any equity interest or other interest in any other Entity or enter into a joint venture with any other Entity;
(v) (A) lend money to any Person, (B) incur or guarantee any indebtedness for borrowed money, or (C) guarantee any debt securities of others ;
(vi) acquire any material asset or sell, lease, license or otherwise irrevocably dispose of any of its assets or properties, or grant any Encumbrance with respect to such assets
or properties;
(vii) sell, assign, transfer, license, sublicense or otherwise dispose of any material Parent IP Rights (other than pursuant to non-exclusive licenses in the Ordinary Course of
Business);
56
(viii) waive, settle or compromise any pending or threatened Legal Proceeding against Parent or any of its Subsidiaries, other than waivers, settlements or agreements (A) for an
amount not in excess of $100,000 in the aggregate (excluding amounts to be paid under existing insurance policies or renewals thereof) and (B) that do not impose any material restrictions on the operations or businesses of Parent or its
Subsidiaries, taken as a whole, or any equitable relief on, or the admission of wrongdoing by Parent or any of its Subsidiaries;
(ix) enter into, amend in a manner adverse to Parent or terminate any Parent Material Contract outside of the Ordinary Course of Business;
(x) make any capital expenditures, capital additions or capital improvements, or enter into any contract, exceeding $250,000 individually or in the aggregate;
(xi) (A) adopt, establish or enter into any Parent Employee Plan, including, for the avoidance of doubt, any equity awards plans, (B) cause or permit any Parent Employee Plan to
be amended other than as required by law or in order to make amendments for the purposes of compliance with Section 409A of the Code, (C) pay any bonus or make any profit-sharing or similar payment to (except with respect to obligations in place on
the date of this Agreement pursuant to any Parent Employee Plan disclosed to the Company), or increase the amount of the wages, salary, commissions, fringe benefits or other compensation or remuneration payable to, any of its directors, officers,
employees or consultants, (D) increase the severance or change of control benefits offered to any current or new employees, directors or consultants, or (E) hire any officer, employee or consultant;
(xii) agree, resolve or commit to do any of the foregoing.
Nothing contained in this Agreement shall give the Company, directly or indirectly, the right to control or direct the operations of Parent prior to the Effective Time. Prior to the Effective Time, Parent shall
exercise, consistent with the terms and conditions of this Agreement, complete unilateral control and supervision over its business operations.
5.2 Operation of the Company’s Business.
(a) Except (i) as expressly contemplated or permitted by this Agreement, (ii) as set forth in Section 5.2(a) of the Company Disclosure Letter, (iii) as required by applicable Law,
or (iv) unless Parent shall otherwise consent in writing (which consent shall not be unreasonably withheld, delayed or conditioned), during the Pre-Closing Period the Company shall, and shall cause its Subsidiaries to, use commercially reasonable
efforts to (x) conduct its business and operations in the Ordinary Course of Business and in material compliance with all applicable Law and the requirements of all Contracts that constitute Company Material Contracts and (y) continue to pay
material outstanding accounts payable and other material current Liabilities (including payroll) when due and payable.
(b) Except (i) as expressly contemplated or permitted by this Agreement, (ii) as set forth in Section 5.2(b) of the Company Disclosure Letter, (iii) as required by applicable Law,
or (iv) with the prior written consent of Parent (which consent shall not be unreasonably withheld, delayed or conditioned), at all times during the Pre-Closing Period, the Company shall not, nor shall it cause or permit any of its Subsidiaries to,
do any of the following:
(i) declare, accrue, set aside or pay any dividend or make any other distribution in respect of any shares of its capital stock; or repurchase, redeem or otherwise reacquire any
shares of Company Capital Stock or other securities (except for shares of Company Common Stock from terminated employees, directors or consultants of the Company);
57
(ii) except as required to give effect to anything in contemplation of the Closing, amend any of its or its Subsidiaries’ Organizational Documents, or effect or be a party to
any merger, consolidation, share exchange, business combination, recapitalization, reclassification of shares, stock split, reverse stock split or similar transaction except, for the avoidance of doubt, the Contemplated Transactions;
(iii) sell, issue grant, or authorize any of the foregoing actions with respect to more than 25% of the shares of Company Capital Stock outstanding as of the date of this
Agreement: (A) any capital stock or other security of the Company or any of its Subsidiaries (except for shares of outstanding Company Common Stock issued upon the valid exercise of Company Options), (B) any option, warrant or right to acquire any
capital stock or any other security or (C) any instrument convertible into or exchangeable for any capital stock or other security of the Company or any of its Subsidiaries;
(iv) acquire any equity interest or other interest in any other Entity or enter into a joint venture with any other Entity;
(v) (A) lend money to any Person, (B) incur or guarantee any indebtedness for borrowed money, or (C) guarantee any debt securities of others;
(vi) acquire any material asset or sell, lease, license or otherwise irrevocably dispose of any of its assets or properties, or grant any Encumbrance with respect to such assets
or properties;
(vii) sell, assign, transfer, license, sublicense or otherwise dispose of any material Company IP Rights (other than pursuant to non-exclusive licenses in the Ordinary Course of
Business);
(viii) waive, settle or compromise any pending or threatened Legal Proceeding against the Company, other than waivers, settlements or agreements (A) for an amount not in excess
of $100,000 in the aggregate (excluding amounts to be paid under existing insurance policies or renewals thereof) and (B) that do not impose any material restrictions on the operations or businesses of the Company or any equitable relief on, or the
admission of wrongdoing by the Company;
(ix) enter into, amend in a manner adverse to the Company or terminate any Company Material Contract; or
(x) agree, resolve or commit to do any of the foregoing.
Nothing contained in this Agreement shall give Parent, directly or indirectly, the right to control or direct the operations of the Company prior to the Effective Time. Prior to the Effective Time,
the Company shall exercise, consistent with the terms and conditions of this Agreement, complete unilateral control and supervision over its business operations.
5.3 Access and Investigation.
58
(a) Subject to the terms of the Confidentiality Agreement, which the Parties agree will continue in full force following the date of this Agreement, during the Pre-Closing Period, upon reasonable
notice, Parent, on the one hand, and the Company, on the other hand, shall and shall use commercially reasonable efforts to cause such Party’s Representatives to: (a) provide the other Party and such other Party’s Representatives with reasonable
access during normal business hours to such Party’s Representatives, personnel, property and assets and to all existing books, records, Tax Returns, work papers and other documents and information relating to such Party and its Subsidiaries, (b)
provide the other Party and such other Party’s Representatives with such copies of the existing books, records, Tax Returns, work papers, product data, and other documents and information relating to such Party and its Subsidiaries, and with such
additional financial, operating and other data and information regarding such Party and its Subsidiaries as the other Party may reasonably request, (c) permit the other Party’s officers and other employees to meet, upon reasonable notice and during
normal business hours, with the chief financial officer and other officers and managers of such Party responsible for such Party’s financial statements and the internal controls of such Party to discuss such matters as the other Party may deem
necessary, and (d) make available to the other Party copies of any material notice, report or other document filed with or sent to or received from any Governmental Authority in connection with the Contemplated Transactions. Any investigation
conducted by either Parent or the Company pursuant to this Section 5.3 shall be conducted in such manner as not to interfere unreasonably with the conduct of the business of the other Party.
(b) Notwithstanding anything herein to the contrary in this Section 5.3, no access or examination contemplated by this Section 5.3 shall be permitted to the extent
that it would require any Party or its Subsidiaries to waive the attorney-client privilege or attorney work product privilege, or violate any applicable Law; provided, that such Party or its Subsidiary (i) shall be entitled to withhold only
such information that may not be provided without causing such violation or waiver, (ii) shall provide to the other Party all related information that may be provided without causing such violation or waiver (including, to the extent permitted,
redacted versions of any such information) and (iii) shall enter into such effective and appropriate joint-defense agreements or other protective arrangements as may be reasonably requested by the other Party in order that all such information may
be provided to the other Party without causing such violation or waiver.
5.4 No Solicitation.
(a) The Company agrees that, during the Pre-Closing Period, neither it nor any of its Subsidiaries shall, nor shall it or any of its Subsidiaries authorize or permit any of its Representatives to,
directly or indirectly: (i) solicit, initiate or knowingly encourage, induce or facilitate the communication, making, submission or announcement of any Acquisition Proposal or Acquisition Inquiry, (ii) furnish any non-public information regarding
such Party to any Person in connection with or in response to an Acquisition Proposal or Acquisition Inquiry, (iii) engage in discussions or negotiations with any Person with respect to any Acquisition Proposal or Acquisition Inquiry, (iv) execute
or enter into any letter of intent or any Contract contemplating or otherwise relating to any Acquisition Transaction or (v) publicly propose to do any of the foregoing. Without limiting the generality of the foregoing, the Company acknowledges and
agrees that, in the event any Representative of the Company takes any action that, if taken by the Company, would constitute a breach of this Section 5.4 by the Company, the taking of such action by such Representative shall be
deemed to constitute a breach of this Section 5.4 by the Company for purposes of this Agreement.
(b) If the Company or any of its Representatives receives an Acquisition Proposal or Acquisition Inquiry at any time during the Pre-Closing Period, then the Company shall promptly (and in no event
later than one (1) Business Day after the Company becomes aware of such Acquisition Proposal or Acquisition Inquiry) advise Parent in writing of such Acquisition Proposal or Acquisition Inquiry (including the identity of the Person making or
submitting such Acquisition Proposal or Acquisition Inquiry, and the terms thereof). The Company shall keep Parent reasonably informed with respect to the status and terms of any such Acquisition Proposal or Acquisition Inquiry and any material
modification or material proposed modification thereto.
59
(c) The Company shall immediately cease and cause to be terminated any existing discussions, negotiations and communications with any Person that relate to any Acquisition Proposal or Acquisition
Inquiry as of the date of this Agreement and request the destruction or return of any nonpublic information provided to such Person promptly following the execution and delivery of this Agreement.
5.5 Notification of Certain Matters. During the Pre-Closing Period, each of the Company, on the one hand, and Parent, on the other hand, shall promptly notify the other (and, if in writing,
furnish copies of) if any of the following occurs: (a) any notice or other communication is received from any Person alleging that the Consent of such Person is or may be required in connection with any of the Contemplated Transactions, (b) any
Legal Proceeding against or involving or otherwise affecting such Party or its Subsidiaries is commenced, or, to the Knowledge of such Party, threatened against such Party or, to the Knowledge of such Party, any director or officer of such Party,
(c) such Party becomes aware of any inaccuracy in any representation or warranty made by such Party in this Agreement or (d) the failure of such Party to comply with any covenant or obligation of such Party; in each case that could reasonably be
expected to make the timely satisfaction of any of the conditions set forth in Section 7, Section 8 or Section 9, as applicable, impossible or materially less likely. No such notice shall be deemed to supplement or amend the
Company Disclosure Letter or the Parent Disclosure Letter for the purpose of (x) determining the accuracy of any of the representations and warranties made by the Company in this Agreement or (y) determining whether any condition set forth in Section
7, Section 8 or Section 9 has been satisfied. Any failure by either Party to provide notice pursuant to this Section 5.5 shall not be deemed to be a breach for purposes of Section 8.2 or Section
9.2, as applicable, unless such failure to provide such notice was knowing and intentional.
Section 6. Additional Agreements of the Parties.
6.1 Proxy Statement.
(a) As promptly as practicable after the Closing Date and in any event on or before the date that is 45 days after the date hereof, Parent shall prepare and file with the SEC a proxy statement
relating to the Parent Stockholder Meeting to be held in connection with the Preferred Stock Conversion Proposal (together with any amendments thereof or supplements thereto, the “Proxy Statement”). Parent
shall use its commercially reasonable efforts to (i) cause the Proxy Statement to comply with applicable rules and regulations promulgated by the SEC and (ii) respond promptly to any comments or requests of the SEC or its staff related to the Proxy
Statement.
(b) Parent covenants and agrees that the Proxy Statement (and the letters to stockholders, notice of meeting and form of proxy included therewith) will (i) comply as to form in all material
respects with the requirements of applicable U.S. federal securities Laws and the DGCL, and (ii) will not contain any untrue statement of a material fact or omit to state any material fact required to be stated therein or necessary in order to make
the statements made therein, in light of the circumstances under which they were made, not misleading.
60
(c) Parent shall use commercially reasonable efforts to cause the Proxy Statement to be mailed to Parent’s stockholders as promptly as practicable after the Proxy Statement has been filed with the
SEC and either (i) the SEC has indicated that it does not intend to review the Proxy Statement or that its review of the Proxy Statement has been completed or (ii) at least ten (10) days shall have passed since the Proxy Statement was filed with
the SEC without receiving any correspondence from the SEC commenting upon, or indicating that it intends to review, the Proxy Statement, all in compliance with applicable U.S. federal securities laws and the DGCL. If Parent, Merger Sub or the
Surviving Entity (A) become aware of any event or information that, pursuant to the Securities Act or the Exchange Act, should be disclosed in an amendment or supplement to the Proxy Statement, (B) receives notice of any SEC request for an
amendment or supplement to the Proxy Statement or for additional information related thereto, or (C) receives SEC comments on the Proxy Statement, as the case may be, then such Party, as the case may be, shall promptly inform the other Parties
thereof and shall cooperate with such other Parties in Parent filing such amendment or supplement with the SEC and, if appropriate, in mailing such amendment or supplement to the Parent stockholders.
(d) The Company shall reasonably cooperate with Parent and provide, and cause its Representatives to provide, Parent and its Representatives, with all true, correct and complete information
regarding the Company that is required by Law to be included in the Proxy Statement or reasonably requested by Parent to be included in the Proxy Statement (collectively, the “Company Required Proxy Information”).
The Company and its legal counsel shall be given reasonable opportunity to review and comment on the Proxy Statement, including all amendments and supplements thereto, prior to the filing thereof with the SEC, and on the response to any comments of
the SEC on the Proxy Statement, prior to the filing thereof with the SEC, and Parent shall give reasonable and good faith consideration to any comments by the Company and its legal counsel. To the extent not prohibited by Law, Parent shall provide
the Company and its legal counsel with (A) any comments or other communications, whether written or oral, that Parent or its legal counsel may receive from time to time from the SEC or its staff with respect to the Proxy Statement promptly after
receipt of those comments or other communications and (B) a reasonable opportunity to participate in the response of Parent to those comments and to provide comments on that response (to which reasonable and good faith consideration shall be
given), including by participating with the Company or its legal counsel in any discussions or meetings with the SEC (to the extent permitted by the SEC). Parent may file the Proxy Statement, or any amendment or supplement thereto, without the
prior consent of the Company; provided, that Parent has included the Company Required Proxy Information in the Proxy Statement in substantially the same form as it was provided to Parent by the Company pursuant to this Section 6.1;
provided, further, that if the prior consent of the Company is not obtained then, notwithstanding anything else herein, the Company makes no covenant or representation regarding the portion of such information supplied by or on
behalf of the Company to Parent for inclusion in such Proxy Statement that the Company reasonably identifies prior to such filing of the Proxy Statement.
6.2 Company Stockholder Written Consent.
(a) Promptly after the execution and delivery of this Agreement, and in any event no later than ten (10) Business Days thereafter, the Company shall obtain the approval by written consent from
Company stockholders sufficient for the Required Company Stockholder Vote in lieu of a meeting pursuant to Section 228 of the DGCL, for purposes of (i) adopting and approving this Agreement and the Contemplated Transactions, (ii) acknowledging that
the approval given thereby is irrevocable and that such stockholder is aware of its rights to demand appraisal for its shares pursuant to Section 262 of the DGCL, and that such stockholder has received and read a copy of Section 262 of the DGCL and
(iii) acknowledging that by its approval of the Merger it is not entitled to appraisal rights with respect to its shares in connection with the Merger and thereby waives any rights to receive payment of the fair value of its capital stock under the
DGCL (the “Company Stockholder Written Consents”). Under no circumstances shall the Company assert that any other approval or consent is necessary by its stockholders to approve this Agreement and the
Contemplated Transactions.
61
(b) Reasonably promptly following receipt of the Required Company Stockholder Vote, and in any event no later than ten (10) days thereafter, the Company shall prepare and mail a notice (the “Stockholder Notice”) to every stockholder of the Company that did not execute the Company Stockholder Written Consent. The Stockholder Notice shall (i) be a statement to the effect that the Company Board
determined that the Merger is advisable in accordance with Section 251(b) of the DGCL and in the best interests of the stockholders of the Company and approved and adopted this Agreement, the Merger and the other Contemplated Transactions, (ii)
provide the stockholders of the Company to whom it is sent with notice of the actions taken in the Company Stockholder Written Consent, including the adoption and approval of this Agreement, the Merger and the other Contemplated Transactions in
accordance with Section 228(e) of the DGCL and the certificate of incorporation and bylaws of the Company and (iii) include a description of the appraisal rights of the Company’s stockholders available under the DGCL, along with such other
information as is required thereunder and pursuant to applicable Law. All materials (including any amendments thereto) submitted to the stockholders of the Company in accordance with this Section 6.2(b) shall be subject to Parent’s
advance review and reasonable approval.
(c) The Company agrees that: (i) the Company Board shall recommend that the Company’s stockholders vote to adopt and approve this Agreement and the Contemplated Transactions and shall use
commercially reasonable efforts to solicit such approval within the time set forth in Section 6.2(a) (the recommendation of the Company Board that the Company’s stockholders vote to adopt and approve this Agreement being referred to
as the “Company Board Recommendation”), and (ii) except to the extent the fiduciary duties of the Company Board otherwise require prior to the time the Required Company Stockholder Vote is obtained, the
Company Board Recommendation shall not be withdrawn or modified (and the Company Board shall not publicly propose to withdraw or modify the Company Board Recommendation) in a manner adverse to Parent, and no resolution by the Company Board or any
committee thereof to withdraw or modify the Company Board Recommendation in a manner adverse to Parent or to adopt, approve or recommend (or publicly propose to adopt, approve or recommend) any Acquisition Proposal shall be adopted or proposed.
6.3 Parent Stockholder Meeting.
(a) As promptly as practicable following the Closing Date, Parent shall take all action necessary under applicable Law to call, give notice of and hold a meeting of the holders of Parent Common
Stock for the purpose of seeking the affirmative vote of a majority of the shares of Parent Common Stock present or represented and entitled to vote at a meeting of stockholders of Parent to approve, for purposes of the Nasdaq Stock Market Rules,
the issuance of shares of Parent Common Stock to the stockholders of the Company upon conversion of any and all Designated Holder Preferred Stock Payment Shares and the Non-Designated Holder Preferred Stock Payment Shares in accordance with the
terms of the Certificates of Designation (the “Preferred Stock Conversion Proposal,” and such meeting, the “Parent Stockholder Meeting”). Parent agrees to use
reasonable best efforts to call and hold the Parent Stockholders’ Meeting as soon as practicable after the date hereof. Parent shall take reasonable measures to ensure that all proxies solicited in connection with the Parent Stockholder Meeting are
solicited in compliance with all applicable Law. Notwithstanding anything to the contrary contained herein, if on the date of the Parent Stockholder Meeting, or a date preceding the date on which the Parent Stockholder Meeting is scheduled, Parent
reasonably believes that (i) it will not receive proxies sufficient to obtain approval of the Preferred Stock Conversion Proposal, whether or not a quorum would be present, (ii) it will not have sufficient shares of Parent Common Stock represented
(whether in person or by proxy) to constitute a quorum necessary to conduct the business of the Parent Stockholder Meeting or (iii) in good faith after consultation with outside counsel, that the failure to postpone or adjourn the Parent
Stockholder Meeting would reasonably be expected to be inconsistent with its fiduciary obligations under applicable Law, Parent may postpone or adjourn, or make one or more successive postponements or adjournments of, the Parent Stockholder
Meeting; provided, however, that Parent shall postpone or adjourn the meeting only in increments of ten (10) Business Days and in no event more than thirty (30) Business Days in the aggregate without the prior written consent of the holders of a
majority-in-interest of the Designated Holder Preferred Stock Payment Shares and Non-Designated Holder Preferred Stock Payment Shares. If the Parent Stockholders’ Meeting is not so adjourned, and/or if the approval of the Preferred Stock Conversion
Proposal is not then obtained, Parent shall use its reasonable best efforts to obtain such approvals as soon as practicable thereafter, and in any event to obtain such approvals at the next occurring annual meeting of the stockholders of Parent or,
if such annual meeting is not held within six months after the Parent Stockholders’ Meeting, a special meeting of the stockholders of Parent to be held within six months after the Parent Stockholders’ Meeting. Parent shall hold an annual meeting or
special meeting of its stockholders, at which a vote of the stockholders of Parent to approve the Preferred Stock Conversion Proposal will be solicited and taken, at least once every six months until Parent obtains approval of the Preferred Stock
Conversion Proposal.
62
(b) Parent agrees that (i) the Parent Board shall recommend that the holders of Parent Common Stock vote to approve the Preferred Stock Conversion Proposal and shall use its reasonable best
efforts to solicit and obtain such approval within the time frames set forth in Section 6.3(a) above, and (ii) except to the extent the fiduciary duties of the Parent Board otherwise require (but in no event shall Parent be
relieved of the requirement to call and a the Parent Stockholder Meeting as required by Section 6.4(a)): (A) the Proxy Statement shall include a statement to the effect that the Parent Board recommends that Parent’s stockholders vote to approve the
Preferred Stock Conversion Proposal (the recommendation of the Parent Board being referred to as the “Parent Board Recommendation”), and (B) such Parent Board Recommendation shall not be modified or
withdrawn. Nothing herein shall prohibit the Parent Board from making any disclosure that outside counsel advises is required under applicable Law with respect to any material event or circumstance that was not known to the Parent Board on the date
of this Agreement (or, if known, the consequences of which were not known to the Parent Board as of the date of this Agreement).
6.4 Efforts; Regulatory Approvals.
(a) The Parties shall use reasonable best efforts to consummate the Contemplated Transactions. Without limiting the generality of the foregoing, each Party: (i) shall make all filings and other
submissions (if any) and give all notices (if any) required to be made and given by such Party in connection with the Contemplated Transactions, (ii) shall use commercially reasonable efforts to obtain each Consent (if any) reasonably required to
be obtained (pursuant to any applicable Law or Contract, or otherwise) by such Party in connection with the Contemplated Transactions or for such Contract to remain in full force and effect, (iii) shall use commercially reasonable efforts to lift
any injunction prohibiting, or any other legal bar to, the Contemplated Transactions and (iv) shall use commercially reasonable efforts to satisfy the conditions precedent to the consummation of this Agreement.
(b) Notwithstanding the generality of the foregoing, each Party shall use commercially reasonable efforts to file or otherwise submit, as soon as practicable after the date of this Agreement, all
applications, notices, reports and other documents reasonably required to be filed by such Party with or otherwise submitted by such Party to any Governmental Authority with respect to the Contemplated Transactions, and to submit promptly any
additional information requested by any such Governmental Authority. Without limiting the generality of the foregoing, the Parties shall prepare and file, if required, (a) the notification and report forms required to be filed under the HSR Act,
and (b) any notification or other document required to be filed in connection with the Merger under any applicable foreign Law relating to antitrust or competition matters, no later than ten (10) Business Days after the later of (x) with respect to
the filing required under the HSR Act, the date hereof and (y) with respect to any other filing, date the Company and Parent receive notification (in writing or otherwise) from the Federal Trade Commission, the Department of Justice, any state
attorney general, foreign antitrust or competition authority or other Governmental Authority that a filing is required in connection with antitrust or competition matters. Each Party will promptly furnish to the other (i) such necessary information
and reasonable assistance as the other may request in connection with its preparation of any required submissions with respect to this Agreement required under the HSR Act or any other applicable Law and (ii) copies of all written communications
(and memoranda setting forth the substance of all oral communications) between each of them, any of their Affiliates or any of its or their representatives, on the one hand, and any Governmental Authority, on the other hand, with respect to this
Agreement or the transactions contemplated hereby to the extent necessary to make all submissions as are required under the HSR Act or any other applicable Law. Without limiting the generality of the foregoing, each such Party will promptly notify
the other of the receipt and content of any inquiries or requests for additional information made by any Governmental Authority in connection with the HSR Act and will use its reasonable best efforts (A) to promptly comply with any such inquiry or
reasonable request for additional information, documents or other materials, and (B) to the extent reasonably practicable, consult and permit the other to review in advance, and cooperate with the other Party and consider in good faith the views of
the other Party in connection with, any proposed material or substantive oral or written analysis, appearance, presentation, memorandum, brief, argument, opinion, proposal or other communication to be made or submitted in connection with any such
inquiry or request.
63
(c) Without limiting the generality of the foregoing, Parent shall give the Company prompt written notice (email being sufficient) of any litigation against Parent and/or its directors relating to
this Agreement or the Contemplated Transactions (“Transaction Litigation”) (including by providing copies of all pleadings with respect thereto) and keep the Company reasonably informed with respect to the
status thereof. Parent will (i) give the Company the opportunity to participate in, but not control, the defense, settlement or prosecution of any Transaction Litigation (to the extent that the attorney-client privilege is not undermined or
otherwise adversely affected; provided that Parent and the Company will use commercially reasonable efforts to find alternative solutions to not undermine or adversely effect the privilege such as entering into common interest agreements,
joint defense agreements or similar agreements), (ii) consult with the Company with respect to the defense, settlement and prosecution of any Transaction Litigation and (iii) consider in good faith the Company’s advice with respect to such
Transaction Litigation. Parent will obtain the prior written consent of the Company (such consent not to be unreasonably withheld, conditioned or delayed) prior to settling or satisfying any such claim.
6.5 Company RSUs; Company Options.
(a) Company RSUs. At the Effective Time, each unvested restricted stock unit award of the Company (each such award, a “Company RSU”) set forth on Schedule
6.5(a) of the Company Disclosure Letter, that is outstanding immediately prior to the Effective Time, shall vest and be settled for Cullgen Common Stock and shall have the right to receive the Merger Consideration pursuant to Section
2.5(a)(ii).
(b) Company Options. At the Effective Time, Parent shall assume each Company Stock Plan and each In-the-Money Company Option, whether vested or unvested, that is outstanding immediately
prior to the Effective Time shall, at the Effective Time, cease to represent a right to acquire shares of Company Common Stock and shall be converted, at the Effective Time, into an option to purchase shares of Parent Common Stock (an “Assumed Option”), on the same terms and conditions (including any vesting provisions and any provisions providing for accelerated vesting upon certain events) as were applicable under such Company Option as of
immediately prior to the Effective Time, except for administrative or ministerial changes as determined by the Company Board (or, following the Effective Time, the Parent Board or compensation committee). At the Effective Time, each Company Option
that is not an In-the-Money Company Option shall be automatically cancelled and extinguished, with no consideration payable in connection with such cancellation and no further rights to the holder thereof. The number of shares of Parent Common
Stock subject to each such Assumed Option shall be equal to (i) the number of shares of Company Common Stock subject to the respective Company Option immediately prior to the Effective Time multiplied by (ii) the Exchange Ratio, rounded down, if
necessary, to the nearest whole share of Parent Common Stock, and such Assumed Option shall have an exercise price per share (rounded up to the nearest whole cent) equal to (A) the exercise price per share of the Company Common Stock otherwise
purchasable pursuant to the respective Company Option immediately prior to the Effective Time divided by (B) the Exchange Ratio; provided, that in the case of any Company Option to which Section 421 of the Code applies as of immediately
prior to the Effective Time (taking into account the effect of any accelerated vesting thereof, if applicable) by reason of its qualification under Section 422 of the Code, the exercise price, the number of shares of Parent Common Stock subject to
such option and the terms and conditions of exercise of such option shall be determined in a manner consistent with the requirements of Section 424(a) of the Code; provided further, that in the case of any Assumed Option to which Section
409A of the Code applies as of the Effective Time, the exercise price, the number of shares of Parent Common Stock subject to such option and the terms and conditions of exercise of such option shall be determined in a manner consistent with the
requirements of Section 409A of the Code in order to avoid the imposition of any additional taxes thereunder. The Company Board shall, prior to the Effective Time, take all actions necessary to effect the foregoing.
64
(c) Parent shall file with the SEC, promptly after the Effective Time (and in any event, not later than thirty (30) days thereafter), a registration statement on Form S-8 (or any successor form),
if available for use by Parent, relating to the shares of Parent Common Stock issuable with respect to any Assumed RSUs or Assumed Options, as applicable.
6.6 Employee Benefits.
(a) Parent shall comply with the terms of any employment, severance, retention, change of control, or similar agreement specified on Section 4.17(d) or contemplated by Section
5.1 of the Parent Disclosure Letter, subject to the provisions of such agreements. From and after the Effective Time, with respect to each health or welfare benefit plan maintained by Parent, the Company, the Surviving Entity, or any of
their Affiliates for the benefit of each Continuing Employee Parent shall use commercially reasonable efforts to do the following, subject to any required approval of the applicable insurance provider, if any: (i) cause to be waived any eligibility
waiting periods, any evidence of insurability requirements and the application of any preexisting condition limitations under such plan (except to the extent such waiting periods, requirements or limitations applied to the Continuing Employee under
comparable Company Employee Plans prior to the Closing); (ii) provide that Continuing Employees shall receive credit for purposes of eligibility (including for purposes of any vacation, sick, personal time off plans or programs, to the extent
applicable) and vesting for years of service with the Company and/or any of its Subsidiaries prior to the Closing in the applicable welfare benefit plans and defined contribution plan of Parent (but excluding any defined benefit pension plan),
except to the extent providing such credit would result in any duplication of benefits; and (iii) cause each Continuing Employee to be given credit under such plan for all amounts paid by such Continuing Employee under any similar Company Employee
Plan for the plan year that includes the Closing Date for purposes of applying deductibles, co-payments and out-of-pocket maximums as though such amounts had been paid in accordance with the terms and conditions of the applicable plan maintained by
Parent, the Company or the Surviving Entity, as applicable, for the plan year in which the Closing occurs (to the extent such credit would have been given under comparable Company Employee Plans prior to the Closing).
(b) Notwithstanding the foregoing, none of the Company, the Surviving Entity, Parent, and their respective Affiliates shall be obligated to continue to employ any Continuing Employee for any
specific period of time following the Closing Date, and nothing in this Agreement shall interfere with or restrict in any way the rights of Parent or any of its Affiliates (including, from and after the Closing, the Surviving Entity and its
Subsidiaries), which rights are hereby expressly reserved, to discharge or terminate the services of any employee at any time for any reason whatsoever, with or without cause. In addition, nothing contained in this Agreement shall, or shall be
construed so as to (i) constitute an amendment or modification of any Company Benefit Plan or employee benefit plan, (ii) create any third party rights in any such current or former service provider of the Company or its Affiliates (including any
beneficiary or dependent thereof); or (iii) obligate Parent to adopt or maintain any particular plan or program or other compensatory or benefits arrangement at any time or prevent Parent from modifying or terminating any such plan, program or
other compensatory or benefits arrangement at any time.
65
6.7 Indemnification of Officers and Directors.
(a) From the Effective Time through the sixth anniversary of the date on which the Effective Time occurs, each of Parent and the Surviving Entity shall indemnify and hold harmless each person who
is now, or has been at any time prior to the date hereof, or who becomes prior to the Effective Time, a director or officer of the Company or any of its Subsidiaries (the “D&O Indemnified Parties”),
against all claims, losses, liabilities, damages, judgments, fines and reasonable fees, costs and expenses, including attorneys’ fees and disbursements (collectively, “Costs”), incurred in connection with any
claim, action, suit, proceeding or investigation, whether civil, criminal, administrative or investigative, arising out of or pertaining to the fact that the D&O Indemnified Party is or was a director or officer of the Company or any of its
Subsidiaries, whether asserted or claimed prior to, at or after the Effective Time, in each case, to the fullest extent permitted under the DGCL. Each D&O Indemnified Party will be entitled to advancement of expenses incurred in the defense of
any such claim, action, suit, proceeding or investigation from each of Parent and the Surviving Entity, jointly and severally, upon receipt by Parent or the Surviving Entity from the D&O Indemnified Party of a request therefor; provided
that any such person to whom expenses are advanced provides an undertaking to Parent, to the extent then required by the DGCL, to repay such advances if it is ultimately determined that such person is not entitled to indemnification.
(b) The certificate of formation and limited liability company agreement of the Surviving Entity shall contain, and Parent shall cause the certificate of formation and limited liability company
agreement of the Surviving Entity to so contain, provisions no less favorable with respect to indemnification, advancement of expenses and exculpation of present and former directors and officers as those presently set forth in the certificate of
incorporation and bylaws of Parent.
(c) From and after the Effective Time, the Surviving Entity shall fulfill and honor in all respects the obligations of the Company and its Subsidiaries to their D&O Indemnified Parties as of
immediately prior to the Closing pursuant to any indemnification provisions under the Organizational Documents of the Company or its Subsidiaries, as applicable, and pursuant to any indemnification agreements between the Company and/or its
Subsidiaries and such D&O Indemnified Parties, with respect to claims arising out of matters occurring at or prior to the Effective Time.
(d) Reserved.
(e) From and after the Effective Time, Parent shall pay all expenses, including reasonable attorneys’ fees, that are incurred by the persons referred to in this Section 6.7 in
connection with their enforcement of the rights provided to such persons in this Section 6.7.
(f) The provisions of this Section 6.7 are intended to be in addition to the rights otherwise available to the current and former officers and directors of the Company and its
Subsidiaries by Law, charter, statute, bylaw or agreement, and shall operate for the benefit of, and shall be enforceable by, each of the D&O Indemnified Parties, their heirs and their Representatives.
66
(g) In the event Parent or the Surviving Entity or any of their respective successors or assigns (i) consolidates with or merges into any other Person and shall not be the continuing or surviving
corporation or entity of such consolidation or merger or (ii) transfers all or substantially all of its properties and assets to any Person, then, and in each such case, proper provision shall be made so that the successors and assigns of Parent or
the Surviving Entity, as the case may be, shall succeed to the obligations set forth in this Section 6.7. Parent shall cause the Surviving Entity to perform all of the obligations of the Surviving Entity under this Section 6.7.
6.8 Disclosure. The Parties shall use their commercially reasonable efforts to agree to the text of any initial press release and Parent’s Form 8-K announcing the execution and delivery of
this Agreement. Without limiting any Party’s obligations under the Confidentiality Agreement, no Party shall, and no Party shall permit any of its Subsidiaries or any of its Representatives to, issue any press release or make any public or private
disclosure regarding the Contemplated Transactions unless: (a) the other Party shall have approved such press release or disclosure in writing, such approval not to be unreasonably conditioned, withheld or delayed; or (b) such Party shall have
determined in good faith, upon the advice of outside legal counsel, that such disclosure is required by applicable Law and, to the extent practicable, before such press release or disclosure is issued or made, such Party advises the other Party of,
and consults with the other Party regarding, the text of such press release or disclosure. Notwithstanding anything to the contrary herein, neither Parent or Company shall, and shall not permit any of its Subsidiaries or any of its Representatives
to, make any public or private statement to the press, analysts, investors or potential investors, and may not attend, or communicate with anybody attending, industry conferences, during the Pre-Closing Period, without the prior written consent of
the other Party.
6.9 Listing. Parent shall use its commercially reasonable efforts to (a) maintain its listing on Nasdaq, and (b) to the extent required by the rules and regulations of Nasdaq, prepare and
submit to Nasdaq a notification form for the listing of (I) the Designated Holder Common Stock Payment Shares and the Non-Designated Holder Common Stock Payment Shares, and (II) the Parent Common Stock to be issued upon conversion of the Designated
Holder Preferred Stock Payment Shares and the Non-Designated Holder Preferred Stock Payment Shares, in each case, to be issued in connection with the Contemplated Transactions, and to cause such shares to be approved for listing (subject to
official notice of issuance).
6.10 Tax Matters.
(a) The Parties shall use reasonable best efforts (and each shall cause its Affiliates) to cause the Merger to qualify for the Intended Tax Treatment, No Party shall take any actions, or fail to
take any action, which action or failure to act would reasonably be expected to prevent or impede the Intended Tax Treatment. The Parties shall report the Contemplated Transactions for all applicable Tax purposes in a manner that is consistent with
the Intended Tax Treatment. No Party shall take any position for Tax purposes that is inconsistent with the Intended Tax Treatment, except as otherwise required by a ”determination” as described in Section 1313(a) of the Code. The Parties shall
comply with the recordkeeping and information reporting requirements imposed on them with respect to the Contemplated Transactions and the Intended Tax Treatment including, but not limited to, those set forth in Treasury Regulation Section 1.368-3.
(b) Parent shall promptly notify the Company if, at any time before the Effective Time, Parent becomes aware of any fact or circumstance that would reasonably be expected to prevent, cause a
failure of, or impede the Intended Tax Treatment. The Company shall promptly notify Parent if, at any time before the Effective Time, the Company becomes aware of any fact or circumstance that would reasonably be expected to prevent, cause a
failure of, or impede the Intended Tax Treatment.
(c) Parent and the Company shall reasonably cooperate in the preparation, execution and filing of all Tax Returns, questionnaires, applications or other documents regarding any real property
transfer, sales, use, transfer, value added, stock transfer and stamp taxes, and transfer, recording, registration and other fees and similar Taxes which become payable in connection with the Merger that are required or permitted to be filed on or
before the Effective Time. Each of Parent and the Company shall pay, without deduction from any consideration or other amounts payable or otherwise deliverable pursuant to this Agreement and without reimbursement from the other party, any such
Taxes or fees imposed on it by any Governmental Authority, which becomes payable in connection with the Merger.
67
6.11 Legends. Parent shall be entitled to place appropriate legends on the book entries and/or certificates evidencing any shares of Parent Capital Stock to be received in the Merger by
equityholders of the Company who may be considered “affiliates” of Parent for purposes of Rules 144 and 145 under the Securities Act reflecting the restrictions set forth in Rules 144 and 145 and to issue appropriate stop transfer instructions to
the transfer agent for any such shares of Parent Capital Stock.
6.12 Officers and Directors. Until successors are duly elected or appointed and qualified in accordance with applicable Law, the Parties shall use commercially reasonable efforts and take
all necessary action so that the Persons listed on Section 6.12 of the Parent Disclosure Letter are elected or appointed, as applicable, to the positions of officers or directors of Parent, to serve in such positions effective as of the
Effective Time. If any Person listed on Section 6.12 of the Parent Disclosure Letter is unable or unwilling to serve as officer or director of Parent, the Party appointing such Person (as set forth on Section 6.12 of the Parent
Disclosure Letter) shall designate a successor.
6.13 Termination of Certain Agreements and Rights. The Company shall cause any stockholder agreements, voting agreements, registration rights agreements, co-sale agreements and any other
similar Contracts between the Company and any holders of Company Capital Stock, including any such Contract granting any Person investor rights, rights of first refusal, registration rights or director registration rights, to be terminated
immediately prior to the Effective Time, without any liability being imposed on the part of Parent or the Surviving Entity.
6.14 Section 16 Matters. Prior to the Effective Time, Parent shall take all such steps as may be required to cause any acquisitions of Parent Common Stock and any options to purchase Parent
Common Stock in connection with the Contemplated Transactions, by each individual who is reasonably expected to become subject to the reporting requirements of Section 16(a) of the Exchange Act with respect to Parent, to be exempt under Rule 16b-3
promulgated under the Exchange Act.
6.15 Allocation Information. The Company will prepare and deliver to Parent prior to the Closing a spreadsheet setting forth (as of immediately prior to the Effective Time) (a) each holder
of (i) Company Capital Stock, and (ii) Company Options, (b) such holder’s name and address, (c) with respect to holders of Company Capital Stock, the number or percentage and type of Company Capital Stock held as of the Closing Date for each such
holder and (d) the number of shares of Parent Capital Stock and Assumed Options to be issued to such holder, or to underlie any Assumed Option to be issued to such holder, in each case pursuant to this Agreement in respect of the Company Capital
Stock and Company Options, respectively, held by such holder as of immediately prior to the Effective Time (the “Allocation Certificate”).
6.16 Parent SEC Documents. From the date of this Agreement to the Effective Time, Parent shall use commercially reasonable efforts to timely file with the SEC all registrations, proxy
statements, Certifications, reports, schedules, exhibits, forms and other documents required to be filed by Parent with the SEC under the Exchange Act or the Securities Act (“SEC Documents”). As of its filing
date, or if amended after the date of this Agreement, as of the date of the last such amendment, each SEC Document filed by Parent with SEC (a) shall comply in all material respects with the applicable requirements of the Exchange Act and the
Securities Act, and (b) except for information in such SEC Documents that is “furnished” instead of “filed” under Items 2.02 or 7.01 in the Parent’s Current Reports on Form 8-K, shall not contain any untrue statement of a material fact or omit to
state any material fact required to be stated therein or necessary in order to make the statements made therein, in light of the circumstances under which they were made, not misleading.
68
6.17 Obligations of Merger Sub. Parent will take all action necessary to cause Merger Sub to perform its obligations under this Agreement and to consummate the Merger on the terms and
conditions set forth in this Agreement.
6.18 Operations of the Combined Company. Parent agrees to use its reasonable best efforts to operate in accordance with, and pursuant to, the Agreed Business Plan.
6.19 Reservation of Parent Common Stock; Issuance of Shares of Parent Common Stock. For as long as any Designated Holder Preferred Stock Payment Shares or Non-Designated Holder Preferred
Stock Payment Shares remain outstanding, Parent shall at all times reserve and keep available, free from preemptive rights, out of its authorized but unissued Parent Common Stock or shares of Parent Common Stock held in treasury by Parent, for the
purpose of effecting the conversion of the Designated Holder Preferred Stock Payment Shares and Non-Designated Holder Preferred Stock Payment Shares, the full number of shares of Parent Common Stock then issuable upon the conversion of all
Designated Holder Preferred Stock Payment Shares and Non-Designated Holder Preferred Stock Payment Shares then outstanding. All shares of Parent Common Stock delivered upon conversion of the Designated Holder Preferred Stock Payment Shares and the
Non-Designated Holder Preferred Stock Payment Shares shall be newly issued shares or shares held in treasury by Parent, shall have been duly authorized and validly issued and shall be fully paid and nonassessable, and shall be free from preemptive
rights and free of any Encumbrance.
Section 7. Conditions Precedent to Obligations of Each Party. The obligations of each Party to effect the Merger and otherwise consummate the Contemplated Transactions to be consummated at
the Closing are subject to the satisfaction or, to the extent permitted by applicable law, the written waiver by each of the Parties, at or prior to the Closing, of each of the following conditions:
7.1 Regulatory Approvals. Any applicable waiting periods (or any extensions thereof) under the HSR Act (if applicable) shall have expired or otherwise been terminated.
7.2 Foreign Person Status. Neither the Parent nor any of the Merger Sub is a “foreign person” or a “foreign entity,” as defined in Section 721 of the Defense Production Act of 1950, as
amended, including all implementing regulations thereof.
7.3 No Restraints. No Order preventing the consummation of the Contemplated Transactions shall have been issued by any Governmental Authority of competent jurisdiction and remain in effect
and there shall not be any Law which has the effect of making the consummation of the Contemplated Transactions illegal.
7.4 Lock-Up Agreements. The Lock-Up Agreements shall be in full force and effect.
7.5 Certificate of Designation. Parent shall have filed the Certificate of Designation with the Secretary of State of the State of Delaware.
7.6 Registration Rights Agreement. The Registration Rights Agreement shall have been duly executed by Parent, the Company and certain of the holders
of Company Capital Stock.
69
Section 8. Additional Conditions Precedent to Obligations of Parent and Merger Sub. The obligations of Parent and Merger Sub to effect the Merger and otherwise consummate the transactions
to be consummated at the Closing are subject to the satisfaction or the written waiver by Parent, at or prior to the Closing, of each of the following conditions:
8.1 Accuracy of Representations. The Company Fundamental Representations shall have been true and correct in all material respects as of the date of this Agreement and shall be true and
correct on and as of the Closing Date with the same force and effect as if made on and as of such date (except to the extent such representations and warranties are specifically made as of a particular date, in which case such representations and
warranties shall be true and correct as of such date). The Company Capitalization Representations shall have been true and correct in all respects as of the date of this Agreement and shall be true and correct on and as of the Closing Date with the
same force and effect as if made on and as of such date, except, in each case, (x) for such inaccuracies which are de minimis, individually or in the aggregate, (y) for those representations and warranties which address matters only as of a
particular date (which representations and warranties shall have been true and correct, subject to the qualifications as set forth in the preceding clause (x), as of such particular date). The representations and warranties of the Company contained
in this Agreement (other than the Company Fundamental Representations and the Company Capitalization Representations) shall have been true and correct as of the date of this Agreement and shall be true and correct on and as of the Closing Date with
the same force and effect as if made on the Closing Date except (a) in each case, or in the aggregate, where the failure to be so true and correct would not reasonably be expected to have a Company Material Adverse Effect (without giving effect to
any references therein to any Company Material Adverse Effect or other materiality qualifications) or (b) for those representations and warranties which address matters only as of a particular date (which representations shall have been true and
correct, subject to the qualifications as set forth in the preceding clause (a), as of such particular date) (it being understood that, for purposes of determining the accuracy of such representations and warranties, any update of or modification
to the Company Disclosure Letter made or purported to have been made after the date of this Agreement shall be disregarded).
8.2 Performance of Covenants. The Company shall have performed or complied with in all material respects all agreements and covenants required to be performed or complied with by it under
this Agreement at or prior to the Effective Time.
8.3 Documents. Parent shall have received the following documents, each of which shall be in full force and effect:
(a) a certificate executed by the Chief Executive Officer or Chief Financial Officer of the Company certifying (i) that the conditions set forth in Sections 8.1, 8.2, 8.4
and 8.5 have been duly satisfied and (ii) that the information (other than emails and addresses) set forth in the Allocation Certificate delivered by the company in accordance with Section 6.15 is true and accurate in all
respects as of the Closing Date;
(b) a certificate pursuant to Treasury Regulations Sections 1.1445-2(c) and 1.897-2(h), together with a form of notice to the IRS in accordance with the requirements of Treasury Regulations
Section 1.897-2(h), in each case, in form and substance reasonably acceptable to Parent; and
(c) the Allocation Certificate.
8.4 No Company Material Adverse Effect. Since the date of this Agreement, there shall not have occurred any Company Material Adverse Effect that is continuing.
70
8.5 Company Stockholder Written Consent. The Company Stockholder Written Consent executed by the stockholders of the Company shall be in full force and effect.
Section 9. Additional Conditions Precedent to Obligation of the Company.
The obligations of the Company to effect the Merger and otherwise consummate the transactions to be consummated at the Closing are subject to the satisfaction or the written waiver by the Company,
at or prior to the Closing, of each of the following conditions:
9.1 Accuracy of Representations. The Parent Fundamental Representations shall have been true and correct in all material respects as of the date of this Agreement and shall be true and
correct on and as of the Closing Date with the same force and effect as if made on and as of such date (except to the extent such representations and warranties are specifically made as of a particular date, in which case such representations and
warranties shall be true and correct as of such date). The Parent Capitalization Representations shall have been true and correct in all respects as of the date of this Agreement and shall be true and correct on and as of the Closing Date with the
same force and effect as if made on and as of such date, except, in each case, (x) for such inaccuracies which are de minimis, individually or in the aggregate, (y) for those representations and warranties which address matters only as of a
particular date (which representations and warranties shall have been true and correct, subject to the qualifications as set forth in the preceding clause (x), as of such particular date). The representations and warranties of Parent and Merger Sub
contained in this Agreement (other than the Parent Fundamental Representations and the Parent Capitalization Representations) shall have been true and correct as of the date of this Agreement and shall be true and correct on and as of the Closing
Date with the same force and effect as if made on the Closing Date except (a) in each case, or in the aggregate, where the failure to be so true and correct would not reasonably be expected to have a Parent Material Adverse Effect (without giving
effect to any references therein to any Parent Material Adverse Effect or other materiality qualifications) or (b) for those representations and warranties which address matters only as of a particular date (which representations shall have been
true and correct, subject to the qualifications as set forth in the preceding clause (a), as of such particular date) (it being understood that, for purposes of determining the accuracy of such representations and warranties, any update of or
modification to the Parent Disclosure Letter made or purported to have been made after the date of this Agreement shall be disregarded).
9.2 Performance of Covenants. Parent and Merger Sub shall have performed or complied with in all material respects all of their agreements and covenants required to be performed or complied
with by each of them under this Agreement at or prior to the Effective Time.
9.3 Documents. The Company shall have received a certificate executed by an executive officer of Parent certifying that the conditions set forth in Sections 9.1, 9.2 and
9.4 have been duly satisfied.
9.4 No Parent Material Adverse Effect. Since the date of this Agreement, there shall not have occurred any Parent Material Adverse Effect that is continuing.
Section 10. Termination.
10.1 Termination. This Agreement may be terminated prior to the Effective Time (whether before or after adoption of this Agreement by the Company’s stockholders, unless otherwise specified
below):
(a) by mutual written consent of Parent and the Company;
71
(b) by either Parent or the Company if the Merger shall not have been consummated by the date that is the six (6) month anniversary of the date hereof (subject to possible extension as provided in
this Section 10.1(b), the “End Date”); provided, however, that the right to terminate this Agreement under this Section 10.1(b) shall not be
available to the Company or Parent if such Party’s (or in the case of Parent, Merger Sub’s) action or failure to act has been a principal cause of the failure of the Merger to occur on or before the End Date and such action or failure to act
constitutes a breach of this Agreement;
(c) by either Parent or the Company if a court of competent jurisdiction or other Governmental Authority shall have issued a final and non-appealable Order having the effect of permanently
restraining, enjoining or otherwise prohibiting the Contemplated Transactions;
(d) by Parent if the Required Company Stockholder Vote shall not have been obtained within ten (10) Business Days following the execution and delivery of this Agreement; provided, however,
that once the Required Company Stockholder Vote has been obtained, Parent may not terminate this Agreement pursuant to this Section 10.1(d);
(e) by the Company, upon a breach of any representation, warranty, covenant or agreement set forth in this Agreement by Parent or Merger Sub or if any representation or warranty of Parent or
Merger Sub shall have become inaccurate, in either case, such that the conditions set forth in Section 9.1 or Section 9.2 would not be satisfied as of the time of such breach or as of the time such representation or
warranty shall have become inaccurate; provided, that the Company is not then in material breach of any representation, warranty, covenant or agreement under this Agreement; provided further, that if such inaccuracy in Parent’s or
Merger Sub’s representations and warranties or breach by Parent or Merger Sub is curable by Parent or Merger Sub, then the Company shall not be permitted to terminate this Agreement pursuant to this Section 10.1(e) as a
result of such particular breach or inaccuracy until the earlier of (i) the expiration of a thirty (30) day period commencing upon delivery of written notice from the Company to Parent or Merger Sub of such breach or inaccuracy and its intention to
terminate pursuant to this Section 10.1(e) and (ii) Parent or Merger Sub (as applicable) ceasing to exercise commercially reasonable efforts to cure such breach following delivery of written notice from the Company to Parent
or Merger Sub of such breach or inaccuracy and its intention to terminate pursuant to this Section 10.1(e) (it being understood that the Company shall not be permitted to terminate this Agreement pursuant to this Section
10.1(e) as a result of such particular breach or inaccuracy if such breach by Parent or Merger Sub is cured prior to such termination becoming effective); or
(f) by Parent, upon a breach of any representation, warranty, covenant or agreement set forth in this Agreement by the Company or if any representation or warranty of the Company shall have
become inaccurate, in either case, such that the conditions set forth in Section 8.1 or Section 8.2 would not be satisfied as of the time of such breach or as of the time such representation or warranty shall have
become inaccurate; provided that Parent is not then in material breach of any representation, warranty, covenant or agreement under this Agreement; provided, further, that if such inaccuracy in the Company’s representations
and warranties or breach by the Company is curable by the Company then Parent shall not be permitted to terminate this Agreement pursuant to this Section 10.1(f) as a result of such particular breach or inaccuracy until the
earlier of (i) the expiration of a thirty (30) day period commencing upon delivery of written notice from Parent to the Company of such breach or inaccuracy and its intention to terminate pursuant to this Section 10.1(f) and
(ii) the Company ceasing to exercise commercially reasonable efforts to cure such breach following delivery of written notice from Parent to the Company of such breach or inaccuracy and its intention to terminate pursuant to this Section 10.1(f)
(it being understood that Parent shall not be permitted to terminate this Agreement pursuant to this Section 10.1(f) as a result of such particular breach or inaccuracy if such breach by the Company is cured prior to
such termination becoming effective).
72
The Party desiring to terminate this Agreement pursuant to this Section 10.1 (other than pursuant to Section 10.1(a)) shall give a notice of such termination
to the other Party specifying the provisions hereof pursuant to which such termination is made and the basis therefor described in reasonable detail.
10.2 Effect of Termination. In the event of the termination of this Agreement as provided in Section 10.1, this Agreement shall be of no further force or effect; provided,
however, that (a) this Section 10.2, Section 10.3 and Section 11 (other than Section 11.8) and the related definitions of the defined terms in such sections shall survive the
termination of this Agreement and shall remain in full force and effect and (b) the termination of this Agreement and the provisions of Section 10.3 shall not relieve any Party of any liability for fraud or for any willful and
material breach of any representation, warranty, covenant, obligation or other provision contained in this Agreement.
10.3 Expenses.
(a) Except as set forth in this Section 10.3 and Section 6.9 all fees and expenses incurred in connection with this Agreement and the Contemplated Transactions
shall be paid by the Party incurring such expenses, whether or not the Merger is consummated. For the avoidance of doubt, each Party shall pay the respective filing fees required under the HSR Act and for any other similar filings required in any
other jurisdiction.
Section 11. Miscellaneous Provisions.
11.1 Non-Survival of Representations and Warranties. The representations and warranties of the Company, Parent and Merger Sub contained in this Agreement or any certificate or instrument
delivered pursuant to this Agreement shall terminate at the Effective Time, and only the covenants that by their terms survive the Effective Time and this Section 11 shall survive the Effective Time.
11.2 Amendment. This Agreement may be amended with the approval of the respective boards of directors of the Company, Merger Sub and Parent at any time (whether before or after the adoption
and approval of this Agreement by the Company’s stockholders); provided, however, that after any such approval of this Agreement by a Party’s stockholders, no amendment shall be made which by Law requires further approval of such
stockholders without the further approval of such stockholders. This Agreement may not be amended except by an instrument in writing signed on behalf of each of the Company, Merger Sub and Parent.
11.3 Waiver.
(a) Any provision hereof may be waived by the waiving Party solely on such Party’s own behalf, without the consent of any other Party. No failure on the part of any Party to exercise any power,
right, privilege or remedy under this Agreement, and no delay on the part of any Party in exercising any power, right, privilege or remedy under this Agreement, shall operate as a waiver of such power, right, privilege or remedy; and no single or
partial exercise of any such power, right, privilege or remedy shall preclude any other or further exercise thereof or of any other power, right, privilege or remedy.
(b) No Party shall be deemed to have waived any claim arising out of this Agreement, or any power, right, privilege or remedy under this Agreement, unless the waiver of such claim, power, right,
privilege or remedy is expressly set forth in a written instrument duly executed and delivered on behalf of such Party and any such waiver shall not be applicable or have any effect except in the specific instance in which it is given.
73
11.4 Entire Agreement; Counterparts; Exchanges by Electronic Transmission or Facsimile. This Agreement and the other schedules, exhibits, certificates, instruments and agreements referred
to in this Agreement constitute the entire agreement and supersede all prior agreements and understandings, both written and oral, among or between any of the Parties with respect to the subject matter hereof and thereof; provided, however,
that the Confidentiality Agreement shall not be superseded and shall remain in full force and effect in accordance with its terms; provided, further, that only Exhibit D is incorporated by reference and made a part hereof
for purposes of Section 251 of the DGCL. This Agreement may be executed in several counterparts, each of which shall be deemed an original and all of which shall constitute one and the same instrument. The exchange of a fully executed Agreement (in
counterparts or otherwise) by all Parties by facsimile or electronic transmission in PDF format shall be sufficient to bind the Parties to the terms and conditions of this Agreement.
11.5 Applicable Law; Jurisdiction. This Agreement shall be governed by, and construed in accordance with, the laws of the State of Delaware, regardless of the laws that might otherwise
govern under applicable principles of conflicts of laws. In any action or proceeding between any of the Parties arising out of or relating to this Agreement or any of the Contemplated Transactions, each of the Parties: (a) irrevocably and
unconditionally consents and submits to the exclusive jurisdiction and venue of the Court of Chancery of the State of Delaware or, to the extent such court does not have subject matter jurisdiction, the Superior Court of the State of Delaware or
the United States District Court for the District of Delaware, (b) agrees that all claims in respect of such action or proceeding shall be heard and determined exclusively in accordance with clause (a) of this Section 11.5, (c)
waives any objection to laying venue in any such action or proceeding in such courts, (d) waives any objection that such courts are an inconvenient forum or do not have jurisdiction over any Party, (e) agrees that service of process upon such Party
in any such action or proceeding shall be effective if notice is given in accordance with Section 11.7 of this Agreement and (f) irrevocably and unconditionally waives the right to trial by jury.
11.6 Assignability. This Agreement shall be binding upon, and shall be enforceable by and inure solely to the benefit of, the Parties and their respective successors and permitted assigns;
provided, however, that neither this Agreement nor any of a Party’s rights or obligations hereunder may be assigned or delegated by such Party without the prior written consent of the other Party, and any attempted assignment or
delegation of this Agreement or any of such rights or obligations by such Party without the other Party’s prior written consent shall be void and of no effect.
11.7 Notices. All notices and other communications hereunder shall be in writing and shall be deemed to have been duly delivered and received hereunder (a) one (1) Business Day after being
sent for next Business Day delivery, fees prepaid, via a reputable international overnight courier service, (b) upon delivery in the case of delivery by hand or (c) on the date delivered in the place of delivery if sent by email or facsimile (with
a written or electronic confirmation of delivery) prior to 6:00 p.m. (New York City time), otherwise on the next succeeding Business Day, in each case to the intended recipient as set forth below:
if to Parent or Merger Sub:
Gyre Therapeutics, Inc.
12770 High Bluff Drive Suite 150
San Diego, California
Attention: David Epstein, Chair of Special Committee
74
with a copy to (which shall not constitute notice):
Gibson, Dunn & Crutcher LLP
One Embarcadero Center, Suite 2600
San Francisco, CA 94111
Attention: Ryan Murr, Branden Berns, Evan Shepherd
if to the Company:
Cullgen Inc.
12730 High Bluff Drive, Suite 250
San Diego, CA 92130
Attention: Thomas Eastling
with a copy to (which shall not constitute notice):
Mintz, Levin, Cohn, Ferris, Glovsky & Popeo, P.C.
3580 Carmel Mountain Road
Suite 300
San Diego, CA 92130
Attention: Scott Stanton, Jason McCaffrey
11.8 Cooperation. Each Party agrees to cooperate fully with the other Party and to execute and deliver such further documents, certificates, agreements and instruments and to take such
other actions as may be reasonably requested by the other Party to evidence or reflect the Contemplated Transactions and to carry out the intent and purposes of this Agreement.
11.9 Severability. Any term or provision of this Agreement that is invalid or unenforceable in any situation in any jurisdiction shall not affect the validity or enforceability of the
remaining terms and provisions of this Agreement or the validity or enforceability of the offending term or provision in any other situation or in any other jurisdiction. If a final judgment of a court of competent jurisdiction declares that any
term or provision of this Agreement is invalid or unenforceable, the Parties agree that the court making such determination shall have the power to limit such term or provision, to delete specific words or phrases or to replace such term or
provision with a term or provision that is valid and enforceable and that comes closest to expressing the intention of the invalid or unenforceable term or provision, and this Agreement shall be valid and enforceable as so modified. In the event
such court does not exercise the power granted to it in the prior sentence, the Parties agree to replace such invalid or unenforceable term or provision with a valid and enforceable term or provision that will achieve, to the extent possible, the
economic, business and other purposes of such invalid or unenforceable term or provision.
11.10 Other Remedies; Specific Performance. Except as otherwise provided herein, any and all remedies herein expressly conferred upon a Party will be deemed cumulative with and not
exclusive of any other remedy conferred hereby, or by law or equity upon such Party, and the exercise by a Party of any one remedy will not preclude the exercise of any other remedy. The Parties agree that irreparable damage for which monetary
damages, even if available, would not be an adequate remedy, would occur in the event that any of the provisions of this Agreement were not performed in accordance with their specific terms (including failing to take such actions as are required of
it hereunder to consummate this Agreement) or were otherwise breached. It is accordingly agreed that the Parties shall be entitled to an injunction or injunctions to prevent breaches of this Agreement and to enforce specifically the terms and
provisions hereof in the Court of Chancery of the State of Delaware or, to the extent such court does not have subject matter jurisdiction, the Superior Court of the State of Delaware or the United States District Court for the District of
Delaware, this being in addition to any other remedy to which they are entitled at law or in equity, and each of the Parties waives any bond, surety or other security that might be required of any other Party with respect thereto. Each of the
Parties further agrees that it will not oppose the granting of an injunction, specific performance or other equitable relief on the basis that any other Party has an adequate remedy at law or that any award of specific performance is not an
appropriate remedy for any reason at law or in equity.
75
11.11 No Third-Party Beneficiaries. Nothing in this Agreement, express or implied, is intended to or shall confer upon any Person (other than the Parties and the D&O Indemnified Parties
to the extent of their respective rights pursuant to Section 6.7) any right, benefit or remedy of any nature whatsoever under or by reason of this Agreement.
[Remainder of page intentionally left blank]
76
In Witness Whereof, the Parties have caused this Agreement to be executed as of the date
first above written.
|
GYRE THERAPEUTICS, INC.
|
||
|
By:
|
||
|
Name:
|
||
|
Title:
|
||
|
HELIX MERGER SUB CORP.
|
||
|
By:
|
||
|
Name:
|
||
|
Title:
|
||
[Signature Page to Agreement and Plan of Merger and Reorganization]
In Witness Whereof, the Parties have caused this Agreement to be
executed as of the date first above written.
|
CULLGEN INC.
|
||
|
By:
|
||
|
Name:
|
||
|
Title:
|
||
[Signature Page to Agreement and Plan of Merger and Reorganization]
Exhibit 3.1
Confidential
GYRE THERAPEUTICS, INC.
CERTIFICATE OF DESIGNATION OF PREFERENCES,
RIGHTS AND LIMITATIONS
OF
SERIES B CONVERTIBLE PREFERRED STOCK
Pursuant to Section 151 of the
General Corporation Law of the State of Delaware
THE UNDERSIGNED DOES HEREBY CERTIFY, on behalf of Gyre Therapeutics, Inc., a Delaware corporation (the “Corporation”), that the following resolution was duly adopted by the Board of Directors of the Corporation (the “Board of Directors”), in accordance with the provisions of Section 151 of the General Corporation Law of the State of Delaware (the “DGCL”), at a meeting duly called and held on February 27, 2026, which resolution provides for the creation of a series of the Corporation’s Preferred Stock, par value $0.001 per share, which is designated as “Series B
Convertible Preferred Stock,” with the preferences, rights and limitations set forth therein relating to dividends, conversion, redemption, dissolution and distribution of assets of the Corporation.
WHEREAS: the Restated Certificate of Incorporation of the Corporation, as amended (the “Certificate of Incorporation”),
provides for a class of its authorized stock known as Preferred Stock, consisting of 5,000,000 shares, $0.001 par value per share (the “Preferred
Stock”), issuable from time to time in one or more series.
RESOLVED: that, pursuant to authority conferred upon the Board of Directors by the Certificate of Incorporation, (i) a new series of Preferred Stock of the Corporation be, and hereby is authorized by the Board of
Directors, (ii) the Board of Directors hereby authorizes the issuance of [•] shares of “Series B Convertible Preferred Stock” pursuant to the terms of the Agreement and Plan of
Merger and Reorganization, dated as of March 2, 2026, by and among the Corporation, Helix Merger Sub Corp., a Delaware corporation and wholly owned subsidiary of the Corporation, and Cullgen, Inc., a Delaware corporation (the “Merger Agreement”), and (iii) the Board of Directors hereby fixes the designations, powers, preferences and relative, participating, optional
or other special rights, and the qualifications, limitations or restrictions thereof, of such shares of Preferred Stock, in addition to any provisions set forth in the Certificate of Incorporation that are applicable to the Preferred Stock of all
classes and series, as follows:
TERMS OF SERIES B CONVERTIBLE PREFERRED STOCK
1. Definitions. For the purposes hereof, the following terms shall have the following meanings:
“Affiliate” means any Person that,
directly or indirectly through one or more intermediaries, controls or is controlled by or is under common control with a Person, as such terms are used in and construed under Rule 405 of the Securities Act of 1933, as amended.
“Business Day” means any day except any
Saturday, any Sunday, any day which is a federal legal holiday in the United States or any day on which banking institutions in the State of New York are authorized or required by law or other governmental action to close.
“Buy-In” shall have the meaning set
forth in Section 6.5.4.
“Closing Sale Price” means, for any
security as of any date, the last closing trade price for such security immediately prior to 4:00 p.m., New York City time, on the principal Trading Market where such security is listed or traded, as reported by Bloomberg, L.P. (or an equivalent,
reliable reporting service), or if the foregoing do not apply, the last trade price of such security in the over-the-counter market on the electronic bulletin board for such security as reported by Bloomberg, L.P., or, if no last trade price is
reported for such security by Bloomberg, L.P., the average of the bid prices of any market makers for such security as reported on the OTC Pink Market by OTC Markets Group, Inc. If the Closing Sale Price cannot be calculated for a security on a
particular date on any of the foregoing bases, the Closing Sale Price of such security on such date shall be the fair market value as determined in good faith by the Board of Directors of the Corporation.
“Commission” means the United States
Securities and Exchange Commission.
“Common Stock” means the Corporation’s
common stock, par value $0.001 per share, and stock of any other class of securities into which such securities may hereafter be reclassified or changed.
“Conversion Shares” means,
collectively, the shares of Common Stock issuable upon conversion of the shares of Series B Preferred Stock in accordance with the terms hereof.
“Effective Time” means the time at which the merger pursuant to
the terms of the Merger Agreement becomes effective.
“Exchange Act” means the Securities
Exchange Act of 1934, as amended, and the rules and regulations promulgated thereunder.
“Holder” means a holder of shares of
Series B Preferred Stock.
“Person” means an individual or
corporation, partnership, trust, incorporated or unincorporated association, joint venture, limited liability company, joint stock company, government (or an agency or subdivision thereof) or other entity of any kind.
“Trading Day” means a day on which the
principal Trading Market is open for business.
“Trading Market” means any of the
following markets or exchanges on which the Common Stock is listed or quoted for trading on the date in question: the NYSE American, the Nasdaq Capital Market, the Nasdaq Global Market, the Nasdaq Global Select Market, or the New York Stock Exchange
(or any successors to any of the foregoing).
2. Designation, Amount and Par Value. The series of Preferred Stock shall be designated as the Corporation’s Series B Convertible Preferred Stock (the “Series B Preferred Stock”) and the number of shares so designated shall be [•]. Each share of Series B Preferred Stock shall have
a par value of $0.001 per share.
3. Dividends. Holders shall be entitled to receive, and the Corporation shall pay, dividends on shares of the Series B Preferred Stock (on an as-if-converted-to-Common-Stock basis, without regard to the Beneficial Ownership
Limitation (as defined below)) equal to and in the same form, and in the same manner, as dividends (other than dividends on shares of the Common Stock payable in the form of Common Stock) actually paid on shares of the Common Stock when, as and if
such dividends (other than dividends payable in the form of Common Stock) are paid on shares of the Common Stock. Other than as set forth in the previous sentence, no other dividends shall be paid on shares of Series B Preferred Stock, and the
Corporation shall pay no dividends (other than dividends payable in the form of Common Stock) on shares of the Common Stock unless it simultaneously complies with the previous sentence.
2
4. Voting Rights.
4.1 On all matters submitted to a vote of the
Corporation’s stockholders, the holders of shares of Series B Preferred Stock shall vote together with the holders of Common Stock as a single class and, in connection therewith, each holder of outstanding shares of Series B Preferred Stock shall
be entitled to cast one vote for each share of Series B Preferred Stock held by such holder as of the record date for determining stockholders entitled to vote on such matter. In addition to any other vote required herein or by the DGCL, as long
as any shares of Series B Preferred Stock are outstanding, the Corporation shall not, without the affirmative vote of the holders of a majority of the then outstanding shares of the Series B Preferred Stock: (i) alter or change adversely the
powers, preferences or rights given to the Series B Preferred Stock or alter or amend this Certificate of Designation, amend or repeal any provision of, or add any provision to, the Certificate of Incorporation or Bylaws of the Corporation (as
amended, restated or amended and restated from time to time), or file any articles of amendment, certificate of designations, preferences, limitations and relative rights of any series of Preferred Stock, if such action would adversely alter or
change the preferences, rights, privileges or powers of, or restrictions provided for the benefit of the Series B Preferred Stock, regardless of whether any of the foregoing actions shall be by means of amendment to the Certificate of Incorporation
or by merger, consolidation, recapitalization, reclassification, conversion or otherwise, (ii) issue further shares of Series B Preferred Stock or increase or decrease (other than by conversion) the number of authorized shares of Series B Preferred
Stock, (iii) at any time prior to the approval Stockholder Approval, (A) consummate (I) any Fundamental Transaction (as defined below) or (II) any merger or consolidation of the Corporation with or into another entity or any stock sale to, or other
business combination in which the stockholders of the Corporation immediately before such transaction do not hold at least a majority of the capital stock of the Corporation immediately after such transaction, (B) increase the authorized number of
directors constituting the Board of Directors of the Corporation or change the number of votes entitled to be cast by any director or directors on any matter, or (C) enter into any agreement with respect to any of the foregoing. Holders of shares
of Common Stock acquired upon the conversion of shares of Series B Preferred Stock shall be entitled to the same voting rights as each other holder of Common Stock, except that such holders may not vote such shares upon the proposal for Stockholder
Approval in accordance with Rule 5635 of the listing rules of The Nasdaq Stock Market LLC (“Nasdaq”).
4.2 Any vote required or permitted under Section 4.1 may be taken at a meeting of the Holders or the Board of Directors, or through the execution of an action by written consent in lieu of such meeting, provided that
the consent is executed by Holders representing a majority of the outstanding shares of Series B Preferred Stock.
5. Rank; Liquidation.
5.1 The Series B Preferred Stock shall rank on parity
with the Common Stock as to distributions of assets upon liquidation, dissolution or winding up of the Corporation, whether voluntarily or involuntarily.
5.2 Upon any liquidation, dissolution or winding-up of
the Corporation, whether voluntary or involuntary (a “Liquidation”), each Holder shall be entitled to receive out of the assets, whether
capital or surplus, of the Corporation the same amount that a holder of Common Stock would receive if the Series B Preferred Stock were fully converted (disregarding for such purpose any Beneficial Ownership Limitations) to Common Stock which
amounts shall be paid pari passu with all holders of Common Stock, plus an additional amount equal to any dividends declared on but unpaid to such shares.
If, upon any such Liquidation, the assets of the Corporation shall be insufficient to pay the Holders of shares of the Series B Preferred Stock the amount required under the preceding sentence, then all remaining assets of the Corporation shall be
distributed ratably to the Holders and the holders of Common Stock in accordance with the respective amounts that would be payable on all such securities if all amounts payable thereon were paid in full. For the avoidance of any doubt, a
Fundamental Transaction shall not be deemed a Liquidation unless the Corporation expressly declares that such Fundamental Transaction shall be treated as if it were a Liquidation.
3
6. Conversion.
6.1 Prohibition on Conversion. Notwithstanding anything herein to the contrary, the Corporation shall not effect any conversion of any share of Series B Preferred Stock, and a Holder shall not have the right to convert any
portion of the Series B Preferred Stock pursuant to Section 6.2 or otherwise, unless such Holder has, prior to any such conversion of Series B Preferred Stock,
executed and delivered to the Corporation a lock up agreement, substantially in the form set forth as an exhibit to the Merger Agreement. Any purported conversion in violation of this Section 6.1 shall be null and void and of no force or effect
whatsoever.
6.2 Conversion at Option of Holder. Subject to Section 6.4 and Section 6.5.3,
each share of Series B Preferred Stock then outstanding shall be convertible, at any time and from time to time following 5:00 p.m. Eastern time on the third Business Day after the date that the Corporation’s stockholders approve the conversion of
the Series B Preferred Stock into shares of Common Stock in accordance with Nasdaq listing rules, as set forth in Section 4.1 of the Merger Agreement (the “Stockholder Approval”), at the option of the Holder thereof, into a number of shares of Common Stock equal to the Conversion Ratio, subject to the Beneficial Ownership Limitation (as defined below) (each, an “Optional Conversion”). Holders shall effect conversions by providing the Corporation with the form of conversion notice attached hereto as Annex A (a “Notice of Conversion”), duly
completed and executed. Provided the Corporation’s transfer agent is participating in the Depository Trust Company (“DTC”) Fast Automated
Securities Transfer program, the Notice of Conversion may specify, at the Holder’s election, whether the applicable Conversion Shares shall be credited to the account of the Holder’s prime broker with DTC through its Deposit Withdrawal Agent
Commission system (a “DWAC Delivery”). The date on which an Optional Conversion shall be deemed effective (the “Conversion Date”) shall be the Trading Day that the Notice of Conversion, completed and executed, is sent via email to, and received during regular business
hours by, the Corporation; provided, that the original certificate(s) (if any) representing such shares of Series B Preferred Stock being converted, duly endorsed, and the accompanying Notice of Conversion, are received by the Corporation within
two (2) Trading Days thereafter. In all other cases, the Conversion Date shall be defined as the Trading Day on which the original certificate(s) (if any) representing such shares of Series B Preferred Stock being converted, duly endorsed, and the
accompanying Notice of Conversion, are received by the Corporation. The calculations set forth in the Notice of Conversion shall control in the absence of manifest or mathematical error.
6.3 Conversion Ratio. The “Conversion Ratio” for each share of Series B Preferred Stock shall be five (5) shares
of Common Stock issuable upon the conversion (the “Conversion”) of each share of Series B Preferred Stock (corresponding to a ratio of
5:1), subject to adjustment as provided herein.
4
6.4 Beneficial Ownership Limitation. Notwithstanding anything herein to the contrary, and except as set forth herein, the Corporation shall not effect
any conversion of any share of Series B Preferred Stock, and a Holder shall not have the right to convert any portion of the Series B Preferred Stock pursuant to Section 6.2,
to the extent that, after giving effect to such attempted conversion set forth on an applicable Notice of Conversion (as defined in the Certificate of Designation) with respect to the Series B Preferred Stock, such Holder (or any of such Holder’s
Affiliates or any other Person who would be a beneficial owner of Common Stock beneficially owned by the Holder for purposes of Section 13(d) or Section 16 of the Exchange Act and the applicable rules and regulations of the Commission, including
any “group” of which the Holder is a member (the foregoing, “Attribution Parties”)) would beneficially own a number of shares of Common
Stock in excess of the Beneficial Ownership Limitation. For purposes of the foregoing sentence, the aggregate number of shares of Common Stock beneficially owned by such Holder and its Attribution Parties shall include the number of shares of
Common Stock issuable upon conversion of the Series B Preferred Stock subject to the Notice of Conversion, with respect to which such determination is being made, but shall exclude the number of shares of Common Stock which are issuable upon (A)
conversion of the remaining, unconverted Series B Preferred Stock beneficially owned by such Holder or any of its Attribution Parties, and (B) exercise or conversion of the unexercised or unconverted portion of any other securities of the
Corporation (including any warrants) beneficially owned by such Holder or any of its Attribution Parties that are subject to and would exceed a limitation on conversion or exercise similar to the limitation contained herein. Except as set forth in
the preceding sentence, for purposes of this Section 6.4, beneficial ownership shall be calculated in accordance with Section 13(d) of the Exchange Act and the applicable
rules and regulations of the Commission, and the terms “beneficial ownership” and “beneficially own” have the meanings ascribed to such terms therein. In addition, for purposes hereof, “group” has the meaning set forth in Section 13(d) of the
Exchange Act and the applicable rules and regulations of the Commission. For purposes of this Section 6.4, in determining the number of outstanding shares of Common
Stock, a Holder may rely on the number of outstanding shares of Common Stock as stated in the most recent of the following: (A) the Corporation’s most recent periodic or annual filing with the Commission, as the case may be, (B) a more recent
public announcement by the Corporation that is filed with the Commission, or (C) a more recent notice by the Corporation or the Corporation’s transfer agent to the Holder setting forth the number of shares of Common Stock then outstanding. Upon the
written request of a Holder (which may be by email), the Corporation shall, within two (2) Trading Days thereof, confirm in writing to such Holder (which may be via email) the number of shares of Common Stock then outstanding. In any case, the
number of outstanding shares of Common Stock shall be determined after giving effect to any actual conversion or exercise of securities of the Corporation, including shares of Series B Preferred Stock, by such Holder or its Attribution Parties
since the date as of which such number of outstanding shares of Common Stock was last publicly reported or confirmed to the Holder. The “Beneficial
Ownership Limitation” shall initially be set at the discretion of each Holder to a percentage designated by such Holder on its signature page to the Purchase Agreement or otherwise between 0% and 19.99% of the number of shares of the
Common Stock outstanding or deemed to be outstanding as of the applicable measurement date, and such percentage shall be set at 19.99% for any Holder that does not make such designation in the Purchase Agreement or otherwise; provided,
however, that any Holder that holds in excess of 19.99% of the number of shares of Common Stock outstanding immediately prior to the Effective Time shall not be subject to the ownership limitations set forth in this Section 6.4. The Corporation shall be entitled to rely on representations made to it by the Holder in any Notice of Conversion regarding its Beneficial Ownership Limitation. Notwithstanding the
foregoing, by written notice to the Corporation (email being sufficient), (i) the Holder may reset the Beneficial Ownership Limitation percentage to a higher percentage, not to exceed 19.99%, which increase will not be effective until the
sixty-first (61st) day after such written notice is delivered to the Corporation, and (ii) the Holder may reset the Beneficial Ownership Limitation percentage to a lower percentage provided that such decrease shall not become effective until the
later of (x) 5:00 p.m. Eastern time on the third Business Day after the date of the Stockholder Approval and (y) if Stockholder Approval is not obtained within 12 months after the initial issuance of the Series B Preferred Stock, the date that is
three Business Days after the date that is 12 months after the initial issuance of the Series B Preferred Stock. Upon such a change by a Holder of the Beneficial Ownership Limitation, not to exceed 19.99%, the Beneficial Ownership Limitation may
not be further amended by such Holder without first providing the minimum notice required by this Section 6.4. Notwithstanding the foregoing, at any time following notice
of a Fundamental Transaction, the Holder may waive and/or change the Beneficial Ownership Limitation effective immediately upon written notice to the Corporation and may reinstitute a Beneficial Ownership Limitation at any time thereafter effective
immediately upon written notice to the Corporation. The provisions of this Section 6.4 shall be construed, corrected and implemented in a manner so as to effectuate the
intended Beneficial Ownership Limitation herein contained and the shares of Common Stock underlying the Series B Preferred Stock in excess of the Beneficial Ownership Limitation shall not be deemed to be beneficially owned by the Holder for any
purpose including for purposes of Section 13(d) or Rule 16a-1(a)(1) of the Exchange Act.
6.5 Mechanics of Conversion.
6.5.1 Delivery of Certificate or Electronic Issuance. Upon Conversion not later than two (2) Trading Days after the applicable Conversion Date, or if the Holder requests the issuance of physical certificate(s), two (2)
Trading Days after receipt by the Corporation of the original certificate(s) representing such shares of Series B Preferred Stock being converted, duly endorsed, and the accompanying Notice of Conversion (the “Share Delivery Date”), the Corporation shall either: (a) deliver, or cause to be delivered, to the converting Holder a physical certificate or certificates
representing the number of Conversion Shares being acquired upon the conversion of shares of Series B Preferred Stock, or (b) in the case of a DWAC Delivery (if so requested by the Holder), electronically transfer such Conversion Shares by
crediting the account of the Holder’s prime broker with DTC through its DWAC system. If in the case of any Notice of Conversion such certificate or certificates for the Conversion Shares are not delivered to or as directed by or, in the case of a
DWAC Delivery, such shares are not electronically delivered to or as directed by, the applicable Holder by the Share Delivery Date, the applicable Holder shall be entitled to elect to rescind such Notice of Conversion by written notice to the
Corporation at any time on or before its receipt of such certificate or certificates for Conversion Shares or electronic receipt of such shares, as applicable, in which event the Corporation shall promptly return to such Holder any original Series
B Preferred Stock certificate delivered to the Corporation and such Holder shall promptly return to the Corporation any Common Stock certificates or otherwise direct the return of any shares of Common Stock delivered to the Holder through the DWAC
system, representing the shares of Series B Preferred Stock unsuccessfully tendered for conversion to the Corporation.
5
6.5.2 Obligation Absolute. Subject to Section 6.4 and subject to Holder’s right to rescind a Notice of Conversion pursuant to Section 6.5.1, the Corporation’s obligation to issue and deliver the Conversion Shares upon conversion of Series B Preferred Stock in accordance with the terms hereof are absolute and unconditional, irrespective of any action or
inaction by a Holder to enforce the same, any waiver or consent with respect to any provision hereof, the recovery of any judgment against any Person or any action to enforce the same, or any setoff, counterclaim, recoupment, limitation or
termination, or any breach or alleged breach by such Holder or any other Person of any obligation to the Corporation or any violation or alleged violation of law by such Holder or any other Person, and irrespective of any other circumstance which
might otherwise limit such obligation of the Corporation to such Holder in connection with the issuance of such Conversion Shares. Subject to Section 6.4 and subject to
Holder’s right to rescind a Notice of Conversion pursuant to Section 6.5.1, in
the event a Holder shall elect to convert any or all of its Series B Preferred Stock, the Corporation may not refuse conversion based on any claim that such Holder or anyone associated or affiliated with such Holder has been engaged in any
violation of law, agreement or for any other reason, unless an injunction from a court, on notice to Holder, restraining and/or enjoining conversion of all or part of the Series B Preferred Stock of such Holder shall have been sought and obtained
by the Corporation, and the Corporation posts a surety bond for the benefit of such Holder in the amount of 150% of the value of the Conversion Shares into which would be converted the Series B Preferred Stock which is subject to such injunction,
which bond shall remain in effect until the completion of arbitration/litigation of the underlying dispute and the proceeds of which shall be payable to such Holder to the extent it obtains judgment. In the absence of such injunction, the
Corporation shall, subject to Section 6.4 and subject to Holder’s right to
rescind a Notice of Conversion pursuant to Section 6.5.1, issue Conversion Shares upon a properly noticed conversion.
6.5.3 Cash Settlement. If, at any time after the earlier of Stockholder Approval or 12 months after the initial issuance of the Series B Preferred Stock, the Corporation fails to deliver to a Holder such certificate or
certificates, or electronically deliver (or instruct its transfer agent to electronically deliver) such shares in the case of a DWAC Delivery, pursuant to Section 6.5.1
on or prior to the third (3rd) Trading Day after the Share Delivery Date applicable to such conversion (other than a failure caused by (i) materially incorrect or incomplete information provided by Holder to the Corporation or (ii) the application
of the Beneficial Ownership Limitation after Stockholder Approval (but, prior to the Stockholder Approval, disregarding for such purpose any Beneficial Ownership Limitation), then, unless the Holder has rescinded the applicable Notice of Conversion
pursuant to Section 6.5.1, the Corporation shall, at the request of the Holder, pay an amount equal to the Fair Value (as defined below) of such undelivered
shares, with such payment to be made within two Business Days from the date of request by the Holder, whereupon the Corporation’s obligations to deliver such shares underlying the Notice of Conversion shall be extinguished upon payment in full of
the Fair Value of such undelivered shares; provided, however that such request shall be presumed to have been made by such Holder if Stockholder Approval shall not have been obtained prior to the date on which the Notice of Conversion is delivered
to the Corporation. For purposes of this Section 6.5.3, the “Fair Value” of shares shall be fixed with reference to the last reported Closing Sale Price on the
principal Trading Market on which the Common Stock is listed as of the Trading Day immediately prior to the date on which the Notice of Conversion is delivered to the Corporation. For the avoidance of doubt, the cash settlement provisions set forth
in this Section 6.5.3 shall be available irrespective of the reason for the Corporation’s failure to timely deliver Conversion Shares (other than a failure caused
by (i) materially incorrect or incomplete information provided by Holder to the Corporation or (ii) the application of the Beneficial Ownership Limitation after Stockholder Approval (but, prior to the Stockholder Approval, disregarding for such
purpose any Beneficial Ownership Limitation)), including due to limitations set forth in Section 6.5.6, the lack of obtaining Stockholder Approval, or due to
applicable Trading Market rules.
6
6.5.4 Buy-In on Failure to Timely Deliver Certificates. If the Corporation fails to deliver to a Holder the applicable certificate or certificates or to effect a DWAC Delivery, as applicable, by the Share
Delivery Date pursuant to Section 6.5.1 (other than a failure caused by materially incorrect or incomplete information provided by Holder to the Corporation or the
application of the Beneficial Ownership Limitation), and if after such Share Delivery Date such Holder is required by its brokerage firm to purchase (in an open market transaction or otherwise), or the Holder’s brokerage firm otherwise purchases, shares of Common Stock to deliver in satisfaction of a sale by such Holder of the Conversion Shares which such Holder was entitled to receive upon
the conversion relating to such Share Delivery Date (a “Buy-In”), then the Corporation shall (A) pay in cash to such Holder (in addition to any other remedies available to or elected
by such Holder) the amount by which (x) such Holder’s total purchase price (including any brokerage commissions) for the shares of Common Stock so purchased
exceeds (y) the product of (1) the aggregate number of shares of Common Stock that such Holder was entitled to receive from the conversion at issue multiplied by (2) the actual sale price at which the sell order giving rise to such purchase
obligation was executed (including any brokerage commissions) and (B) at the option of such Holder, either reissue (if surrendered) the shares of Series B Preferred Stock equal to the number of shares of Series B Preferred Stock submitted for
conversion or deliver to such Holder the number of shares of Common Stock that would have been issued if the Corporation had timely complied with its delivery requirements under Section
6.5.1. For example, if a Holder purchases shares of Common Stock having a total purchase price of $11,000 to cover a Buy-In with respect to an attempted conversion of shares of Series B Preferred Stock with respect to which the
actual sale price (including any brokerage commissions) giving rise to such purchase obligation was a total of $10,000 under clause (A) of the immediately preceding sentence, the Corporation shall be required to pay such Holder $1,000. The Holder
shall provide the Corporation written notice, within three (3) Trading Days after the occurrence of a Buy-In, indicating the amounts payable to such Holder in respect of such Buy-In together with applicable confirmations and other evidence
reasonably requested by the Corporation. Nothing herein shall limit a Holder’s right to pursue any other remedies available to it hereunder, at law or in
equity including, without limitation, a decree of specific performance and/or injunctive relief with respect to the Corporation’s failure to timely deliver
certificates representing shares of Common Stock upon conversion of the shares of Series B Preferred Stock as required pursuant to the terms hereof or the cash settlement remedy set forth in Section 6.5.3; provided, however, that the Holder shall not be entitled to both (i) require the reissuance of the shares of Series B Preferred Stock submitted for conversion for which such conversion was not timely
honored and (ii) receive the number of shares of Common Stock that would have been issued if the Corporation had timely complied with its delivery requirements under Section 6.5.1.
6.5.5 Reservation of Shares Issuable Upon Conversion. The Corporation covenants that at all times it will reserve and keep available out of its authorized and unissued shares of Common Stock for the sole purpose of issuance
upon conversion of the Series B Preferred Stock, free from preemptive rights or any other actual contingent purchase rights of Persons other than the Holders of the Series B Preferred Stock, not less than such aggregate number of shares of the
Common Stock as shall be issuable (taking into account the adjustments of Section 7) upon the conversion of all outstanding shares of Series B Preferred Stock. The
Corporation covenants that all shares of Common Stock that shall be so issuable shall, upon issue, be duly authorized, validly issued, fully paid and Partial-assessable.
6.5.6 Fractional Shares. No fractional shares of Common Stock shall be issued upon conversion of the Series B Preferred Stock, no certificates or scrip for any such fractional shares shall be issued and no cash shall be paid
for any such fractional shares. Any fractional shares of Common Stock that a Holder of Series B Preferred Stock would otherwise be entitled to receive shall be aggregated with all fractional shares of Common Stock issuable to such Holder and any
remaining fractional shares shall be rounded up to the nearest whole share. Whether or not fractional shares would be issuable upon such conversion shall be determined on the basis of the total number of shares of Series B Preferred Stock the
Holder seeks to convert into Common Stock and the aggregate number of shares of Common Stock issuable upon such conversion.
7
6.5.7 Transfer Taxes. The issuance of certificates for shares of the Common Stock upon conversion of the Series B Preferred Stock shall be made without charge to any Holder for any documentary stamp or similar taxes that may
be payable in respect of the issue or delivery of such certificates, provided that the Corporation shall not be required to pay any tax that may be payable in respect of any transfer involved in the issuance and delivery of any such certificate
upon conversion in a name other than that of the registered Holder(s) of such shares of Series B Preferred Stock and the Corporation shall not be required to issue or deliver such certificates unless or until the Person or Persons requesting the
issuance thereof shall have paid to the Corporation the amount of such tax or shall have established to the satisfaction of the Corporation that such tax has been paid.
6.6 Status as Stockholder. Upon each Conversion Date, (i) the shares of Series B Preferred Stock being converted shall be deemed converted into shares of Common Stock and (ii) the Holder’s rights as a holder of such
converted shares of Series B Preferred Stock shall cease and terminate, excepting only the right to receive certificates for such shares of Common Stock and to any remedies provided herein or otherwise available at law or in equity to such Holder
because of a failure by the Corporation to comply with the terms of this Certificate of Designation. In all cases, the Holder shall retain all of its rights and remedies for the Corporation’s failure to convert Series B Preferred Stock. In no
event shall the Series B Preferred Stock convert into shares of Common Stock prior to the Stockholder Approval.
7. Certain Adjustments.
7.1 Stock Dividends and Stock Splits. If the Corporation, at any time while this Series B Preferred Stock is outstanding: (A) pays a stock dividend or otherwise makes a distribution or distributions payable in shares of
Common Stock (which, for avoidance of doubt, shall not include any shares of Common Stock issued by the Corporation upon conversion of this Series B Preferred Stock) with respect to the then outstanding shares of Common Stock; (B) subdivides
outstanding shares of Common Stock into a larger number of shares; or (C) combines (including by way of a reverse stock split) outstanding shares of Common Stock into a smaller number of shares, then the Conversion Ratio shall be multiplied by a
fraction of which the numerator shall be the number of shares of Common Stock (excluding any treasury shares of the Corporation) outstanding immediately after such event and of which the denominator shall be the number of shares of Common Stock
outstanding immediately before such event (excluding any treasury shares of the Corporation). Any adjustment made pursuant to this Section 7.1 shall become effective
immediately after the record date for the determination of stockholders entitled to receive such dividend or distribution and shall become effective immediately after the effective date in the case of a subdivision or combination.
7.2 Fundamental Transaction. If, at any time while this Series B Preferred Stock is outstanding, (A) the Corporation effects any merger or consolidation of the Corporation with or into another Person or any stock sale to,
or other business combination (including, without limitation, a reorganization, recapitalization, spin-off, share exchange or scheme of arrangement) with or into another Person (other than such a transaction in which the Corporation is the
surviving or continuing entity and its Common Stock is not exchanged for or converted into other securities, cash or property), (B) the Corporation effects any sale, lease, transfer or exclusive license of all or substantially all of its assets in
one transaction or a series of related transactions, (C) any tender offer or exchange offer (whether by the Corporation or another Person) is completed pursuant to which more than 50% of the Common Stock not held by the Corporation or such Person
is exchanged for or converted into other securities, cash or property, or (D) the Corporation effects any reclassification of the Common Stock or any compulsory share exchange pursuant (other than as a result of a dividend, subdivision or
combination covered by Section 7.1) to which the Common Stock is effectively converted into or exchanged for other securities, cash or property (in any such case, a “Fundamental Transaction”), then, upon any subsequent conversion of this Series B Preferred Stock the Holders shall have the right to receive,
in lieu of the right to receive Conversion Shares, for each Conversion Share that would have been issuable upon such conversion immediately prior to the occurrence of such Fundamental Transaction, the same kind and amount of securities, cash or
property as it would have been entitled to receive upon the occurrence of such Fundamental Transaction if it had been, immediately prior to such Fundamental Transaction, the holder of one share of Common Stock (the “Alternate Consideration”). For purposes of any such subsequent conversion, the determination of the Conversion Ratio shall be appropriately adjusted to apply to such
Alternate Consideration based on the amount of Alternate Consideration issuable in respect of one share of Common Stock in such Fundamental Transaction, and the Corporation shall adjust the Conversion Ratio in a reasonable manner reflecting the
relative value of any different components of the Alternate Consideration. If holders of Common Stock are given any choice as to the securities, cash or property to be received in a Fundamental Transaction, then the Holders shall be given the same
choice as to the Alternate Consideration it receives upon any conversion of this Series B Preferred Stock following such Fundamental Transaction. To the extent necessary to effectuate the foregoing provisions, any successor to the Corporation or
surviving entity in such Fundamental Transaction shall file a new certificate of designations with the same terms and conditions and issue to the Holders new preferred stock consistent with the foregoing provisions and evidencing the Holders’ right
to convert such preferred stock into Alternate Consideration. The terms of any agreement to which the Corporation is a party and pursuant to which a Fundamental Transaction is effected shall include terms requiring any such successor or surviving
entity to comply with the provisions of this Section 7.2 and ensuring that this Series B Preferred Stock (or any such replacement security) will be similarly adjusted
upon any subsequent transaction analogous to a Fundamental Transaction. The Corporation shall cause to be delivered to each Holder, at its last address as it shall appear upon the stock books of the Corporation, written notice of any Fundamental
Transaction at least 20 calendar days prior to the date on which such Fundamental Transaction is expected to become effective or close.
8
7.3 Calculations. All calculations under this Section 7 shall be made to the nearest cent or the nearest 1/100th of a share, as the case may be. For
purposes of this Section 7, the number of shares of Common Stock deemed to be issued and outstanding as of a given date shall be the sum of the number of shares of Common
Stock (excluding any treasury shares of the Corporation) issued and outstanding.
8. Redemption. The shares of Series B Preferred Stock shall not be redeemable; provided, however, that the foregoing shall not limit the ability of the Corporation to purchase or otherwise deal in such shares to the
extent otherwise permitted hereby and by law, nor shall the foregoing limit the Holder’s rights under Section 6.5.3.
9. Transfer. The shares of Series B Preferred Stock, together with the accompanying rights set forth herein, shall not be transferable, assignable, pledged or otherwise encumbered by the holder thereof without the
prior written consent of the Board of Directors of the Corporation, which consent may be withheld in its sole discretion. Any attempted transfer in violation of this provision shall be null and void ab initio.
10. Series B Preferred Stock Register. The Corporation shall maintain at its principal executive offices (or such other office or agency of the Corporation as it may designate by notice to the Holders in accordance with Section 11), a register for the Series B Preferred Stock, in which the Corporation shall record (i) the name, address, and electronic mail address of each holder in whose name
the shares of Series B Preferred Stock have been issued and (ii) the name, address, and electronic mail address of each transferee of any shares of Series B Preferred Stock. The Corporation may deem and treat the registered Holder of shares of
Series B Preferred Stock as the absolute owner thereof for the purpose of any conversion thereof and for all other purposes. The Corporation shall keep the register open and available at all times during business hours for inspection by any holder
of Series B Preferred Stock or his, her or its legal representatives.
11. Notices. Any notice required or permitted by the provisions of this Certificate of Designation to be given to a Holder of shares of Series B Preferred Stock shall be mailed, postage prepaid, to the post office address last
shown on the records of the Corporation, or given by electronic communication in compliance with the provisions of the DGCL, and shall be deemed sent upon such mailing or electronic transmission.
9
12. Book-Entry; Certificates. The Series B Preferred Stock will be issued in book-entry form; provided that, if a Holder requests that such Holder’s shares of Series B Preferred Stock be issued in certificated form, the
Corporation will instead issue a stock certificate to such Holder representing such Holder’s shares of Series B Preferred Stock. To the extent that any shares of Series B Preferred Stock are issued in book-entry form, references herein to
“certificates” shall instead refer to the book-entry notation relating to such shares.
13. Waiver. Any waiver by the Corporation or a Holder of a breach of any provision of this Certificate of Designation shall not operate as or be construed to be a waiver of any other breach of such provision or of any breach of
any other provision of this Certificate of Designation or a waiver by any other Holders. The failure of the Corporation or a Holder to insist upon strict adherence to any term of this Certificate of Designation on one or more occasions shall not be
considered a waiver or deprive that party (or any other Holder) of the right thereafter to insist upon strict adherence to that term or any other term of this Certificate of Designation. Any waiver by the Corporation or a Holder must be in writing.
Notwithstanding any provision in this Certificate of Designation to the contrary, any provision contained herein and any right of the Holders of Series B Preferred Stock granted hereunder may be waived as to all shares of Series B Preferred Stock
(and the Holders thereof) upon the written consent of the Holders of not less than a majority of the shares of Series B Preferred Stock then outstanding, provided, however, that the Beneficial Ownership Limitation applicable to a Holder, and any
provisions contained herein that are related to such Beneficial Ownership Limitation, cannot be modified, waived or terminated without the consent of such Holder, provided further, that any proposed waiver that would, by its terms, have a
disproportionate and materially adverse effect on any Holder shall require the consent of such Holder(s).
14. Severability. Whenever possible, each provision hereof shall be interpreted in a manner as to be effective and valid under applicable law, but if any provision hereof is held to be prohibited by or invalid under applicable
law, then such provision shall be ineffective only to the extent of such prohibition or invalidity, without invalidating or otherwise adversely affecting the remaining provisions hereof.
15. Status of Converted Series B Preferred Stock. If any shares of Series B Preferred Stock shall be converted or redeemed by the Corporation, such shares shall, to the fullest extent permitted by applicable law, be retired
and cancelled upon such acquisition, and shall not be reissued as a share of Series B Preferred Stock. Any share of Series B Preferred Stock so acquired shall, upon its retirement and cancellation, and upon the taking of any action required by
applicable law, resume the status of authorized but unissued shares of preferred stock and shall no longer be designated as Series B Preferred Stock.
[Remainder of Page Intentionally Left Blank]
10
IN WITNESS WHEREOF, Gyre Therapeutics, Inc. has caused this
Certificate of Designation of Preferences, Rights and Limitations of Series B Convertible Preferred Stock to be duly executed by its Chief Executive Officer on [•], 2026.
|
GYRE THERAPEUTICS, INC.
|
|||
| By: | |||
| Name: | |||
| Title: |
|
ANNEX A
NOTICE OF CONVERSION
(TO BE EXECUTED BY THE REGISTERED HOLDER IN ORDER TO CONVERT SHARES OF SERIES B CONVERTIBLE PREFERRED STOCK)
The undersigned Holder hereby irrevocably elects to convert the number of shares of Series B Preferred Stock indicated below, represented in book-entry form, into shares
of common stock, par value $0.001 per share (the “Common Stock”), of Gyre Therapeutics, Inc., a Delaware corporation (the “Corporation”), as of the date written below. If securities are to be issued in the name of a Person other than the undersigned, the undersigned
will pay all transfer taxes payable with respect thereto. Capitalized terms utilized but not defined herein shall have the meaning ascribed to such terms in that certain Certificate of Designation of Preferences, Rights and Limitations of Series B
Convertible Preferred Stock (the “Certificate of Designation”) filed by the Corporation with the Secretary of State of the State of Delaware
on [•], 2026.
As of the date hereof, the number of shares of Common Stock beneficially owned by the undersigned Holder (together with such Holder’s Attribution Parties), including the
number of shares of Common Stock issuable upon conversion of the Series B Preferred Stock subject to this Notice of Conversion, but excluding the number of shares of Common Stock which are issuable upon (A) conversion of the remaining, unconverted
Series B Preferred Stock beneficially owned by such Holder or any of its Attribution Parties, and (B) exercise or conversion of the unexercised or unconverted portion of any other securities of the Corporation (including any warrants) beneficially
owned by such Holder or any of its Attribution Parties that are subject to a limitation on conversion or exercise similar to the limitation contained in Section 6.4 of the
Certificate of Designation, is _____%. For purposes hereof, beneficial ownership shall be calculated in accordance with Section 13(d) of the Exchange Act and the applicable regulations of the Commission. In addition, for purposes hereof, “group” has
the meaning set forth in Section 13(d) of the Exchange Act and the applicable regulations of the Commission.
CONVERSION CALCULATIONS:
|
Date to Effect Conversion:
|
|
|
Number of shares of Series B Preferred Stock owned prior to Conversion:
|
|
|
Number of shares of Series B Preferred Stock to be Converted:
|
|
|
Number of shares of Common Stock to be Issued:
|
|
|
Address for delivery of physical certificates:
|
For DWAC Delivery, please provide the following:
| Broker No.: |
| Account No.: |
|
[HOLDER]
|
|||
| By: | |||
| Name: | |||
| Title: |
|
Exhibit 10.1
Confidential
This Support Agreement (this “Agreement”) is made and entered into as of March 2, 2026, by and among Cullgen Inc., a Delaware corporation (the “Company”), Gyre Therapeutics, Inc., a
Delaware corporation (“Parent”), and the undersigned stockholder (the “Stockholder”) of the Company. Capitalized terms used herein but not otherwise defined shall have the respective meanings ascribed to such terms in the Merger
Agreement (as defined below).
RECITALS
WHEREAS, concurrently with the execution and delivery hereof, Parent, the Company, and Helix Merger Sub Corp., a Delaware corporation and a wholly owned subsidiary of Parent (“Merger Sub”),
have entered into an Agreement and Plan of Merger and Reorganization, dated of even date herewith (as such agreement may be amended or supplemented from time to time pursuant to the terms thereof, the “Merger Agreement”), pursuant to which
Merger Sub will merge with and into the Company (the “Merger”), with the Company surviving the Merger as the surviving corporation and a wholly owned subsidiary of Parent, upon the terms and subject to the conditions set forth in the Merger
Agreement.
WHEREAS, as of the date hereof, the Stockholder is the beneficial owner (as defined in Rule 13d-3 under the Exchange Act) and has sole or shared voting power with respect to such number of shares
of Company Capital Stock, and holds Company Options to acquire the number of Shares, as indicated in Appendix A.
WHEREAS, as an inducement and a condition to the willingness of Parent to enter into the Merger Agreement, each Stockholder has agreed to enter into and perform this Agreement.
NOW, THEREFORE, intending to be legally bound, the parties hereby agree as follows:
1. Certain Definitions. Capitalized terms used but not otherwise defined herein shall have the meanings ascribed thereto in the Merger Agreement. For all
purposes of this Agreement, the following terms shall have the following respective meanings:
(a) “Constructive Sale” means, with respect to any security, a short sale with respect to such security, entering into or acquiring a derivative contract
with respect to such security, entering into or acquiring a futures or forward contract to deliver such security or entering into any other hedging or other derivative transaction that has the effect of either directly or indirectly materially
changing the economic benefits or risks of ownership of such security.
(b) “Shares” means (i) all shares of Company Capital Stock owned, beneficially or of record, by the Stockholder as of the date hereof, (ii) all additional
shares of Company Capital Stock acquired by the Stockholder, beneficially or of record, during the period commencing with the execution and delivery of this Agreement and expiring on the Expiration Date (as defined below) and (iii) any shares of
capital stock or other equity securities of the Company that such Stockholder acquires or with respect to which such Stockholder otherwise acquires sole or shared voting power (including any proxy), whether beneficial or of record, or otherwise
owned by such Stockholder after the execution and delivery of this Agreement, the Third Amended and Restated Voting Agreement of the Company, dated as of April 13, 2023 (the “Voting Agreement”), and expiring on the Expiration Date, whether by
exercise of any Company Options or otherwise, including, without limitation, by gift, succession, in the event of a stock split or as a dividend or distribution of any Shares.
Page 1
(c) “Transfer” or “Transferred” means, with respect to any security, the direct or indirect assignment, sale, transfer, tender, exchange, pledge or
hypothecation, or the grant, creation or suffrage of an Encumbrance, lien, security interest or encumbrance in or upon, or the gift, grant or placement in trust, or the Constructive Sale or other disposition of such security (including transfers
by testamentary or intestate succession, by domestic relations order or other court order, or otherwise by operation of law) or any right, title or interest therein (including any right or power to vote to which the holder thereof may be
entitled, whether such right or power is granted by proxy or otherwise), or the record or beneficial ownership thereof, the offer to make such a sale, transfer, Constructive Sale or other disposition, and each agreement, arrangement or
understanding, whether or not in writing, to effect any of the foregoing.
2. Transfer and Voting Restrictions. The Stockholder covenants to the Company and Parent as follows:
(a) Except as otherwise permitted by Section 2(c), during the period commencing with the execution and delivery of this Agreement and expiring on the Expiration
Date (as defined below), the Stockholder shall not Transfer any of the Stockholder’s Shares, publicly announce its intention to Transfer any of its Shares, or enter into any contract with respect to any Transfer of, its Shares or any interest
therein.
(b) Except as otherwise permitted by this Agreement or otherwise permitted or required or by order of a court of competent jurisdiction or a Governmental
Authority, the Stockholder will not commit any act that would restrict the Stockholder’s legal power, authority and right to vote all of the Shares held by the Stockholder or otherwise prevent or disable the Stockholder from performing any of
his, her or its obligations under this Agreement. Without limiting the generality of the foregoing, except for this Agreement, and as otherwise permitted by this Agreement, the Stockholder shall not enter into any voting agreement with any person
or entity with respect to any of the Stockholder’s Shares, grant any person or entity any proxy (revocable or irrevocable) or power of attorney with respect to any of the Shares, deposit any Shares in a voting trust or otherwise enter into any
agreement or arrangement with any person or entity in each case, which has the effect of limiting or affecting the Stockholder’s legal power, authority or right to execute and deliver the Company Stockholder Written Consents.
(c) Except as otherwise permitted by this Agreement or otherwise permitted or required by order of a court of competent jurisdiction or a Governmental Authority,
the Stockholder will not enter into any Contract, option, commitment or other arrangement or understanding with respect to the direct or indirect Transfer of any right, title or interest (including any right or power to vote to which the holder
thereof may be entitled whether such right or power is granted by proxy or otherwise) to any Shares or take any action that would reasonably be expected to make any representation or warranty of such Stockholder contained herein untrue or
incorrect or have the effect of restricting the Stockholder’s legal power, authority and right to vote all of the Shares or would otherwise prevent or disable such Stockholder from performing any of such Stockholder’s obligations under this
Agreement.
(d) Notwithstanding anything else herein to the contrary, the Stockholder may, at any time, Transfer Shares (i) by will or other testamentary document or by
intestacy, (ii) to such Stockholder’s Affiliates (in each case, directly or indirectly), (iii) to any member of the Stockholder’s immediate family (or, if the Stockholder is a corporation, partnership or other entity, to an immediate family
member of a beneficial owner of the Shares held by the Stockholder), (iv) to any trust or other entity for the direct or indirect benefit of the Stockholder or the immediate family of the Stockholder (or, if the Stockholder is a corporation,
partnership or other entity, for the direct or indirect benefit of an immediate family member of a beneficial owner of the Shares held by the Stockholder) or otherwise for estate tax or estate planning purposes, (v) in the case of a Stockholder
who is not a natural person, by pro rata distributions from the Stockholder to its members, partners, or shareholders pursuant to the Stockholder’s organizational documents, (vi) with respect to such Stockholder’s Company Options (and any Shares
underlying such Company Options) which expire on or prior to the Expiration Date, Transfers of Shares to the Company (or effecting a “net exercise” of a Company Option) as payment for the (a) exercise price of such Stockholder’s Company Options
and (b) taxes applicable to the exercise of such Stockholder’s Company Options, (vii) transfers to another holder of capital stock of the Company that has signed a support agreement that is reasonably acceptable to Parent, and (viii) transfers,
sales or other dispositions as Parent may otherwise agree in writing in its sole discretion; provided, that in the cases of clauses (i)-(viii), (1) such Transferred Shares shall continue to be bound by this Agreement and (2) the
applicable direct transferee (if any) of such Transferred Shares shall have executed and delivered to Parent and the Company a support agreement substantially identical to this Agreement upon consummation of the Transfer if not already a party
thereto, or (ix) to the extent required by applicable Law. Any action taken in violation of Section 2(a) through 2(d) shall be null and void ab initio.
Page 2
(e) Notwithstanding anything to the contrary herein, nothing in this Agreement shall obligate the Stockholder to exercise any option or any other right to acquire
any shares of Company Capital Stock.
3. Agreement to Vote Shares. The Stockholder covenants to the Company and Parent as follows:
(a) Promptly after the execution and delivery of this Agreement, and in any event no later than ten (10) Business Days thereafter, the Stockholder shall deliver in
accordance with Section 228 of the DGCL a written consent (i) adopting and approving this Agreement and the Contemplated Transactions, (ii) acknowledging that the approval given thereby is irrevocable and that such stockholder is aware of its
rights to demand appraisal for its shares pursuant to Section 262 of the DGCL, and that such stockholder has received and read a copy of Section 262 of the DGCL and (iii) acknowledging that by its approval of the Merger it is not entitled to
appraisal rights with respect to its shares in connection with the Merger and thereby waives any rights to receive payment of the fair value of its capital stock under the DGCL.
(b) Until the Expiration Date (as defined below), at any meeting of the stockholders of the Company, however called, and at every adjournment or postponement
thereof, and on every action or approval by written consent of the stockholders of the Company, the Stockholder shall be present (in person or by proxy) and vote (including by submitting the proxy solicited by the Company with respect to such
meeting), or exercise its right to consent with respect to, all Shares held by the Stockholder (A) in favor of the adoption and approval of the Merger Agreement, (B) in favor of approval of the Contemplated Transactions, including any matter that
could reasonably be expected to facilitate the Contemplated Transactions, and (C) against any Acquisition Proposal.
(c) If the Stockholder is the beneficial owner, but not the record holder, of Shares, the Stockholder agrees to take all actions necessary to cause the record
holder and any nominees to be present (in person or by proxy) and vote all the Stockholder’s Shares in accordance with this Section 3.
(d) In the event of a stock split, stock dividend or distribution, or any change in the capital stock of the Company by reason of any split-up, reverse stock
split, recapitalization, combination, reclassification, reincorporation, exchange of shares or the like, the term “Shares” shall be deemed to refer to and include such shares as well as all such stock dividends and distributions and any
securities into which or for which any or all of such shares may be changed or exchanged or which are received in such transaction.
4. Action in Stockholder Capacity Only. The Stockholder is entering into this Agreement solely in the Stockholder’s capacity as a record holder and/or
beneficial owner, as applicable, of its Shares and not in the Stockholder’s capacity as a director or officer of the Company. Nothing herein shall limit or affect the Stockholder’s ability to act as an officer or director of the Company.
Page 3
5. Irrevocable Proxy. The Stockholder hereby revokes (or agrees to cause to be revoked) any proxies that the Stockholder has heretofore granted with respect
to its Shares. The Stockholder hereby irrevocably grants to, and appoints, the president of the Company, and any individual designated in writing by the Company, and each of them individually, as his, her or its proxy and attorney-in-fact (with
full power of substitution), for and in its name, place and stead, to vote his, her or its Shares in any action by written consent of Company stockholders or at any meeting of the Company’s stockholders called with respect to any of the matters
specified in, and in accordance and consistent with, Section 3 of this Agreement, if and only the Stockholder fails to vote the Shares in accordance with Section 3 at any applicable meeting of the stockholders of the Company or
pursuant to any applicable written consent of the stockholders of the Company. The Company agrees not to exercise the proxy granted herein for any purpose other than the purposes described in this Agreement and the Stockholder affirms that the
proxy set forth in this Section 5 is given in connection with, and granted in consideration of, and as an inducement to the Company, Parent and Merger Subs to enter into the Merger Agreement and that such proxy is given to secure the
obligations of the Stockholder under Section 3. Except as otherwise provided for herein, the Stockholder hereby affirms that the irrevocable proxy is coupled with an interest and may under no circumstances be revoked and that such
irrevocable proxy is executed and intended to be irrevocable. The irrevocable proxy and power of attorney granted herein shall survive and not be affected by the death or incapacity of such Stockholder. Notwithstanding any other provisions of
this Agreement, the irrevocable proxy granted hereunder shall automatically terminate on the Expiration Date.
6. No Solicitation. Subject to Section 4, the Stockholder agrees not to, directly or indirectly, including through any of its officers, directors or
agents, take any action that the Company is prohibited from taking pursuant to Section 5.4 of the Merger Agreement and Section 5.4 of the Merger Agreement is hereby incorporated by reference mutatis mutandis.
7. Documentation and Information. The Stockholder shall permit and hereby authorizes Parent and the Company to publish and disclose in all documents and
schedules filed with the SEC, and any press release or other disclosure document that Parent or the Company reasonably determines to be necessary in connection with the Merger and any of the Contemplated Transactions, a copy of this Agreement,
the Stockholder’s identity and ownership of the Shares and the nature of the Stockholder’s commitments and obligations under this Agreement; provided, that, Parent and the Company provide such documents, schedules, press release or other
disclosure document to the Stockholder in advance for its review and comment. Each of Parent and the Company is an intended third-party beneficiary of this Section 7.
8. No Exercise of Appraisal Rights; Waivers. The Stockholder hereby irrevocably and unconditionally (a) waives, and agrees to cause to be
waived and to prevent the exercise of, any rights of appraisal, any dissenters’ rights and any similar rights (including any notice requirements related thereto) relating to the Merger that Stockholder may have by virtue of, or with respect to,
any Shares (including all rights under Section 262 of the DGCL or any right to assert a claim for quasi-appraisal or similar equitable remedy) and (b) agrees that the Stockholder will not bring, commence, institute, maintain, prosecute or
voluntarily aid or participate in any action, claim, suit or cause of action, in law or in equity, in any court or before any Governmental Authority, which (i) challenges the validity of or seeks to enjoin the operation of any provision of this
Agreement or (ii) alleges that (A) the execution and delivery of this Agreement by the Stockholder breaches any duty such Stockholder has (or may be alleged to have) to the Company or to the other Company stockholders or (B) the approval of the
Merger Agreement by the Company Board breaches any fiduciary duty of the Company Board or any member thereof; provided, that (1) the Stockholder may defend against, contest or settle any such action, claim, suit or cause of action brought against
the Stockholder that relates solely to the Stockholder’s capacity as a director, officer or securityholder of the Company and (2) the foregoing shall not limit or restrict in any manner the Stockholder from enforcing the Stockholder’s rights
under this Agreement and the other agreements entered into by the Stockholder in connection herewith, or otherwise in connection with the Merger, including the Stockholder’s right to receive the Merger Consideration pursuant to the terms of the
Merger Agreement.
Page 4
9. Representations and Warranties of the Stockholder. The Stockholder hereby represents and warrants to Parent and the Company as follows:
(a) (i) The Stockholder is the beneficial or record owner of the shares of Company Capital Stock, and/or Company Options indicated in Appendix A (each of
which shall be deemed to be “held” by the Stockholder for purposes of Section 3 unless otherwise expressly stated with respect to any shares in Appendix A), free and clear of any and all Encumbrances (except for any Encumbrance
that may be imposed pursuant to this Agreement, the Voting Agreement, the Investors’ Rights Agreement of the Company, dated as of April 13, 2023 (the “Investors’ Rights Agreement”), the Right of First Refusal and Co-Sale Agreement of the
Company, dated as of April 13, 2023 (the “ROFR”), any lock-up agreement entered into by and between the Stockholder, the Company and Parent, and Encumbrances arising under applicable securities or community property laws); and (ii) the
Stockholder does not beneficially own any securities of the Company other than the shares of Company Capital Stock and rights to purchase shares of Company Capital Stock set forth in Appendix A.
(b) With respect to any Stockholder that is an entity, the Stockholder is duly organized, validly existing and in good standing under the laws of the jurisdiction
of its formation and is qualified to conduct its business in those jurisdictions necessary to perform this Agreement.
(c) Except as otherwise provided in this Agreement, the Stockholder has full power, legal capacity and authority to (i) make, enter into and carry out the terms of
this Agreement and (ii) vote all of its Shares in the manner set forth in this Agreement without the consent or approval of, or any other action on the part of, any other person or entity (including any Governmental Authority). Without limiting
the generality of the foregoing, except for the Voting Agreement, the Stockholder has not entered into any voting agreement (other than this Agreement) with any person with respect to any of the Stockholder’s Shares, granted any person any proxy
(revocable or irrevocable) or power of attorney with respect to any of the Stockholder’s Shares, deposited any of the Stockholder’s Shares in a voting trust or entered into any arrangement or agreement with any person limiting or affecting the
Stockholder’s legal power, authority or right to vote the Stockholder’s Shares on any matter.
(d) This Agreement has been duly and validly executed and delivered by the Stockholder and (assuming the due authorization, execution and delivery by the other
parties hereto) constitutes a valid and binding agreement of the Stockholder enforceable against the Stockholder in accordance with its terms, subject to the Enforceability Exceptions. The execution and delivery of this Agreement by the
Stockholder and the performance by the Stockholder of the agreements and obligations hereunder will not result in any breach or violation of or be in conflict with or constitute a default under any term of any Contract or if applicable any
provision of an organizational document (including a certificate of incorporation) to or by which the Stockholder is a party or bound, or any applicable law to which the Stockholder (or any of the Stockholder’s assets) is subject or bound, except
for any such breach, violation, conflict or default which, individually or in the aggregate, would not reasonably be expected to materially impair or adversely affect the Stockholder’s ability to perform its obligations under this Agreement.
(e) The execution, delivery and performance of this Agreement by the Stockholder do not and will not require any consent, approval, authorization or permit of,
action by, filing with or notification to, any Governmental Authority, except for any such consent, approval, authorization, permit, action, filing or notification the failure of which to make or obtain, individually or in the aggregate, has not
and would not materially impair the Stockholder’s ability to perform its obligations under this Agreement.
Page 5
(f) The Stockholder has had the opportunity to review the Merger Agreement and this Agreement with counsel of the Stockholder’s own choosing. The Stockholder has
had an opportunity to review with its own tax advisors the tax consequences of the Merger and the Contemplated Transactions. The Stockholder understands that it must rely solely on its advisors and not on any statements or representations made by
Parent, the Company or any of their respective agents or representatives with respect to the tax consequences of the Merger and the Contemplated Transactions. The Stockholder understands that such Stockholder (and not Parent, the Company, or the
Surviving Entity) shall be responsible for such Stockholder’s tax liability that may arise as a result of the Merger or the other Contemplated Transactions. The Stockholder understands and acknowledges that the Company, Parent and Merger Sub are
entering into the Merger Agreement in reliance upon the Stockholder’s execution, delivery and performance of this Agreement.
(g) With respect to the Stockholder, as of the date hereof, there is no action, suit, investigation or proceeding pending against, or, to the knowledge of the
Stockholder, threatened against, the Stockholder or any of the Stockholder’s properties or assets (including the Shares) that would reasonably be expected to prevent or materially delay or impair the ability of the Stockholder to perform its
obligations hereunder or to consummate the transactions contemplated hereby.
10. Certain Agreements. Each Stockholder, by this Agreement, and with respect to such Stockholder’s Shares, severally and not jointly, hereby agrees to
terminate, subject to the occurrence of, and effective immediately prior to, the Effective Time each of (a) the Voting Agreement, the Investors’ Rights Agreement and the ROFR and (b) any rights under any letter agreement providing for redemption
rights, put rights, purchase rights, information rights, rights to consult with and advise management, inspection rights, preemptive rights, board of directors observer rights or rights to receive information delivered to the board of directors
or other similar rights not generally available to stockholders of the Company between the Stockholder and the Company, but excluding, for the avoidance of doubt, any rights the Stockholder may have that relate to any indemnification, commercial,
development or employment agreements or arrangements between such Stockholder and the Company or any subsidiary of the Company, which shall survive in accordance with their terms. Each Stockholder hereby terminates and waives all rights of first
refusal, redemption rights and rights of notice of the Merger and the other transactions contemplated by the Merger Agreement, effective as of immediately prior to, and contingent upon, the Effective Time.
11. Termination. This Agreement shall terminate and shall cease to be of any further force or effect as of the earliest of (a) such date and time as the
Merger Agreement shall have been terminated pursuant to the terms thereof, (b) the Effective Time or (c) the time this Agreement is terminated upon mutual written agreement of the parties to this Agreement (clauses (a)-(c), the “Expiration Date”); provided, however, that (i) Section 12 shall survive the termination of this Agreement, and (ii) the
termination of this Agreement shall not relieve any party hereto from any liability for any material and willful breach of this Agreement prior to the Expiration Date.
12. Miscellaneous Provisions.
(a) Amendments. No amendment of this Agreement shall be effective against any party unless it shall be in writing and signed by each of the parties hereto.
(b) Entire Agreement; Counterparts; Exchanges by Electronic Transmission or Facsimile. This Agreement constitutes the entire agreement between the parties to this Agreement and
supersedes all other prior agreements, arrangements and understandings, both written and oral, among the parties with respect to the subject matter hereof. This Agreement may be executed in several counterparts, each of which shall be deemed an
original and all of which shall constitute one and the same instrument. The exchange of a fully executed Agreement (in counterparts or otherwise) by all parties by facsimile or electronic transmission in PDF format shall be sufficient to bind the
parties to the terms and conditions of this Agreement.
Page 6
(c) Applicable Law; Jurisdiction. This Agreement shall be governed by, and construed in accordance with, the laws of the State of Delaware, regardless of the laws that might
otherwise govern under applicable principles of conflicts of laws. In any action or proceeding between any of the parties arising out of or relating to this Agreement, each of the parties: (i) irrevocably and unconditionally consents and submits to
the exclusive jurisdiction and venue of the Court of Chancery of the State of Delaware or, to the extent such court does not have subject matter jurisdiction, the Superior Court of the State of Delaware or the United States District Court for the
District of Delaware, (ii) agrees that all claims in respect of such action or proceeding shall be heard and determined exclusively in accordance with clause (i) of this Section 12(c), (iii) waives any objection to laying venue in any such
action or proceeding in such courts, (iv) waives any objection that such courts are an inconvenient forum or do not have jurisdiction over any party, (v) agrees that service of process upon such party in any such action or proceeding shall be
effective if notice is given in accordance with Section 12h) of this Agreement and (vi) irrevocably and unconditionally waives the right to trial by jury.
(d) Assignment. This Agreement shall be binding upon, and shall be enforceable by and inure solely to the benefit of, the parties and their respective successors and permitted
assigns; provided, however, that neither this Agreement nor any of a party’s rights or obligations hereunder may be assigned or delegated (except by Merger) by such party without the prior written consent of the other party, and any
attempted assignment or delegation of this Agreement or any of such rights or obligations by such party without the other party’s prior written consent shall be void and of no effect.
(e) No Third Party Rights. Nothing in this Agreement, express or implied, is intended to or shall confer upon any Person any right, benefit or remedy of any nature whatsoever under
or by reason of this Agreement.
(f) Severability. Any term or provision of this Agreement that is invalid or unenforceable in any situation in any jurisdiction shall not affect the validity or enforceability of
the remaining terms and provisions of this Agreement or the validity or enforceability of the offending term or provision in any other situation or in any other jurisdiction. If a final judgment of a court of competent jurisdiction declares that
any term or provision of this Agreement is invalid or unenforceable, the parties agree that the court making such determination shall have the power to limit such term or provision, to delete specific words or phrases or to replace such term or
provision with a term or provision that is valid and enforceable and that comes closest to expressing the intention of the invalid or unenforceable term or provision, and this Agreement shall be valid and enforceable as so modified. In the event
such court does not exercise the power granted to it in the prior sentence, the parties agree to replace such invalid or unenforceable term or provision with a valid and enforceable term or provision that will achieve, to the extent possible, the
economic, business and other purposes of such invalid or unenforceable term or provision.
(g) Specific Performance. Except as otherwise provided herein, any and all remedies herein expressly
conferred upon a party will be deemed cumulative with and not exclusive of any other remedy conferred hereby, or by law or equity upon such party, and the exercise by a party of any one remedy will not preclude the exercise of any other remedy. The
parties agree that irreparable damage for which monetary damages, even if available, would not be an adequate remedy, would occur in the event that any of the provisions of this Agreement were not performed in accordance with their specific terms
(including failing to take such actions as are required of it hereunder to consummate this Agreement) or were otherwise breached. It is accordingly agreed that the parties shall be entitled to an injunction or injunctions to prevent breaches of
this Agreement and to enforce specifically the terms and provisions hereof the Court of Chancery of the State of Delaware or, to the extent such court does not have subject matter jurisdiction, the Superior Court of the State of Delaware or the
United States District Court for the District of Delaware, this being in addition to any other remedy to which they are entitled at law or in equity, and each of the parties waives any bond, surety or other security that might be required of any
other party with respect thereto. Each of the parties further agrees that it will not oppose the granting of an injunction, specific performance or other equitable relief on the basis that any other party has an adequate remedy at law or that any
award of specific performance is not an appropriate remedy for any reason at law or in equity.
Page 7
(h) Notices. All notices and other communications hereunder shall be in writing and shall be deemed duly delivered (i) one (1) Business Day after being sent for next Business Day
delivery, fees prepaid, via a reputable international overnight courier service, (ii) upon delivery in the case of delivery by hand or (iii) on the date delivered in the place of delivery if sent by email or facsimile (with a written or electronic
confirmation of delivery) prior to 6:00 p.m. (New York City time), otherwise on the next succeeding Business Day, (A) if to the Company or Parent, to the address, electronic mail address or facsimile provided in Section 11.7 of the Merger
Agreement, including to the persons designated therein to receive copies; and/or (B) if to the Stockholder, to the Stockholder’s address, electronic mail address or facsimile shown below Stockholder’s signature to this Agreement.
(i) Confidentiality. Except to the extent required by applicable Law or regulation, the Stockholder shall hold any non-public information regarding the
Company, this Agreement, the Merger Agreement and the Merger in strict confidence and shall not divulge any such information to any third person until the Company and Parent have publicly disclosed their entry into the Merger Agreement and this
Agreement; provided, however, that the Stockholder may disclose such information to its Affiliates, attorneys, accountants, consultants, and other advisors (provided that such Persons are subject to confidentiality obligations at
least as restrictive as those contained herein). Neither the Stockholder nor any of its Affiliates (other than the Company, whose actions shall be governed by the Merger Agreement), shall issue or cause the publication of any press release or
other public announcement with respect to the Company, this Agreement, the Merger, the Merger Agreement or the other transactions contemplated hereby or thereby without the prior written consent of the Company and Parent, except as may be required
by applicable Law in which circumstance such announcing party shall make reasonable efforts to consult with the Company and Parent to the extent practicable.
Page 8
(j) Interpretation. The words “hereof,” “herein” and “hereunder” and words of like import used in this Agreement shall refer to this Agreement as a whole and not to any particular
provision of this Agreement. The captions herein are included for convenience of reference only and shall be ignored in the construction or interpretation hereof. References to Sections and Appendixes are to Sections and Appendixes of this
Agreement unless otherwise specified. Any capitalized terms used in any Appendix but not otherwise defined therein shall have the meaning as defined in this Agreement. Any singular term in this Agreement shall be deemed to include the plural, and
any plural term the singular, the masculine gender shall include the feminine and neuter genders; the feminine gender shall include the masculine and neuter genders; and the neuter gender shall include masculine and feminine gender. Whenever the
words “include,” “includes” or “including” are used in this Agreement, they shall be deemed to be followed by the words “without limitation,” whether or not they are in fact followed by those words or words of like import. The word “or” is not
exclusive. “Writing,” “written” and comparable terms refer to printing, typing and other means of reproducing words (including electronic media) in a visible form. References to any agreement or Contract are to that agreement or Contract as
amended, modified or supplemented from time to time in accordance with the terms hereof and thereof. References to any Person include the successors and permitted assigns of that Person. References to any statute are to that statute and to the
rules and regulations promulgated thereunder, in each case as amended, modified, re-enacted thereof, substituted, from time to time. References to “$” and “dollars” are to the currency of the United States. All accounting terms used herein will be
interpreted, and all accounting determinations hereunder will be made, in accordance with GAAP unless otherwise expressly specified. References from or through any date shall mean, unless otherwise specified, from and including or through and
including, respectively. All references to “days” shall be to calendar days unless otherwise indicated as a “Business Day.” Except as otherwise specifically indicated, for purposes of measuring the beginning and ending of time periods in this
Agreement (including for purposes of “Business Day” and for hours in a day or Business Day), the time at which a thing, occurrence or event shall begin or end shall be deemed to occur in the Eastern time zone of the United States. The parties agree
that any rule of construction to the effect that ambiguities are to be resolved against the drafting party shall not be applied in the construction or interpretation of this Agreement.
[Remainder of Page Left Intentionally Blank]
Page 9
IN WITNESS WHEREOF, the undersigned have caused this Agreement to be duly executed as of the date first above written.
COMPANY:
Cullgen Inc.
|
By:
|
||
|
Title:
|
||
|
PARENT:
|
||
|
Gyre Therapeutics, Inc.
|
||
|
By:
|
||
|
Title:
|
||
|
[STOCKHOLDER],
|
||
|
in his/her capacity as the Stockholder:
|
||
|
Signature:
|
||
|
Address:
|
||
[Signature Page to Company Support Agreement]
Appendix A
|
Name of Stockholder
|
Shares of
Company
Capital Stock
|
Shares Underlying
Company Options
|
||
Exhibit 10.2
FORM OF PARENT STOCKHOLDER SUPPORT AGREEMENT
This Support Agreement (this “Agreement”) is made and entered into as of March 2, 2026, by and among Cullgen Inc., a Delaware corporation (the “Company”), Gyre Therapeutics, Inc., a
Delaware corporation (“Parent”), and the undersigned stockholder (the “Stockholder”) of Parent. Capitalized terms used herein but not otherwise defined shall have the respective meanings ascribed
to such terms in the Merger Agreement (as defined below).
RECITALS
WHEREAS, concurrently with the execution and delivery hereof, Parent, the Company, and Helix Merger Sub Corp., a Delaware corporation and a wholly owned subsidiary of Parent (“Merger Sub”),
have entered into an Agreement and Plan of Merger and Reorganization, dated of even date herewith (as such agreement may be amended or supplemented from time to time pursuant to the terms thereof, the “Merger Agreement”), pursuant to which
Merger Sub will merge with and into the Company (the “Merger”), with the Company surviving the Merger as the surviving corporation and a wholly owned subsidiary of Parent, upon the terms and subject to the conditions set forth in the Merger
Agreement.
WHEREAS, as of the date hereof, the Stockholder is the beneficial owner (as defined in Rule 13d-3 under the Exchange Act) and has sole or shared voting power with respect to such number of shares
of Parent Capital Stock, as indicated in Appendix A.
WHEREAS, as an inducement and a condition to the willingness of the Company to enter into the Merger Agreement, each Stockholder has agreed to enter into and perform this Agreement.
NOW, THEREFORE, intending to be legally bound, the parties hereby agree as follows:
1. Certain Definitions. Capitalized terms used but not otherwise defined herein shall have the meanings ascribed thereto in the Merger Agreement. For all purposes of this Agreement,
the following terms shall have the following respective meanings:
(a) “Constructive Sale” means, with respect to any security, a short sale with respect to such security, entering into or acquiring a derivative contract with respect to such
security, entering into or acquiring a futures or forward contract to deliver such security or entering into any other hedging or other derivative transaction that has the effect of either directly or indirectly materially changing the economic
benefits or risks of ownership of such security.
(b) “Parent Stockholder Matters” means the approval of the Preferred Stock Conversion Proposal.
(c) “Shares” means (i) all shares of Parent Capital Stock owned, beneficially or of record, by the Stockholder as of the date hereof, (ii) all additional shares of Parent Capital
Stock acquired by the Stockholder, beneficially or of record, during the period commencing with the execution and delivery of this Agreement and expiring on the Expiration Date (as defined below) and (iii) any shares of capital stock or other
equity securities of Parent that such Stockholder acquires or with respect to which such Stockholder otherwise acquires sole or shared voting power (including any proxy), whether beneficial or of record, or otherwise owned by such Stockholder after
the execution and delivery of this Agreement and expiring on the Expiration Date, whether by exercise of any Parent Options or otherwise, including, without limitation, by gift, succession, in the event of a stock split or as a dividend or
distribution of any Shares.
Page 1
(d) “Transfer” or “Transferred” means, with respect to any security, the direct or indirect assignment, sale, transfer, tender, exchange, pledge or hypothecation, or the grant,
creation or suffrage of an Encumbrance, lien, security interest or encumbrance in or upon, or the gift, grant or placement in trust, or the Constructive Sale or other disposition of such security (including transfers by testamentary or intestate
succession, by domestic relations order or other court order, or otherwise by operation of law) or any right, title or interest therein (including any right or power to vote to which the holder thereof may be entitled, whether such right or power
is granted by proxy or otherwise), or the record or beneficial ownership thereof, the offer to make such a sale, transfer, Constructive Sale or other disposition, and each agreement, arrangement or understanding, whether or not in writing, to
effect any of the foregoing.
2. Transfer and Voting Restrictions. The Stockholder covenants to the Company and Parent as follows:
(a) Except as otherwise permitted by Section 2(c), during the period commencing with the execution and delivery of this Agreement and expiring on the Expiration Date (as defined
below), the Stockholder shall not Transfer any of the Stockholder’s Shares, publicly announce its intention to Transfer any of its Shares or enter into any contract with respect to any Transfer of, its Shares or any interest therein.
(b) Except as otherwise permitted by this Agreement or otherwise permitted or required by order of a court of competent jurisdiction or a Governmental Authority, the Stockholder will not
commit any act that would restrict the Stockholder’s legal power, authority and right to vote all of the Shares held by the Stockholder or otherwise prevent or disable the Stockholder from performing any of his, her or its obligations under this
Agreement. Without limiting the generality of the foregoing, except for this Agreement, and as otherwise permitted by this Agreement, the Stockholder shall not enter into any voting agreement with any person or entity with respect to any of the
Stockholder’s Shares, grant any person or entity any proxy (revocable or irrevocable) or power of attorney with respect to any of the Shares, deposit any Shares in a voting trust or otherwise enter into any agreement or arrangement with any person
or entity in each case, which has the effect of limiting or affecting the Stockholder’s legal power, authority or right to vote the Stockholder’s Shares in favor of the Parent Stockholder Matters and against any competing proposals.
(c) Except as otherwise permitted by this Agreement or otherwise permitted or required by order of a court of competent jurisdiction or a Governmental Authority, the Stockholder will not
enter into any Contract, option, commitment or other arrangement or understanding with respect to the direct or indirect Transfer of any right, title or interest (including any right or power to vote to which the holder thereof may be entitled
whether such right or power is granted by proxy or otherwise) to any Shares or take any action that would reasonably be expected to make any representation or warranty of such Stockholder contained herein untrue or incorrect or have the effect of
restricting the Stockholder’s legal power, authority and right to vote all of the Shares or would otherwise prevent or disable such Stockholder from performing any of such Stockholder’s obligations under this Agreement.
Page 2
(d) Notwithstanding anything else herein to the contrary, the Stockholder may, at any time, Transfer Shares (i) by will or other testamentary document or by intestacy, (ii) to such
Stockholder’s Affiliates (in each case, directly or indirectly), (iii) to any member of the Stockholder’s immediate family (or, if the Stockholder is a corporation, partnership or other entity, to an immediate family member of a beneficial owner of
the Shares held by the Stockholder), (iv) to any trust or other entity for the direct or indirect benefit of the Stockholder or the immediate family of the Stockholder (or, if the Stockholder is a corporation, partnership or other entity, for the
direct or indirect benefit of an immediate family member of a beneficial owner of the Shares held by the Stockholder) or otherwise for estate tax or estate planning purposes, (v) in the case of a Stockholder who is not a natural person, by pro rata
distributions from the Stockholder to its members, partners, or shareholders pursuant to the Stockholder’s organizational documents, (vi) with respect to such Stockholder’s Parent Options (and any Shares underlying such Parent Options) which expire
on or prior to the Expiration Date, Transfers of Shares to Parent (or effecting a “net exercise” of a Parent Option) as payment for the (a) exercise price of such Stockholder’s Parent Options and (b) taxes applicable to the exercise of such
Stockholder’s Parent Options, (vii) transfers to another holder of capital stock of Parent that has signed a support agreement that is reasonably acceptable to the Company, and (viii) transfers, sales or other dispositions as the Company may
otherwise agree in writing in its sole discretion; provided, that in the cases of clauses (i)-(viii), (1) such Transferred Shares shall continue to be bound by this Agreement and (2) the applicable direct transferee (if any) of such
Transferred Shares shall have executed and delivered to Parent and the Company a support agreement substantially identical to this Agreement upon consummation of the Transfer if not already a party thereto, or (ix) to the extent required by
applicable Law. Any action taken in violation of Section 2(a) through Section 2(d) shall be null and void ab initio.
(e) Notwithstanding anything to the contrary herein, nothing in this Agreement shall obligate the Stockholder to exercise any option or any other right to acquire any shares of Parent
Capital Stock.
3. Agreement to Vote Shares. The Stockholder covenants to the Company and Parent as follows:
(a) Until the Expiration Date (as defined below), at any meeting of the stockholders of Parent called to vote upon the Parent Stockholder Matters, however called, and at every adjournment
or postponement thereof, and on every action or approval by written consent of the stockholders of Parent, the Stockholder shall be present (in person or by proxy) and vote (including by submitting the proxy solicited by Parent with respect to such
meeting), or exercise its right to consent with respect to, all Shares held by the Stockholder (A) in favor of the Parent Stockholder Matters, and (B) against any Acquisition Proposal.
(b) If the Stockholder is the beneficial owner, but not the record holder, of Shares, the Stockholder agrees to take all actions necessary to cause the record holder and any nominees to be
present (in person or by proxy) and vote all the Stockholder’s Shares in accordance with this Section 3.
(c) In the event of a stock split, stock dividend or distribution, or any change in the capital stock of Parent by reason of any split-up, reverse stock split, recapitalization, combination,
reclassification, reincorporation, exchange of shares or the like, the term “Shares” shall be deemed to refer to and include such shares as well as all such stock dividends and distributions and any securities into which or for which any or all of
such shares may be changed or exchanged or which are received in such transaction.
4. Action in Stockholder Capacity Only. The Stockholder is entering into this Agreement solely in the Stockholder’s capacity as a record holder and/or
beneficial owner, as applicable, of its Shares and not in the Stockholder’s capacity as a director or officer of Parent. Nothing herein shall limit or affect the Stockholder’s ability to act as an officer or director of Parent.
Page 3
5. Irrevocable Proxy. The Stockholder hereby revokes (or agrees to cause to be revoked) any proxies that the Stockholder has heretofore granted with respect to its Shares. The
Stockholder hereby irrevocably grants to, and appoints, the chairman of the Board of Directors of Parent, and any individual designated in writing by Parent, and each of them individually, as his, her or its proxy and attorney-in-fact (with full
power of substitution), for and in its name, place and stead, to vote his, her or its Shares in any action by written consent of Parent stockholders or at any meeting of Parent’s stockholders called with respect to any of the matters specified in,
and in accordance and consistent with, Section 3 of this Agreement, if and only the Stockholder fails to vote the Shares in accordance with Section 3 at any applicable meeting of the stockholders of Parent or pursuant to any
applicable written consent of the stockholders of Parent. Parent agrees not to exercise the proxy granted herein for any purpose other than the purposes described in this Agreement and the Stockholder affirms that the proxy set forth in this Section
5 is given in connection with, and granted in consideration of, and as an inducement to the Company, Parent and Merger Sub to enter into the Merger Agreement and that such proxy is given to secure the obligations of the Stockholder under Section
3. Except as otherwise provided for herein, the Stockholder hereby affirms that the irrevocable proxy is coupled with an interest and may under no circumstances be revoked and that such irrevocable proxy is executed and intended to be
irrevocable. The irrevocable proxy and power of attorney granted herein shall survive and not be affected by the death or incapacity of such Stockholder. Notwithstanding any other provisions of this Agreement, the irrevocable proxy granted
hereunder shall automatically terminate on the Expiration Date.
6. No Solicitation. Subject to Section 4, the Stockholder agrees not to, directly or indirectly, including through any of its officers, directors
or agents, take any action that Parent is prohibited from taking pursuant to Section 5.4 of the Merger Agreement and Section 5.4 of the Merger Agreement is hereby incorporated by reference mutatis mutandis.
7. Documentation and Information. The Stockholder shall permit and hereby authorizes Parent and the Company to publish and disclose in all documents and schedules filed with the
SEC, and any press release or other disclosure document that Parent or the Company reasonably determines to be necessary in connection with the Merger and any of the Contemplated Transactions, a copy of this Agreement, the Stockholder’s identity
and ownership of the Shares and the nature of the Stockholder’s commitments and obligations under this Agreement. Each of Parent and the Company is an intended third-party beneficiary of this Section 7.
8. Representations and Warranties of the Stockholder. The Stockholder hereby represents and warrants to Parent and the Company as follows:
(a) (i) The Stockholder is the beneficial or record owner of the shares of Parent Capital Stock indicated in Appendix A (each of which shall be deemed to be “held” by the
Stockholder for purposes of Section 3 unless otherwise expressly stated with respect to any shares in Appendix A), free and clear of any and all Encumbrances (except for any Encumbrance that may be imposed pursuant to this
Agreement, and Encumbrances arising under applicable securities or community property laws); and (ii) the Stockholder does not beneficially own any securities of Parent other than the shares of Parent Capital Stock and rights to purchase shares of
Parent Capital Stock set forth in Appendix A.
(b) With respect to any Stockholder that is an entity, the Stockholder is duly organized, validly existing, and in good standing under the laws of the jurisdiction of its formation and is
qualified to conduct its business in those jurisdictions necessary to perform this Agreement.
(c) Except as otherwise provided in this Agreement, the Stockholder has full power, legal capacity and authority to (i) make, enter into and carry out the terms of this Agreement and (ii)
vote all of its Shares in the manner set forth in this Agreement without the consent or approval of, or any other action on the part of, any other person or entity (including any Governmental Authority). Without limiting the generality of the
foregoing, the Stockholder has not entered into any voting agreement (other than this Agreement) with any person with respect to any of the Stockholder’s Shares, granted any person any proxy (revocable or irrevocable) or power of attorney with
respect to any of the Stockholder’s Shares, deposited any of the Stockholder’s Shares in a voting trust or entered into any arrangement or agreement with any person limiting or affecting the Stockholder’s legal power, authority or right to vote the
Stockholder’s Shares on any matter.
Page 4
(d) This Agreement has been duly and validly executed and delivered by the Stockholder and (assuming the due authorization, execution and delivery by the other parties hereto) constitutes a
valid and binding agreement of the Stockholder enforceable against the Stockholder in accordance with its terms, subject to the Enforceability Exceptions. The execution and delivery of this Agreement by the Stockholder and the performance by the
Stockholder of the agreements and obligations hereunder will not result in any breach or violation of or be in conflict with or constitute a default under any term of any Contract or if applicable any provision of an organizational document
(including a certificate of incorporation) to or by which the Stockholder is a party or bound, or any applicable law to which the Stockholder (or any of the Stockholder’s assets) is subject or bound, except for any such breach, violation, conflict
or default which, individually or in the aggregate, would not reasonably be expected to materially impair or adversely affect the Stockholder’s ability to perform its obligations under this Agreement.
(e) The execution, delivery and performance of this Agreement by the Stockholder do not and will not require any consent, approval, authorization or permit of, action by, filing with or
notification to, any Governmental Authority, except for any such consent, approval, authorization, permit, action, filing or notification the failure of which to make or obtain, individually or in the aggregate, has not and would not materially
impair the Stockholder’s ability to perform its obligations under this Agreement.
(f) The Stockholder has had the opportunity to review the Merger Agreement and this Agreement with counsel of the Stockholder’s own choosing. The Stockholder has had an opportunity to review
with its own tax advisors the tax consequences of the Merger and the other Contemplated Transactions. The Stockholder understands that it must rely solely on its advisors and not on any statements or representations made by Parent, the Company or
any of their respective agents or representatives with respect to the tax consequences of the Merger and the other Contemplated Transactions. The Stockholder understands that such Stockholder (and not Parent, the Company, or the Surviving Entity)
shall be responsible for such Stockholder’s tax liability that may arise as a result of the Merger or the other Contemplated Transactions. The Stockholder understands and acknowledges that the Company, Parent and Merger Sub are entering into the
Merger Agreement in reliance upon the Stockholder’s execution, delivery and performance of this Agreement.
(g) With respect to the Stockholder, as of the date hereof, there is no action, suit, investigation or proceeding pending against, or, to the knowledge of the Stockholder, threatened
against, the Stockholder or any of the Stockholder’s properties or assets (including the Shares) that would reasonably be expected to prevent or materially delay or impair the ability of the Stockholder to perform its obligations hereunder or to
consummate the transactions contemplated hereby.
9. Termination. This Agreement shall terminate and shall cease to be of any further force or effect as of the earliest of (a) such date and time as the Preferred Stock Conversion
Proposal shall have been approved by the stockholders of Parent, (b) twelve (12) months following the date hereof, or (c) the time this Agreement is terminated upon mutual written agreement of the Stockholder, the Company and Parent (clauses
(a)-(c), the “Expiration Date”); provided, however, that (i) Section 10 shall survive the termination of this Agreement, and (ii) the
termination of this Agreement shall not relieve any party hereto from any liability for any material and willful breach of this Agreement prior to the Expiration Date.
(a) Amendments. No amendment of this Agreement shall be effective against any party unless it shall be in writing and signed by each of the parties hereto.
Page 5
(b) Entire Agreement; Counterparts; Exchanges by Electronic Transmission or Facsimile. This Agreement constitutes the entire agreement between the parties to this Agreement and
supersedes all other prior agreements, arrangements and understandings, both written and oral, among the parties with respect to the subject matter hereof. This Agreement may be executed in several counterparts, each of which shall be deemed an
original and all of which shall constitute one and the same instrument. The exchange of a fully executed Agreement (in counterparts or otherwise) by all parties by facsimile or electronic transmission in PDF format shall be sufficient to bind the
parties to the terms and conditions of this Agreement.
(c) Applicable Law; Jurisdiction. This Agreement shall be governed by, and construed in accordance with, the laws of the State of Delaware, regardless
of the laws that might otherwise govern under applicable principles of conflicts of laws. In any action or proceeding between any of the parties arising out of or relating to this Agreement, each of the parties: (i) irrevocably and unconditionally
consents and submits to the exclusive jurisdiction and venue of the Court of Chancery of the State of Delaware or, to the extent such court does not have subject matter jurisdiction, the Superior Court of the State of Delaware or the United States
District Court for the District of Delaware, (ii) agrees that all claims in respect of such action or proceeding shall be heard and determined exclusively in accordance with clause (i) of this Section 10(c), (iii) waives any objection to
laying venue in any such action or proceeding in such courts, (iv) waives any objection that such courts are an inconvenient forum or do not have jurisdiction over any party, (v) agrees that service of process upon such party in any such action or
proceeding shall be effective if notice is given in accordance with Section 10(h) of this Agreement and (vi) irrevocably and unconditionally waives the right to trial by jury.
(d) Assignment. This Agreement shall be binding upon, and shall be enforceable by and inure solely to the benefit of, the parties and their respective successors and permitted
assigns; provided, however, that neither this Agreement nor any of a party’s rights or obligations hereunder may be assigned or delegated (except by the Merger) by such party without the prior written consent of the other party, and
any attempted assignment or delegation of this Agreement or any of such rights or obligations by such party without the other party’s prior written consent shall be void and of no effect.
(e) No Third Party Rights. Nothing in this Agreement, express or implied, is intended to or shall confer upon any Person any right, benefit or remedy of any nature whatsoever under
or by reason of this Agreement.
(f) Severability. Any term or provision of this Agreement that is invalid or unenforceable in any situation in any jurisdiction shall not affect the validity or enforceability of
the remaining terms and provisions of this Agreement or the validity or enforceability of the offending term or provision in any other situation or in any other jurisdiction. If a final judgment of a court of competent jurisdiction declares that
any term or provision of this Agreement is invalid or unenforceable, the parties agree that the court making such determination shall have the power to limit such term or provision, to delete specific words or phrases or to replace such term or
provision with a term or provision that is valid and enforceable and that comes closest to expressing the intention of the invalid or unenforceable term or provision, and this Agreement shall be valid and enforceable as so modified. In the event
such court does not exercise the power granted to it in the prior sentence, the parties agree to replace such invalid or unenforceable term or provision with a valid and enforceable term or provision that will achieve, to the extent possible, the
economic, business and other purposes of such invalid or unenforceable term or provision.
Page 6
(g) Specific Performance. Except as otherwise provided herein, any and all remedies herein expressly
conferred upon a party will be deemed cumulative with and not exclusive of any other remedy conferred hereby, or by law or equity upon such party, and the exercise by a party of any one remedy will not preclude the exercise of any other remedy. The
parties agree that irreparable damage for which monetary damages, even if available, would not be an adequate remedy, would occur in the event that any of the provisions of this Agreement were not performed in accordance with their specific terms
(including failing to take such actions as are required of it hereunder to consummate this Agreement) or were otherwise breached. It is accordingly agreed that the parties shall be entitled to an injunction or injunctions to prevent breaches of
this Agreement and to enforce specifically the terms and provisions hereof the Court of Chancery of the State of Delaware or, to the extent such court does not have subject matter jurisdiction, the Superior Court of the State of Delaware or the
United States District Court for the District of Delaware, this being in addition to any other remedy to which they are entitled at law or in equity, and each of the parties waives any bond, surety or other security that might be required of any
other party with respect thereto. Each of the parties further agrees that it will not oppose the granting of an injunction, specific performance or other equitable relief on the basis that any other party has an adequate remedy at law or that any
award of specific performance is not an appropriate remedy for any reason at law or in equity.
(h) Notices. All notices and other communications hereunder shall be in writing and shall be deemed duly delivered (i) one (1) Business Day after being
sent for next Business Day delivery, fees prepaid, via a reputable international overnight courier service, (ii) upon delivery in the case of delivery by hand or (iii) on the date delivered in the place of delivery if sent by email or facsimile
(with a written or electronic confirmation of delivery) prior to 6:00 p.m. (New York City time), otherwise on the next succeeding Business Day, (A) if to the Company or Parent, to the address, electronic mail address or facsimile provided in
Section 11.7 of the Merger Agreement, including to the persons designated therein to receive copies; and/or (B) if to the Stockholder, to the Stockholder’s address, electronic mail address or facsimile shown below Stockholder’s signature to this
Agreement.
(i) Confidentiality. Except to the extent required by applicable Law or regulation, the Stockholder shall hold any non-public information regarding the
Company, this Agreement, the Merger Agreement and the Merger in strict confidence and shall not divulge any such information to any third person until the Company and Parent have publicly disclosed their entry into the Merger Agreement and this
Agreement; provided, however, that the Stockholder may disclose such information to its Affiliates, attorneys, accountants, consultants, and other advisors (provided that such Persons are subject to confidentiality obligations at least as
restrictive as those contained herein). Neither the Stockholder nor any of its Affiliates (other than Parent, whose actions shall be governed by the Merger Agreement), shall issue or cause the publication of any press release or other public
announcement with respect to Parent, this Agreement, the Merger, the Merger Agreement or the other transactions contemplated hereby or thereby without the prior written consent of the Company and Parent, except as may be required by applicable Law
in which circumstance such announcing party shall make reasonable efforts to consult with the Company and Parent to the extent practicable.
Page 7
(j) Interpretation. The words “hereof,” “herein” and “hereunder” and words of like import used in this Agreement shall refer to this Agreement as a whole and not to any particular
provision of this Agreement. The captions herein are included for convenience of reference only and shall be ignored in the construction or interpretation hereof. References to Sections and Appendixes are to Sections and Appendixes of this
Agreement unless otherwise specified. Any capitalized terms used in any Appendix but not otherwise defined therein shall have the meaning as defined in this Agreement. Any singular term in this Agreement shall be deemed to include the plural, and
any plural term the singular, the masculine gender shall include the feminine and neuter genders; the feminine gender shall include the masculine and neuter genders; and the neuter gender shall include masculine and feminine gender. Whenever the
words “include,” “includes” or “including” are used in this Agreement, they shall be deemed to be followed by the words “without limitation,” whether or not they are in fact followed by those words or words of like import. The word “or” is not
exclusive. “Writing,” “written” and comparable terms refer to printing, typing and other means of reproducing words (including electronic media) in a visible form. References to any agreement or Contract are to that agreement or Contract as
amended, modified or supplemented from time to time in accordance with the terms hereof and thereof. References to any Person include the successors and permitted assigns of that Person. References to any statute are to that statute and to the
rules and regulations promulgated thereunder, in each case as amended, modified, re-enacted thereof, substituted, from time to time. References to “$” and “dollars” are to the currency of the United States. All accounting terms used herein will be
interpreted, and all accounting determinations hereunder will be made, in accordance with GAAP unless otherwise expressly specified. References from or through any date shall mean, unless otherwise specified, from and including or through and
including, respectively. All references to “days” shall be to calendar days unless otherwise indicated as a “Business Day.” Except as otherwise specifically indicated, for purposes of measuring the beginning and ending of time periods in this
Agreement (including for purposes of “Business Day” and for hours in a day or Business Day), the time at which a thing, occurrence or event shall begin or end shall be deemed to occur in the Eastern time zone of the United States. The parties agree
that any rule of construction to the effect that ambiguities are to be resolved against the drafting party shall not be applied in the construction or interpretation of this Agreement.
[Remainder of Page Left Intentionally Blank]
Page 8
IN WITNESS WHEREOF, the undersigned have caused this Agreement to be duly executed as of the date first above written.
|
COMPANY:
|
|
|
CULLGEN INC.
|
|
|
By:
|
|
|
Title:
|
[Signature Page to Parent Stockholder Support Agreement]
|
PARENT:
|
|
|
GYRE THERAPEUTICS, INC.
|
|
|
By:
|
|
|
Title:
|
[Signature Page to Parent Stockholder Support Agreement]
[STOCKHOLDER],
in his/her capacity as the Stockholder:
in his/her capacity as the Stockholder:
|
Signature:
|
||
|
Address:
|
||
[Signature Page to Parent Stockholder Support Agreement]
Appendix A
|
Name, Address and Email
Address of Stockholder
|
Shares of Parent
Capital Stock
|
Shares Underlying
Parent Options
|
||
|
|
Exhibit 10.3
Confidential
This REGISTRATION RIGHTS AGREEMENT (this “Agreement”) is made and entered into as of [●], 2026, among Cullgen Inc., a Delaware corporation (the “Company”), Gyre Therapeutics, Inc. (“Parent”), a Delaware corporation, and each of the several holders of shares of Company Capital Stock signatory hereto (each such holder,
a “Company Holder” and, collectively, the “Company Holders”).
WHEREAS, the Company is party to that certain Agreement and Plan of Merger and Reorganization by and among the Company, Helix Merger Sub Corp., a Delaware corporation (“Merger Sub”), and Parent, dated as of March 2, 2026 (the “Merger Agreement”), pursuant to which Merger Sub will merge with and into the Company, with the Company becoming a wholly-owned
subsidiary of Parent (the “Merger”);
WHEREAS, at the Effective Time, each share of Company Capital Stock outstanding immediately prior to the Effective Time (excluding shares of Company Capital Stock to be cancelled pursuant to Section
2.5(a)(i) of the Merger Agreement and excluding Dissenting Shares) will be converted solely into the right to receive a number of shares of Parent Capital Stock calculated as set forth in the Merger Agreement (collectively, the “Merger Shares”); and
WHEREAS, in connection with the consummation of the transactions contemplated by the Merger Agreement, and pursuant to the terms of the Merger Agreement, the parties desire to enter into this
Agreement in order to grant certain rights to the Company Holders as set forth below.
NOW, THEREFORE, in consideration of the covenants and promises set forth herein, and for other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, the
parties hereby agree as follows:
In addition to the terms defined herein, capitalized terms used and not otherwise defined herein that are defined in the Merger Agreement shall have the meanings given such terms in the Merger
Agreement. As used in this Agreement, the following terms shall have the following meanings:
“Advice” shall have the meaning set forth in Section 6(c).
“Effectiveness Date” means, with respect to the Resale Registration Statement required to be filed hereunder, the 90th calendar day following the date hereof
(or, in the event of a “full review” by the Commission, the 120th calendar day following the date hereof) and with respect to any additional Registration Statements that may be required pursuant to Sections 2(b) and 2(c) or Section
3(c), the 45th calendar day following the date on which an additional Registration Statement is required to be filed hereunder (or, in the event of a “full review” by the Commission, the 60th calendar day following the date thereof); provided,
however, that in the event Parent is notified by the Commission (orally or in writing) that one or more of the above Registration Statements will not be reviewed or is no longer subject to further review and comments, the Effectiveness Date as
to such Registration Statement shall be the fifth (5th) Trading Day following the date on which Parent is so notified if such date precedes the dates otherwise required above, provided, further, if such Effectiveness Date falls on a day that is not a
Trading Day, then the Effectiveness Date shall be the next succeeding Trading Day.
“Effectiveness Period” shall have the meaning set forth in Section 2(a).
“Filing Date” means, with respect to the Resale Registration Statement required hereunder, the 45th calendar day following the date hereof and, with respect to
any additional Registration Statements that may be required pursuant to Sections 2(b) and 2(c) or Section 3(c), the 30th calendar day following
the date on which Parent is permitted by SEC Guidance to file such additional Registration Statement related to the Registrable Securities.
“Holder” or “Holders” means the holder or holders, as the case may be, from time to time of Registrable Securities.
“Indemnified Party” shall have the meaning set forth in Section 5(c).
“Indemnifying Party” shall have the meaning set forth in Section 5(c).
“Losses” shall have the meaning set forth in Section 5(a).
“Other Shares” shall mean shares of Common Stock of the Company (other than Registrable Securities) with registration rights.
“Other Stockholders” shall mean persons other than Holders who, by virtue of agreements with the Company, are entitled to include their securities in certain
registrations hereunder.
“Plan of Distribution” shall have the meaning set forth in Section 2(a).
“Prospectus” means the prospectus included in a Registration Statement (including, without limitation, a prospectus that includes any information previously
omitted from a prospectus filed as part of an effective registration statement in reliance upon Rule 430A promulgated by the Commission pursuant to the Securities Act), as amended or supplemented by any prospectus supplement, with respect to the
terms of the offering of any portion of the Registrable Securities covered by a Registration Statement, and all other amendments and supplements to the Prospectus, including post-effective amendments, and all material incorporated by reference or
deemed to be incorporated by reference in such Prospectus.
“Registrable Securities” means, as of any date of determination, (a) all Merger Shares issued at the closing of the Merger to the Company Holders in respect of
all shares of capital stock of the Company held by the Company Holder as of immediately prior to the Effective Time (as defined in the Merger Agreement),including any shares of Parent common stock issuable upon conversion of any Parent Capital Stock,
(b) all shares of Parent common stock held by a Company Holder as of immediately prior to the Effective Time, if any, and (c) any securities issued or then issuable upon any stock split, dividend or other distribution, recapitalization or similar
event with respect to the foregoing; provided, however, that any such Registrable Securities shall cease to be Registrable Securities (and Parent shall not be required to maintain the effectiveness of any, or file another, Registration
Statement hereunder with respect thereto) upon the earliest to occur of (i) a Registration Statement with respect to the sale of such Registrable Securities is declared effective by the Commission under the Securities Act and such Registrable
Securities have been disposed of by the Holder in accordance with such effective Registration Statement, (ii) such Registrable Securities have been previously sold in accordance with Rule 144, and (iii) such securities become eligible for resale
without volume or manner-of-sale restrictions pursuant to Rule 144 and without the requirement for Parent to be in compliance with the current public information requirement under Rule 144, as reasonably determined by counsel to Parent in accordance
with Section 2(a).
“Registration Statement” means any registration statement required to be filed hereunder pursuant to Section 2(a) and any additional registration
statements contemplated by Section 2(c) or Section 3(c), including (in each case) the Prospectus, amendments and supplements to any such registration statement or Prospectus, including pre- and post-effective amendments, all exhibits
thereto, and all material incorporated by reference or deemed to be incorporated by reference in any such registration statement.
2
“Resale Registration Statement” means the initial Registration Statement covering the resale of all Registrable Securities filed pursuant to this Agreement.
“Rule 415” means Rule 415 promulgated by the Commission pursuant to the Securities Act, as such Rule may be amended or interpreted from time to time, or any
similar rule or regulation hereafter adopted by the Commission having substantially the same purpose and effect as such Rule.
“Rule 424” means Rule 424 promulgated by the Commission pursuant to the Securities Act, as such Rule may be amended or interpreted from time to time, or any
similar rule or regulation hereafter adopted by the Commission having substantially the same purpose and effect as such Rule.
“SEC Guidance” means (i) any publicly-available written or oral guidance of the Commission staff, or any comments, requirements or requests of the Commission
staff (whether or not publicly-available); provided, that any such oral guidance, comments, requirements or requests are reduced to writing by the Commission (and shared with the Company Holders if not publicly-available) and (ii) the Securities Act.
“Selling Stockholder Questionnaire” shall have the meaning set forth in Section 3(a).
“Trading Day” means any day on which Parent common stock is traded on a National Exchange.
(a) On or prior to each Filing Date, Parent shall prepare and file with the Commission a
Registration Statement covering the resale of all of the Registrable Securities that are not then registered on an effective Registration Statement for an offering to be made on a continuous basis pursuant to Rule 415. Each Registration Statement
filed hereunder shall be on Form S-3 (except if Parent is not then eligible to register for resale the Registrable Securities on Form S-3, in which case such registration shall be on another appropriate form in accordance with the provisions of Section
2(d)), and shall contain (unless otherwise directed by Holders holding at least 85% of Registrable Securities) disclosure substantially in the form of the “Plan of Distribution” attached hereto as Annex
A and substantially in the form of the “Selling Stockholder” section attached hereto as Annex B. Subject to the terms of this Agreement, Parent shall use reasonable best efforts to cause a
Registration Statement filed under this Agreement (including, without limitation, under Section 3(c)) to be declared effective under the Securities Act by the applicable Effectiveness Date, and shall use its reasonable best efforts to keep
such Registration Statement continuously effective under the Securities Act until the earlier of (a) the date that all Registrable Securities covered by such Registration Statement (i) have been sold, thereunder or pursuant to Rule 144, or (ii) may
be sold without volume or manner-of-sale restrictions pursuant to Rule 144 and without the requirement for Parent to be in compliance with the current public information requirement under Rule 144, as reasonably determined by the counsel to Parent
pursuant to a written opinion letter to such effect, addressed and acceptable to Parent’s transfer agent and the affected Holders (the “Effectiveness Period”). Parent shall telephonically request effectiveness
of a Registration Statement as of 5:00 p.m. (New York City time) on a Trading Day. Parent shall notify the Holders via e-mail of the effectiveness of a Registration Statement promptly following confirmation of effectiveness with the Commission.
Parent shall, in accordance with SEC Guidance, promptly file a final Prospectus with the Commission as required by Rule 424 by 9:00 a.m. on the second (2nd) Trading Day
following the date each Registration Statement is declared effective.
3
(b) Notwithstanding the registration obligations set forth in Section 2(a), if the Commission informs Parent that all of the Registrable Securities cannot, as a
result of the application of Rule 415 or other SEC Guidance, be registered for resale as a secondary offering on a single registration statement, Parent agrees to promptly inform each of the Holders thereof and use commercially reasonable efforts to
file amendments to the Resale Registration Statement as required by the Commission, covering the maximum number of Registrable Securities permitted to be registered by the Commission, on Form S-3 or such other form available to register for resale
the Registrable Securities as a secondary offering; with respect to filing on Form S-3 or other appropriate form; provided, however, that prior to filing such amendment, Parent shall be obligated to use commercially reasonable efforts
to advocate with the Commission for the registration of all of the Registrable Securities in accordance with the SEC Guidance, including without limitation, Securities Act Rules Compliance and Disclosure Interpretation 612.09.
(c) Notwithstanding any other provision of this Agreement, if the Commission or any SEC Guidance sets forth a limitation on the number of Registrable Securities permitted to
be registered on a particular Registration Statement as a secondary offering, including as a result of the application of Rule 415 (and notwithstanding that Parent used commercially reasonable efforts to advocate with the Commission for the
registration of all or a greater portion of Registrable Securities), unless otherwise directed in writing by a Holder as to its Registrable Securities, the total number of Registrable Securities to be registered on such Registration Statement will be
reduced as follows:
|
i.
|
First, Parent shall reduce Registrable Securities represented by shares of Common Stock other than the Merger Shares (applied, in the case that some but not all of such shares of Common Stock may be
registered, to the Holders on a pro rata basis based on the total number of such unregistered shares of Common Stock held by such Holders, but excluding from such pro rata calculation any unregistered shares of Common Stock held by a Holder
that the SEC has deemed to be an “underwriter” or otherwise requires such Holder to sell its shares of Common Stock in a primary offering); and
|
|
ii.
|
Second, Parent shall reduce Registrable Securities represented by the Merger Shares (applied, in the case that some but not all of Merger Shares may be registered, to the Holders on a pro rata basis based on
the total number of unregistered Merger Shares held by such Holders, but excluding from such pro rata calculation any unregistered shares of Common Stock held by a Holder that the SEC has deemed to be an “underwriter” or otherwise requires
such Holder to sell its shares of Common Stock in a primary offering).
|
In the event of a cutback hereunder, Parent shall give the Holder at least two (2) Trading Days prior written notice along with the calculations as to such Holder’s allotment. In the event Parent
amends the Resale Registration Statement in accordance with the foregoing, Parent will use its commercially reasonable efforts to file with the Commission, as promptly as allowed by the Commission or SEC Guidance provided to Parent or to registrants
of securities in general, one (1) or more registration statements on Form S-3 or such other form available to register for resale those Registrable Securities that were not registered for resale on the Resale Registration Statement, as amended.
4
(d) If Form S-3 is not available for the registration of the resale of Registrable
Securities hereunder, Parent shall (i) register the resale of the Registrable Securities on another appropriate form and (ii) undertake to register the Registrable Securities on Form S-3 as soon as such form is available, provided that Parent shall
maintain the effectiveness of the Registration Statement then in effect until such time as a Registration Statement on Form S-3 covering the Registrable Securities has been declared effective by the Commission.
In connection with Parent’s registration obligations hereunder, Parent shall:
(a) Not less than five (5) Trading Days prior to the filing of each Registration Statement and
not less than one (1) Trading Day prior to the filing of any related Prospectus or any amendment or supplement thereto (including any document that would be incorporated or deemed to be incorporated therein by reference), Parent shall (i) furnish to
each Holder copies of all such documents proposed to be filed, which documents (other than those incorporated or deemed to be incorporated by reference) will be subject to the review of such Holders, and (ii) use commercially reasonable efforts to
cause its officers and directors, counsel and independent registered public accountants to respond to such inquiries as shall be necessary, in the reasonable opinion of respective counsel to each Holder, to conduct a reasonable investigation within
the meaning of the Securities Act. Parent shall not file a Registration Statement or any such Prospectus or any amendments or supplements thereto to which the Required Holders (as defined below) shall reasonably object in good faith, provided that,
Parent is notified of such objection in writing no later than three (3) Trading Days after the Holders have been so furnished copies of a Registration Statement or one (1) Trading Day after the Holders have been so furnished copies of any related
Prospectus or amendments or supplements thereto. Each Holder agrees to furnish to Parent a completed questionnaire in the form attached to this Agreement as Annex C or such other form as reasonably acceptable to Parent (a “Selling Stockholder Questionnaire”) on a date that is not less than two (2) Trading Days prior to the Filing Date or by the end of the third (3rd) Trading Day following the date on which such Holder receives draft
materials in accordance with this Section 3. Parent shall not be required to include any Registrable Securities in the Registration Statement for any Holder that has not provided such Selling Stockholder Questionnaire.
(b) (i) Prepare and file with the Commission such amendments, including post-effective
amendments, to a Registration Statement and the Prospectus used in connection therewith as may be necessary to keep a Registration Statement continuously effective as to the applicable Registrable Securities for the Effectiveness Period and prepare
and file with the Commission such additional Registration Statements in order to register for resale under the Securities Act all of the Registrable Securities, (ii) cause the related Prospectus to be amended or supplemented by any required
Prospectus supplement (subject to the terms of this Agreement), and, as so supplemented or amended, to be filed pursuant to Rule 424, (iii) respond as promptly as reasonably possible to any comments received from the Commission with respect to a
Registration Statement or any amendment thereto and provide as promptly as reasonably possible to the Holders true and complete copies of all correspondence from and to the Commission relating to a Registration Statement (provided that, Parent shall
excise any information contained therein that would constitute material non-public information regarding Parent or any of its subsidiaries), and (iv) comply in all material respects with the applicable provisions of the Securities Act and the
Exchange Act with respect to the disposition of all Registrable Securities covered by a Registration Statement during the applicable period in accordance (subject to the terms of this Agreement) with the intended methods of disposition by the Holders
thereof set forth in such Registration Statement as so amended or in such Prospectus as so supplemented.
5
(c) If, during the Effectiveness Period, the number of Registrable Securities at any time
exceeds 100% of the number of shares of Common Stock then registered in a Registration Statement, then Parent shall, subject to Sections 2(b) and 2(c), if applicable, file as soon as reasonably practicable, an additional Registration
Statement covering the resale by the Holders of not less than the number of such Registrable Securities.
(d) Notify the Holders of Registrable Securities to be sold (which notice shall, pursuant to
clauses (iii) through (v) hereof, be accompanied by an instruction to suspend the use of the Prospectus until the requisite changes have been made) as promptly as reasonably possible (and, in the case of (i)(A) below, not less than one (1) Trading
Day prior to such filing) and (if requested by any such Person) confirm such notice in writing no later than one (1) Trading Day following the day (i)(A) when a Prospectus or any Prospectus supplement or post-effective amendment to a Registration
Statement is proposed to be filed, (B) when the Commission notifies Parent whether there will be a “review” of such Registration Statement and whenever the Commission comments in writing on such Registration Statement, and (C) with respect to a
Registration Statement or any post-effective amendment, when the same has become effective, (ii) of any request by the Commission or any other federal or state governmental authority for amendments or supplements to a Registration Statement or
Prospectus or for additional information, (iii) of the issuance by the Commission or any other federal or state governmental authority of any stop order suspending the effectiveness of a Registration Statement covering any or all of the Registrable
Securities or the initiation of any Proceeding for that purpose, (iv) of the receipt by Parent of any notification with respect to the suspension of the qualification or exemption from qualification of any of the Registrable Securities for sale in
any jurisdiction, or the initiation or threatening of any action, suit, proceeding, inquiry or investigation before or brought by any Governmental Authority (a “Proceeding”) for such purpose, and (v) of the
occurrence of any event or passage of time that makes the financial statements included in a Registration Statement ineligible for inclusion therein or any statement made in a Registration Statement or Prospectus or any document incorporated or
deemed to be incorporated therein by reference untrue in any material respect or that requires any revisions to a Registration Statement, Prospectus or other documents so that, in the case of a Registration Statement or the Prospectus, as the case
may be, it will not contain any untrue statement of a material fact or omit to state any material fact required to be stated therein or necessary to make the statements therein, in light of the circumstances under which they were made, not
misleading; provided, however, that in no event shall any such notice contain any information that would constitute material, non-public information regarding Parent or any of its subsidiaries.
(e) Use its commercially reasonable efforts to avoid the issuance of, or, if issued, obtain the withdrawal of (i) any
order stopping or suspending the effectiveness of a Registration Statement, or (ii) any suspension of the qualification (or exemption from qualification) of any of the Registrable Securities for sale in any jurisdiction, at the earliest practicable
moment.
(f) If requested by a Holder, furnish to each Holder, without charge, an electronic copy of the conformed copy of each
such Registration Statement and each amendment thereto, including financial statements and schedules, all documents incorporated or deemed to be incorporated therein by reference to the extent requested by such Person, and all exhibits to the extent
requested by such Person (including those previously furnished or incorporated by reference) promptly after the filing of such documents with the Commission, provided that any such item that is available on the EDGAR system (or successor thereto)
need not be furnished.
(g) Subject to the terms of this Agreement, Parent hereby consents to the use of such Prospectus and each amendment or
supplement thereto by each of the selling Holders in connection with the offering and sale of the Registrable Securities covered by such Prospectus and any amendment or supplement thereto, except after the giving of any notice pursuant to Section
3(d).
6
(h) Prior to any resale of Registrable Securities by a Holder, use its commercially reasonable efforts to register or
qualify or cooperate with the selling Holders in connection with the registration or qualification (or exemption from the registration or qualification) of such Registrable Securities for the resale by the Holder under the securities or Blue Sky laws
of such jurisdictions within the United States as any Holder reasonably requests in writing, to keep each registration or qualification (or exemption therefrom) effective during the Effectiveness Period and to do any and all other acts or things
reasonably necessary to enable the disposition in such jurisdictions of the Registrable Securities covered by each Registration Statement, provided that Parent shall not be required to qualify generally to do business in any jurisdiction where it is
not then so qualified, subject Parent to any material tax in any such jurisdiction where it is not then so subject or file a general consent to service of process in any such jurisdiction.
(i) If requested by a Holder, promptly (and in any event within two (2) Trading Days of such request) deliver to Holder
certificates or book entry statements, as applicable, representing Registrable Securities to be delivered to a transferee pursuant to an effective Registration Statement, which certificates shall be free of all restrictive legends (and have its
counsel provide such opinions that such restrictive legends need not appear on such certificates as may be reasonably requested), and to enable such Registrable Securities to be in such denominations and registered in the name of the transferee as
such Holder may reasonably request. If requested by a Holder other than in connection with a transfer pursuant to an effective registration statement, promptly (and in any event within two (2) Trading Days of such request) deliver to Holder
certificates or book entry statements, as applicable, representing Registrable Securities to be delivered to a transferee, which certificates shall be free of all restrictive legends, and to enable such Registrable Securities to be in such
denominations and registered in such names as any such Holder may reasonably request; provided that Holder timely furnishes to Company a completed Holder Representation Letter in substantially the form attached hereto as Annex D and such
other customary representations as may be reasonably required, in accordance with applicable Law, in connection therewith.
(j) Upon the occurrence of any event contemplated by Section 3(d)(iii) through (v),
as promptly as reasonably possible under the circumstances taking into account Parent’s good faith determination of any adverse consequences to Parent and its stockholders of the premature disclosure of such event, prepare a supplement or amendment,
including a post-effective amendment, to a Registration Statement or a supplement to the related Prospectus or any document incorporated or deemed to be incorporated therein by reference, and file any other required document so that, as thereafter
delivered, neither a Registration Statement nor such Prospectus will contain an untrue statement of a material fact or omit to state a material fact required to be stated therein or necessary to make the statements therein, in light of the
circumstances under which they were made, not misleading. If Parent notifies the Holders in accordance with clauses (iii) through (v) of Section 3(d) above to suspend the use of any Prospectus until the requisite changes to such Prospectus
have been made, then the Holders shall suspend use of such Prospectus; provided that Parent shall only be entitled to exercise its right under this Section 3(j) to suspend the availability of a Registration Statement and Prospectus up to two
(2) occasions in any twelve (12)-month period for a period not to exceed forty-five (45) consecutive days or a total of ninety (90) calendar days, in each case in any such twelve (12)-month period. Parent will use its reasonable best efforts to
ensure that the use of the Prospectus may be resumed as promptly as is reasonably practicable.
7
(k) Otherwise use commercially reasonable efforts to comply with all applicable rules and regulations of the Commission
under the Securities Act and the Exchange Act, including, without limitation, Rule 172 under the Securities Act, file any final Prospectus, including any supplement or amendment thereof, with the Commission pursuant to Rule 424 under the Securities
Act, promptly inform the Holders in writing if, at any time during the Effectiveness Period, Parent does not satisfy the conditions specified in Rule 172 and, as a result thereof, the Holders are required to deliver a Prospectus in connection with
any disposition of Registrable Securities and take such other actions as may be reasonably necessary to facilitate the registration of the Registrable Securities hereunder.
(l) Parent shall use its commercially reasonable efforts to maintain eligibility for use of Form S-3 (or any successor
form thereto) for the registration of the resale of the Registrable Securities once eligible to use such form.
(m) Parent may require each selling Holder to furnish to Parent a certified statement as to the number of shares of
Common Stock beneficially owned by such Holder and, if required by the Commission, the natural persons thereof that have voting and dispositive control over the shares.
(n) Parent shall use its reasonable best efforts to cause all Registrable Securities to be listed on each securities
exchange or market, if any, on which the shares of Parent common stock are listed.
(o) Parent shall, at its sole expense, upon appropriate notice from a Holder stating that Registrable Securities have
been sold or transferred pursuant to an effective Registration Statement, promptly (and in any event within two (2) Trading Days of such notice) prepare and deliver certificates or evidence of book-entry positions representing the Registrable
Securities to be delivered to a transferee pursuant to such Registration Statement, which certificates or book-entry positions shall be free of any restrictive legends and in such denominations and registered in such names as the undersigned may
request.
(p) Neither Parent nor any subsidiary or affiliate thereof shall identify any Holder as an underwriter in any public
disclosure or filing with the Commission or any trading market; provided, that if the Commission requires that a Holder be identified as a statutory underwriter in a Registration Statement, such Holder will have the option, in its sole and absolute
discretion, to either (i) withdraw from such Registration Statement upon its prompt written request to Parent, in which case Parent’s obligation to register such Holder’s Registrable Securities will be deemed satisfied or (ii) be included as such in
such Registration Statement; provided, further, that the foregoing shall not prohibit Parent from including the disclosure found in the “Plan of Distribution” section attached hereto as Exhibit A in the
Registration Statement.
(q) Once a Registration Statement covering the resale of the Registrable Securities is declared effective, Parent shall
remove all restrictive legends on Merger Shares, and Parent shall, upon request of Parent Holder or the transfer agent, provide a blanket opinion of counsel permitting such removal. Further, Parent shall remove all restrictive legends, (i) following
any sale of Merger Shares pursuant to Rule 144 or any other applicable exemption from the registration requirements of the Securities Act, or (ii) if such Merger Shares are eligible for resale under Rule 144(b)(1) or any successor provision. Without
limiting the foregoing, upon request of Parent Holder, subject to receipt by Parent of an opinion of counsel reasonably satisfactory to Parent to the effect that such legend is no longer required under the Securities Act, Parent shall promptly cause
the legend to be removed from any book-entry statements for any Merger Shares in accordance with the terms of this Agreement and deliver, or cause to be delivered, to any Company Holder new book-entry statements representing the Merger Shares that
are free from all restrictive and other legends or, at the request of such Company Holder, via DWAC transfer to such Company Holder’s account.
8
(a) If, at any time prior to the Initial Registration Statement being declared effective by the Commission, the Company shall determine to register any of its securities
either for its own account or the account of a security holder or holders exercising their respective demand registration rights, other than a registration pursuant to Section 2 hereof, a registration relating solely to employee benefit
plans, a registration relating to the offer and sale of debt securities, or a registration relating solely to a Rule 145 transaction, the Company shall:
|
i.
|
promptly give to each Holder written notice thereof; and
|
| ii. |
use commercially reasonable efforts to include in such registration (and any related qualification under blue sky laws or other compliance), except as set forth in Section 4(b), and in any underwriting involved therein, all the
Registrable Securities specified in a written request or requests, made by any Holder and received by the Company within ten (10) days after the written notice from the Company described in clause (i) above is given by the Company. Such
written request may specify all or a part of a Holder’s Registrable Securities.
|
(b) If the registration of which the Company gives notice is for a registered public offering involving an underwriting, the Company shall so advise the Holders as a part of
the written notice given pursuant to Section 4(a)(i). In such event, the right of any Holder to include Registrable Securities in such registration pursuant to this Section 4 shall be conditioned upon such Holder’s participation in
such underwriting and the inclusion of such Holder’s Registrable Securities in the underwriting to the extent provided herein. All Holders proposing to distribute their securities through such underwriting shall (together with the Company and any
Other Stockholders distributing their securities through such underwriting) enter into an underwriting agreement in customary form with the representative of the underwriter or underwriters selected by the Company.
(c) Notwithstanding any other provision of this Section 4, if the representative of the underwriters advises the Company that marketing factors require a limitation
on the number of securities sold other than by the Company, the representative may (subject to the limitations set forth below) exclude all Registrable Securities from, or limit the number of Registrable Securities to be included in, the registration
and underwriting. If such registration is with respect to any Company-initiated registered offering of the Company’s securities to the general public, the Company may limit, to the extent so advised by the underwriters, the amount of securities to
be included in the registration by the Company’s stockholders (including the Holders); provided, however, that the number of Registrable Securities to be included in
such registration by the Company’s stockholders (including the Holders) may not be so reduced to less than twenty-five percent (25%) of the total number of all securities included in such registration (provided that all Other Stockholders shall be
excluded first from such offering). The Company shall so advise all holders of securities requesting registration, and the number of securities that are entitled to be included in the registration and underwriting shall be allocated first to the
Company for securities being sold for its own account and thereafter as set forth in Section 4(e). If any person does not agree to the terms of any such underwriting, such person shall be excluded therefrom by written notice from the Company
or the underwriter. Any securities excluded or withdrawn from such underwriting shall be withdrawn from such registration. If securities are so withdrawn from the registration and if the number of securities to be included in such registration was
previously reduced as a result of marketing factors, the Company shall then offer to all persons who have retained the right to include securities in the registration the right to include additional securities in the registration in an aggregate
amount equal to the number of securities so withdrawn, with such securities to be allocated among the persons requesting additional inclusion in accordance with Section 4(e). To facilitate the allocation of securities in accordance with the
above provisions, the Company or the underwriter(s) may round the number of securities allocated to any Holder to the nearest 100 shares.
9
(d) Right to Terminate Registration. The Company shall have the right to terminate or withdraw any registration initiated by it under this Section 4 prior
to the effectiveness of such registration whether or not any Holder has elected to include securities in such registration.
(e) Allocation of Registration Opportunities. Except as otherwise provided in this Section 4, in any circumstance in which all of the Registrable Securities
and Other Shares requested to be included in a registration on behalf of the Holders or Other Stockholders cannot be so included as a result of limitations in the aggregate number of Registrable Securities and Other Shares that may be so included,
the number of Registrable Securities and Other Shares (if any) shall be excluded, first by excluding Other Shares, pro rata on the basis of the number of Other Shares held by such Other Stockholders, and thereafter by excluding Registrable
Securities, pro rata on the basis of the number of Registrable Securities held by such Holders, until the aggregate number of Registrable Securities and Other Shares (if any) may be included in such registration. If any Holder or Other Stockholder
does not request inclusion of the maximum number of Registrable Securities and Other Shares allocated to such person pursuant to the above described formula, the remaining portion of such person’s allocation shall be reallocated among those
requesting Holders and/or Other Stockholders whose allocations did not satisfy their requests, pro rata on the same basis as described above, and this procedure shall be repeated until all of the Registrable Securities and/or Other Shares that may be
included in such registration on behalf of the Holders and/or Other Stockholders have been so allocated. For purposes of the preceding sentence concerning apportionment, for any selling stockholder that is a Holder of Registrable Securities and that
is a venture capital fund, partnership or corporation, such Holder together with all partners, retired partners, members, retired members and other equity owners or Affiliates of such Holder, or the estates and Family Members of any such individuals
and any trusts for the benefit of any of the foregoing persons shall be deemed to be a single “selling Holder,” and any pro rata reduction with respect to such “selling Holder” shall be based upon the aggregate amount of Registrable Securities owned
by all such related entities and individuals.
5. Registration Expenses. All fees and expenses incident to the performance of or compliance with this Agreement by Parent shall be borne by Parent whether or not any Registrable
Securities are sold pursuant to a Registration Statement. The fees and expenses referred to in the foregoing sentence shall include, without limitation, (i) all registration and filing fees (including, without limitation, fees and expenses of
Parent’s counsel and independent registered public accountants) (A) with respect to filings made with the Commission, (B) with respect to filings required to be made with any National Exchange on which the Common Stock is then listed for trading, and
(C) in compliance with applicable state securities or Blue Sky laws reasonably agreed to by Parent in writing (including, without limitation, fees and disbursements of counsel for Parent in connection with Blue Sky qualifications or exemptions of the
Registrable Securities), (ii) printing expenses (including, without limitation, expenses of printing certificates for Registrable Securities), (iii) messenger, telephone and delivery expenses, (iv) fees and disbursements of counsel for Parent, (v)
Securities Act liability insurance, if Parent so desires such insurance, (vi) fees and expenses of all other Persons retained by Parent in connection with the consummation of the transactions contemplated by this Agreement, including Parent’s
transfer agent, and (vii) solely in connection with the review and filing of the initial Registration Statement, the reasonable fees and expenses, not to exceed $35,000, of one counsel for the selling Holders selected by the Holders of a majority of
the Registrable Securities to be registered. In addition, Parent shall be responsible for all of its internal expenses incurred in connection with the consummation of the transactions contemplated by this Agreement (including, without limitation,
all salaries and expenses of its officers and employees performing legal or accounting duties), the expense of any annual audit and the fees and expenses incurred in connection with the listing of the Registrable Securities on any securities exchange
as required hereunder. In no event shall Parent be responsible for any underwriting, broker or similar fees or commissions of any Holder or, except to the extent provided for in the Merger Agreement or this Agreement, any legal fees or other costs
of the Holders.
10
(a) Indemnification by Parent. Parent shall, notwithstanding any termination of
this Agreement, indemnify and hold harmless each Holder and its affiliates, the officers, directors, members, partners, agents, brokers (including brokers who offer and sell Registrable Securities as principal as a result of a pledge or any failure
to perform under a margin call of Common Stock), investment advisors and employees (and any other Persons with a functionally equivalent role of a Person holding such titles, notwithstanding a lack of such title or any other title) of each of them,
each Person who controls any such Holder (within the meaning of Section 15 of the Securities Act or Section 20 of the Exchange Act) and the officers, directors, members, stockholders, partners, agents and employees (and any other
Persons with a functionally equivalent role of a Person holding such titles, notwithstanding a lack of such title or any other title) of each such controlling Person, to the fullest extent permitted by applicable law, from and against any and all
losses, claims, damages, liabilities, costs (including, without limitation, reasonable and documented attorneys’ fees) and expenses (collectively, “Losses”), as incurred, arising out of or based solely upon (1)
any untrue or alleged untrue statement of a material fact contained in a Registration Statement, any Prospectus or any form of prospectus or in any amendment or supplement thereto or in any preliminary prospectus, or arising out of or relating to any
omission or alleged omission of a material fact required to be stated therein or necessary to make the statements therein (in the case of any Prospectus or supplement thereto, in light of the circumstances under which they were made) not misleading
or (2) any violation or alleged violation by Parent of the Securities Act, the Exchange Act or any state securities law, or any rule or regulation thereunder, in connection with the performance of its obligations under this Agreement, except to the
extent, but only to the extent, that (i) such untrue statements or omissions are based solely upon information regarding such Holder furnished in writing to Parent by such Holder expressly for use therein, or to the extent that such information
relates to such Holder or such Holder’s proposed method of distribution of Registrable Securities and was reviewed and expressly approved in writing by such Holder expressly for use in a Registration Statement, such Prospectus or in any amendment or
supplement thereto (it being understood that the Holder has approved Annex A hereto for this purpose) or (ii) in the case of an occurrence of an event of the type specified in Section 3(d)(iii)-(v), the use by such Holder of an
outdated, defective or otherwise unavailable Prospectus after Parent has notified such Holder in writing that the Prospectus is outdated, defective or otherwise unavailable for use by such Holder and prior to the receipt by such Holder of the Advice
contemplated in Section 7(c). Parent shall notify the Holders promptly of the institution, threat or assertion of any Proceeding arising from or in connection with the transactions contemplated by this Agreement of which Parent is aware.
Such indemnity shall remain in full force and effect regardless of any investigation made by or on behalf of such indemnified person and shall survive the transfer of any Registrable Securities by any of the Holders in accordance with Section
7(f).
11
(b) Indemnification by Holders. Each Holder shall, severally and not jointly,
indemnify and hold harmless Parent, its directors, officers, agents and employees, each Person who controls Parent (within the meaning of Section 15 of the Securities Act and Section 20 of the Exchange Act), and the directors,
officers, agents or employees of such controlling Persons, to the fullest extent permitted by applicable law, from and against all Losses, as incurred, to the extent arising out of or based solely upon any untrue or alleged untrue statement of a
material fact contained in any Registration Statement, any Prospectus, or in any amendment or supplement thereto or in any preliminary prospectus, or arising out of or relating to any omission or alleged omission of a material fact required to be
stated therein or necessary to make the statements therein (in the case of any Prospectus or supplement thereto, in the light of the circumstances under which they were made) not misleading to the extent, but only to the extent, that such untrue
statement or omission is contained in any information so furnished in writing by such Holder to Parent expressly for inclusion in such Registration Statement or such Prospectus, including information provided in the Selling Stockholder Questionnaire
or regarding the proposed method of distribution of Registrable Securities that was reviewed and expressly approved in writing by such Holder expressly for use in a Registration Statement (it being understood that the Holder has approved Annex A
hereto for this purpose), such Prospectus or in any amendment or supplement thereto. In no event shall the liability of a selling Holder be greater in amount than the dollar amount of the proceeds (net of all expenses paid by such Holder in
connection with any claim relating to this Section 5 and the amount of any damages such Holder has otherwise been required to pay by reason of such untrue statement or omission) received by such Holder upon the sale of the Registrable
Securities included in the Registration Statement giving rise to such indemnification obligation.
(c) Conduct of Indemnification Proceedings. If any Proceeding shall be brought or
asserted against any Person entitled to indemnity hereunder (an “Indemnified Party”), such Indemnified Party shall promptly notify the Person from whom indemnity is sought (the “Indemnifying
Party”) in writing, and the Indemnifying Party shall have the right to assume the defense thereof, including the employment of counsel reasonably satisfactory to the Indemnified Party and the payment of all reasonable fees and expenses
incurred in connection with defense thereof, provided that the failure of any Indemnified Party to give such notice shall not relieve the Indemnifying Party of its obligations or liabilities pursuant to this Agreement, except (and only) to the extent
that it shall be finally determined by a court of competent jurisdiction (which determination is not subject to appeal or further review) that such failure shall have materially and adversely prejudiced the Indemnifying Party.
An Indemnified Party shall have the right to employ separate counsel in any such Proceeding and to participate in the defense thereof, but the fees and expenses of such counsel
shall be at the expense of such Indemnified Party or Parties unless: (1) the Indemnifying Party has agreed in writing to pay such fees and expenses, (2) the Indemnifying Party shall have failed promptly to assume the defense of such Proceeding and
to employ counsel reasonably satisfactory to such Indemnified Party in any such Proceeding, or (3) the named parties to any such Proceeding (including any impleaded parties) include both such Indemnified Party and the Indemnifying Party, and counsel
to the Indemnified Party shall reasonably believe that a material conflict of interest is likely to exist if the same counsel were to represent such Indemnified Party and the Indemnifying Party (in which case, if such Indemnified Party notifies the
Indemnifying Party in writing that it elects to employ separate counsel at the expense of the Indemnifying Party, the Indemnifying Party shall not have the right to assume the defense thereof and the reasonable fees and expenses of no more than one
separate counsel shall be at the expense of the Indemnifying Party). Notwithstanding anything in this Section 6, the Indemnifying Party shall not be liable for any settlement of any such Proceeding effected without its written consent, which
consent shall not be unreasonably withheld or delayed. No Indemnifying Party shall, without the prior written consent of the Indemnified Party, effect any settlement of any pending Proceeding in respect of which any Indemnified Party is a party,
unless such settlement includes an unconditional release of such Indemnified Party from all liability on claims that are the subject matter of such Proceeding.
12
Subject to the terms of this Agreement, all reasonable and documented fees and expenses of the Indemnified Party (including reasonable and documented fees and expenses to the
extent incurred in connection with investigating or preparing to defend such Proceeding in a manner not inconsistent with this Section 6) shall be paid to the Indemnified Party, as incurred, within ten (10) Trading Days of written notice
thereof to the Indemnifying Party, provided that the Indemnified Party shall promptly reimburse the Indemnifying Party for that portion of such fees and expenses applicable to such actions for which such Indemnified Party is finally determined by a
court of competent jurisdiction (which determination is not subject to appeal or further review) not to be entitled to indemnification hereunder.
(d) Contribution. If the indemnification under Section 6(a) or 6(b)
is unavailable to an Indemnified Party or insufficient to hold an Indemnified Party harmless for any Losses, then each Indemnifying Party shall contribute to the amount paid or payable by such Indemnified Party, in such proportion as is appropriate
to reflect the relative fault of the Indemnifying Party and Indemnified Party in connection with the actions, statements or omissions that resulted in such Losses as well as any other relevant equitable considerations. The relative fault of such
Indemnifying Party and Indemnified Party shall be determined by reference to, among other things, whether any action in question, including any untrue or alleged untrue statement of a material fact or omission or alleged omission of a material fact,
has been taken or made by, or relates to information supplied by, such Indemnifying Party or Indemnified Party, and the parties’ relative intent, knowledge, access to information and opportunity to correct or prevent such action, statement or
omission; provided, however, that no Person guilty of fraudulent misrepresentation (within the meaning of Section 11(f) of the Securities Act) will be entitled to contribution from any Person who was not guilty of such fraudulent
misrepresentation. The amount paid or payable by a party as a result of any Losses shall be deemed to include, subject to the limitations set forth in this Agreement, any reasonable attorneys’ or other fees or expenses incurred by such party in
connection with any Proceeding to the extent such party would have been indemnified for such fees or expenses if the indemnification provided for in this Section 6 was available to such party in accordance with its terms.
The parties hereto agree that it would not be just and equitable if contribution pursuant to this Section 6(d) were determined by pro rata allocation or by any other method
of allocation that does not take into account the equitable considerations referred to in the immediately preceding paragraph. In no event shall the contribution obligation of a Holder of Registrable Securities be greater in amount than the dollar
amount of the proceeds (net of all expenses paid by such Holder in connection with any claim relating to this Section 6 and the amount of any damages such Holder has otherwise been required to pay by reason of such untrue or alleged untrue
statement or omission or alleged omission) received by it upon the sale of the Registrable Securities giving rise to such contribution obligation.
The indemnity and contribution agreements contained in this Section 6(d) are in addition to any liability that the Indemnifying Parties may have to the Indemnified Parties.
(a) Remedies. In the event of a breach by Parent or by a Holder of any of their respective obligations under
this Agreement, each Holder or Parent, as the case may be, in addition to being entitled to exercise all rights granted by law and under this Agreement, including recovery of damages, shall be entitled to specific performance of its rights under this
Agreement. Each of Parent and each Holder agrees that monetary damages would not provide adequate compensation for any losses incurred by reason of a breach by it of any of the provisions of this Agreement and hereby further agrees that, in the
event of any action for specific performance in respect of such breach, it shall not assert or shall waive the defense that a remedy at law would be adequate.
13
(b) No Piggyback on Registrations; Prohibition on Filing Other Registration Statements.
Except as provided in Section 4, neither Parent nor any of its security holders (other than the Holders in such capacity pursuant hereto) may include securities of Parent in any Registration Statements other than the Registrable Securities. Parent
shall not file any other registration statements until all Registrable Securities are registered pursuant to a Registration Statement that is declared effective by the Commission, provided that Section 4 and this Section 7(b) shall not
prohibit Parent from filing amendments to registration statements filed prior to the date of this Agreement so long as no new securities are registered on any such existing registration statements, nor preparing and filing with the Commission a
registration statements on Form S-8 relating to its equity incentive plans.
(c) Discontinued Disposition. By its acquisition of Registrable Securities, each
Holder agrees that, upon receipt of a notice from Parent of the occurrence of any event of the kind described in Section 3(d)(iii) through (v), such Holder will promptly discontinue disposition of such Registrable Securities under a
Registration Statement until it is advised in writing (the “Advice”) by Parent that the use of the applicable Prospectus (as it may have been supplemented or amended) may be resumed. Parent will use its
commercially reasonable efforts to ensure that the use of the Prospectus may be resumed as promptly as is practicable.
(d) Amendments and Waivers. The provisions of this Agreement, including the
provisions of this sentence, may not be amended, modified or supplemented, and waivers or consents to departures from the provisions hereof may not be given, unless the same shall be in writing and signed by Parent and the Required Holders, provided
that, (i) if any amendment, modification or waiver disproportionately and adversely impacts a Holder (or group of Holders), the consent of such disproportionately impacted Holder (or group of Holders) shall be required, including any amendment or
modification of Section 6, and (ii) any amendment, modification or waiver of Section 6 shall require the consent of each Holder affected by such amendment, modification or waiver. If a Registration Statement does not register all of
the Registrable Securities pursuant to a waiver or amendment done in compliance with the previous sentence, then the number of Registrable Securities to be registered for each Holder shall be reduced pro rata among all Holders and each Holder shall
have the right to designate which of its Registrable Securities shall be omitted from such Registration Statement. Notwithstanding the foregoing, a waiver or consent to depart from the provisions hereof with respect to a matter that relates
exclusively to the rights of a Holder or some Holders and that does not directly or indirectly affect the rights of other Holders may be given only by such Holder or Holders of all of the Registrable Securities to which such waiver or consent
relates; provided, however, that the provisions of this sentence may not be amended, modified, or supplemented except in accordance with the provisions of the first sentence of this Section 6(d). No consideration shall be
offered or paid to any Person to amend or consent to a waiver or modification of any provision of this Agreement unless the same consideration also is offered to all of the parties to this Agreement. As used herein, “Required
Holders” means Holders of 50.1% or more of the then outstanding Registrable Securities (for purposes of clarification, this includes any securities issuable upon conversion or exercise of any Registrable Security).
(e) Notices. Any and all notices or other communications or deliveries required or permitted to be provided hereunder shall be in writing and shall be deemed to
have been duly delivered and received hereunder (a) one (1) Business Day after being sent for next Business Day delivery, fees prepaid, via a reputable international overnight courier service, (b)
upon delivery in the case of delivery by hand or (c) on the date delivered in the place of delivery if sent by email or facsimile (with a written or electronic confirmation of delivery) prior to 6:00 p.m. (New York City
time), otherwise on the next succeeding Business Day, in each case to the intended recipient as set forth below:
14
if to Parent:
Gyre Therapeutics, Inc.
12770 High Bluff Drive, Suite 150
San Deigo, CA 92130
Attention: Thomas Eastling
with a copy to (which shall not constitute notice):
Gibson, Dunn & Crutcher LLP
One Embarcadero Center, Suite 2600
San Francisco, CA 94111
Attention: Ryan Murr, Branden Berns, Evan Shepherd
if to the Company:
Cullgen Inc.
12730 High Bluff Drive, Suite 250
San Diego, CA 92130
Attention: Thomas Eastling
with a copy to (which shall not constitute notice):
Mintz, Levin, Cohn, Ferris, Glovsky & Popeo, P.C.
3580 Carmel Mountain Road
Suite 300
San Diego, CA 92130
Attention: Scott Stanton, Jason McCaffrey
If to a Holder, to the address or email addresses of such Holder as it appears on such Holder’s signature page attached hereto or such other address as may be designated in writing by such Holder.
(f) Successors and Assigns. This Agreement shall inure to the benefit of and be binding upon the successors and permitted assigns of each of the parties and shall
inure to the benefit of each Holder. Parent may not assign (except by merger) its rights or obligations hereunder without the prior written consent of the Required Holders. None of the Holders may assign its rights or obligations hereof without the
prior written consent of Parent; provided, however, that a Holder may assign its rights (in whole or in part) hereunder without Parent consent to (i) any Affiliate, (ii) any fund under common control, (iii) any bona fide transferee of the Registrable
Securities or (iv) in connection with any estate planning, provided such transferee agrees in writing to be bound hereby.
15
(g) No Inconsistent Agreements. Neither Parent nor any of its subsidiaries has entered, as of the date hereof, nor shall Parent or any of its subsidiaries, on or
after the date of this Agreement, enter into any agreement with respect to its securities, that would have the effect of impairing the rights granted to the Holders in this Agreement or otherwise conflicts with the provisions hereof. Neither Parent
nor any of its subsidiaries has previously entered into any agreement granting any registration rights with respect to any of its securities to any Person that have not been satisfied in full.
(h) Execution and Counterparts. This Agreement may be executed in two (2) or more counterparts, all of which when taken together shall be considered one and the
same agreement and shall become effective when counterparts have been signed by each party and delivered to the other party, it being understood that both parties need not sign the same counterpart. In the event that any signature is delivered by
facsimile transmission or by e-mail delivery of a “.pdf” format data file, such signature shall create a valid and binding obligation of the party executing (or on whose behalf such signature is executed) with the same force and effect as if such
facsimile or “.pdf” signature page was an original thereof.
(i) Governing Law. All questions concerning the construction, validity, enforcement and interpretation of this Agreement shall be determined in accordance with the
provisions of the Merger Agreement and Section 11.5 of the Merger Agreement is hereby incorporated herein mutatis mutandi.
(j) Cumulative Remedies. The remedies provided herein are cumulative and not exclusive of any other remedies provided by law.
(k) Severability. If any term, provision, covenant or restriction of this Agreement is held by a court of competent jurisdiction to be invalid, illegal, void or
unenforceable, the remainder of the terms, provisions, covenants and restrictions set forth herein shall remain in full force and effect and shall in no way be affected, impaired or invalidated, and the parties hereto shall use their commercially
reasonable efforts to find and employ an alternative means to achieve the same or substantially the same result as that contemplated by such term, provision, covenant or restriction. It is hereby stipulated and declared to be the intention of the
parties that they would have executed the remaining terms, provisions, covenants and restrictions without including any of such that may be hereafter declared invalid, illegal, void or unenforceable.
(l) Headings. The headings in this Agreement are for convenience only, do not constitute a part of the Agreement and shall not be deemed to limit or affect any of
the provisions hereof.
(m) Independent Nature of Holders’ Obligations and Rights. The obligations of each Holder hereunder are several and not joint with the obligations of any other
Holder hereunder, and no Holder shall be responsible in any way for the performance of the obligations of any other Holder hereunder. Nothing contained herein or in any other agreement or document delivered at any closing, and no action taken by any
Holder pursuant hereto or thereto, shall be deemed to constitute the Holders as a partnership, an association, a joint venture or any other kind of group or entity, or create a presumption that the Holders are in any way acting in concert or as a
group or entity with respect to such obligations or the transactions contemplated by this Agreement or any other matters, and Parent acknowledges that the Holders are not acting in concert or as a group, and Parent shall not assert any such claim,
with respect to such obligations or transactions. Each Holder shall be entitled to protect and enforce its rights, including without limitation the rights arising out of this Agreement, and it shall not be necessary for any other Holder to be joined
as an additional party in any proceeding for such purpose. The use of a single agreement with respect to the obligations of Parent contained was solely in the control of Parent, not the action or decision of any Holder, and was done solely for the
convenience of Parent and not because it was required or requested to do so by any Holder. It is expressly understood and agreed that each provision contained in this Agreement is between Parent and a Holder, solely, and not between Parent and the
Holders collectively and not between and among Holders.
********************
(Signature Pages Follow)
16
IN WITNESS WHEREOF, the parties have executed this Registration Rights Agreement as of the date first written above.
|
CULLGEN INC.
|
||
|
By:
|
||
|
Name:
|
||
|
Title:
|
||
[Signature Page to Registration Rights Agreement]
|
GYRE THERAPEUTICS, INC.
|
||
|
By:
|
||
|
Name:
|
||
|
Title:
|
||
[SIGNATURE PAGE OF HOLDERS FOLLOWS]
[Signature Page to Registration Rights Agreement]
[SIGNATURE PAGE OF HOLDERS TO RRA]
IN WITNESS WHEREOF, the parties have executed this Registration Rights Agreement as of the date first written above.
|
[●]
|
||
|
By:
|
||
|
Name:
|
||
|
Title:
|
||
|
Address:
|
[______]
|
|
Email:
|
[______]
|
|
Attention:
|
[______]
|
[Signature Page to Registration Rights Agreement]
Plan of Distribution
Each Selling Stockholder (the “Selling Stockholders”) of the securities and any of their pledgees, assignees, donees, transferees or other
successors-in-interest may, from time to time, sell, transfer or otherwise dispose of any or all of their securities covered hereby on the principal trading market or any other stock exchange, market or trading facility on which the securities are
traded or in private transactions. These sales may be at fixed or negotiated prices. A Selling Stockholder may use any one or more of the following methods when selling securities:
|
•
|
ordinary brokerage transactions and transactions in which the broker-dealer solicits purchasers;
|
|
•
|
block trades in which the broker-dealer will attempt to sell the securities as agent but may position and resell a portion of the block as principal to facilitate the transaction;
|
|
•
|
purchases by a broker-dealer as principal and resale by the broker-dealer for its account;
|
|
•
|
an exchange distribution in accordance with the rules of the applicable exchange;
|
|
•
|
privately negotiated transactions;
|
|
•
|
settlement of short sales;
|
|
•
|
in transactions through broker-dealers that agree with the Selling Stockholders to sell a specified number of such securities at a stipulated price per security;
|
|
•
|
through the writing or settlement of options or other hedging transactions, whether through an options exchange or otherwise;
|
|
•
|
in a distribution to members, partners, stockholders or other equityholders of the Selling Stockholders;
|
|
•
|
a combination of any such methods of sale or distribution; or
|
|
•
|
any other method permitted pursuant to applicable law.
|
The Selling Stockholders may also sell securities under Rule 144 or any other exemption from registration under the Securities Act of 1933, as amended (the “Securities Act”), if available, rather than under this prospectus.
Broker-dealers engaged by the Selling Stockholders may arrange for other brokers-dealers to participate in sales. Broker-dealers may receive commissions or discounts from the Selling Stockholders
(or, if any broker-dealer acts as agent for the purchaser of securities, from the purchaser) in amounts to be negotiated, but, except as set forth in a supplement to this Prospectus, in the case of an agency transaction not in excess of a customary
brokerage commission in compliance with FINRA Rule 2121; and in the case of a principal transaction a markup or markdown in compliance with FINRA Rule 2121.
Annex A-1
In connection with the sale of the securities or interests therein, the Selling Stockholders may enter into hedging transactions with broker-dealers or other financial institutions, which may in turn
engage in short sales of the securities in the course of hedging the positions they assume. The Selling Stockholders may also sell securities short and deliver these securities to close out their short positions, or loan or pledge the securities to
broker-dealers that in turn may sell these securities. The Selling Stockholders may also enter into option or other transactions with broker-dealers or other financial institutions or create one or more derivative securities that require the
delivery to such broker-dealer or other financial institution of securities offered by this prospectus, which securities such broker-dealer or other financial institution may resell pursuant to this prospectus (as supplemented or amended to reflect
such transaction). The Selling Stockholders also may transfer the securities in other circumstances, in which case the transferees, pledgees, donees or other successors in interest will be the selling beneficial owners for purposes of this
prospectus.
The Selling Stockholders and any broker-dealers or agents that are involved in selling the securities may be deemed to be “underwriters” within the meaning of the Securities Act in connection with
such sales. In such event, any commissions received by such broker-dealers or agents and any profit on the resale of the securities purchased by them may be deemed to be underwriting commissions or discounts under the Securities Act. Each Selling
Stockholder has informed us that it does not have any written or oral agreement or understanding, directly or indirectly, with any person to distribute the securities.
We are required to pay certain fees and expenses incurred by us incident to the registration of the securities. We have agreed to indemnify the Selling Stockholders against certain losses, claims,
damages and liabilities, including liabilities under the Securities Act.
We agreed to keep this prospectus effective until the earlier of the date that the securities (i) have been sold, pursuant to this prospectus or pursuant to Rule 144, or
(ii) the date on which the securities may be resold by the Selling Stockholders without registration and without regard to any volume or manner-of-sale limitations by reason of Rule 144, and without the requirement for us to be in compliance with the
current public information under Rule 144 under the Securities Act or any other rule of similar effect, or. The resale securities will be sold only through registered or licensed brokers or dealers if required under applicable state securities
laws. In addition, in certain states, the resale securities covered hereby may not be sold unless they have been registered or qualified for sale in the applicable state or an exemption from the registration or qualification requirement is available
and is complied with.
Under applicable rules and regulations under the Exchange Act, any person engaged in the distribution of the resale securities may not simultaneously engage in market making activities with respect
to the common stock for the applicable restricted period, as defined in Regulation M, prior to the commencement of the distribution. In addition, the Selling Stockholders will be subject to applicable provisions of the Exchange Act and the rules and
regulations thereunder, including Regulation M, which may limit the timing of purchases and sales of the common stock by the Selling Stockholders or any other person. We will make copies of this prospectus available to the Selling Stockholders and
have informed them of the need to deliver a copy of this prospectus to each purchaser at or prior to the time of the sale (including by compliance with Rule 172 under the Securities Act).
Annex A-2
SELLING STOCKHOLDERS
We are registering the shares of common stock in order to permit the selling stockholders to offer the shares for resale from time to time.
The table below lists the selling stockholders and other information regarding the beneficial ownership of the shares of common stock by each of the selling stockholders. The second column lists the
number of shares of common stock beneficially owned by each selling stockholder, based on its ownership of the shares of common stock, as of [•], 2026.
The third column lists the shares of common stock being offered by this prospectus by the selling stockholders.
The fourth column reflects the number of shares of common stock beneficially owned by each selling stockholder, assuming the sale of all of the shares offered by the selling stockholders pursuant to
this prospectus.
The selling stockholders may sell all, some or none of their shares in this offering. See “Plan of Distribution.”
|
Name of Selling Stockholder
|
Number of Shares
of Common Stock
Owned Prior to
Offering
|
Maximum Number
of Shares of
Common Stock to
be Sold Pursuant to
this Prospectus
|
Number of Shares
of Common Stock
Owned After
Offering
|
|||
Annex B-1
Selling Stockholder Notice and Questionnaire
The undersigned owner of Registrable Securities (as such term is defined in the Registration Rights Agreement) of Cullgen Inc., a Delaware corporation (the “Company”), understands that Parent has filed or intends to file with the Securities and Exchange Commission (the “Commission”) a Registration Statement for the
registration and resale under Rule 415 of the Securities Act of 1933, as amended (the “Securities Act”), of the Registrable Securities, in accordance with the terms of the Registration
Rights Agreement dated as of [•], 2026 to which Parent and the undersigned are parties (the “Registration Rights Agreement”). A copy of the Registration Rights Agreement is available from
Parent upon request at the address set forth below. All capitalized terms not otherwise defined herein shall have the meanings ascribed thereto in the Registration Rights Agreement.
Certain legal consequences arise from being named as a selling stockholder in the Registration Statement and the related prospectus. Accordingly, holders and beneficial owners of Registrable
Securities are advised to consult their own securities law counsel regarding the consequences of being named or not being named as a selling stockholder in the Registration Statement and the related prospectus.
NOTICE
The undersigned beneficial owner (the “Selling Stockholder”) of Registrable Securities hereby elects to include the Registrable Securities
owned by it in the Registration Statement.
The undersigned hereby provides the following information to Parent and represents and warrants that such information is accurate:
QUESTIONNAIRE
1. Name.
| (a) |
Full Legal Name of Selling Stockholder
|
| (b) |
| (c) |
Full Legal Name of Natural Control Person (which means a natural person who directly or indirectly alone or with others has power to vote or dispose of the securities covered by this Questionnaire):
|
2. Address for Notices to Selling Stockholder:
|
Telephone:
|
| Fax: |
| Contact Person: |
Annex C-1
3. Broker-Dealer Status:
| (a) |
Are you a broker-dealer?
|
Yes ☐ No ☐
| (b) |
If “yes” to Section 3(a), did you receive your Registrable Securities as compensation for investment banking services to Parent?
|
Yes ☐ No ☐
| Note: |
If “no” to Section 3(b), the Commission’s staff has indicated that you should be identified as an underwriter in the Registration Statement.
|
| (c) |
Are you an affiliate of a broker-dealer?
|
Yes ☐ No ☐
Yes ☐ No ☐
| Note: |
If “no” to Section 3(d), the Commission’s staff has indicated that you should be identified as an underwriter in the Registration Statement.
|
4. Ownership of Securities of Parent Owned by the Selling Stockholder.
Except as set forth below in this Item 4, the undersigned is not the beneficial or registered owner of any securities of Parent other than the securities issuable pursuant to the
Merger Agreement.
| (a) |
Type and Amount of other Company securities owned by the Selling Stockholder (including beneficially owned, as applicable):
|
5. Relationships with Parent:
Except as set forth below, neither the undersigned nor any of its affiliates, officers, directors or principal equity holders (owners of 5% of more of the equity securities of the
undersigned) has held any position or office or has had any other material relationship with Parent (or its predecessors or affiliates) during the past three years.
State any exceptions here:
Annex C-2
The undersigned agrees to promptly notify Parent of any material inaccuracies or changes in the information provided herein that may occur subsequent to the date hereof at any time while the
Registration Statement remains effective; provided, that the undersigned shall not be required to notify Parent of any changes to the number of securities held or owned by the undersigned or its affiliates.
By signing below, the undersigned consents to the disclosure of the information contained herein in its answers to Items 1 through 5 and the inclusion of such information in the Registration
Statement and the related prospectus and any amendments or supplements thereto. The undersigned understands that such information will be relied upon by Parent in connection with the preparation or amendment of the Registration Statement and the
related prospectus and any amendments or supplements thereto.
IN WITNESS WHEREOF the undersigned, by authority duly given, has caused this Notice and Questionnaire to be executed and delivered either in person or by its duly authorized agent.
|
Date:
|
Beneficial Owner:
|
|
By:
|
||
| Name: | ||
| Title: | ||
PLEASE FAX A COPY (OR EMAIL A .PDF COPY) OF THE COMPLETED AND EXECUTED NOTICE AND QUESTIONNAIRE TO:
Annex C-3
HOLDER REPRESENTATION LETTER
________ __, 20__
Cullgen Inc.
[__]
[__]
[__]To Whom It May Concern:
The undersigned (the “Holder”) hereby requests that the federal securities law restrictive legend be removed from the book entries representing _________ of
shares (the “Shares”) of common stock, par value $0.0001 per share (the “Common Stock”), of Gyre Therapeutics, Inc. (the “Company”). In connection with the legend removal, Holder hereby represents to, and agrees with, you as follows:
|
1.
|
The Shares are owned of record and beneficially by Holder.
|
|
4.
|
Holder (or, in the case of individuals, Holder’s employer) has in place internal policies and procedures reasonably designed to monitor and ensure that no offer, sale or transfer of, or other transaction
involving, the Shares is made in violation of the foregoing restrictions, and Holder will monitor all transactions involving the Shares for the purpose of ensuring that they comply with all federal and state securities laws.
|
|
5.
|
Holder is familiar with the requirements for effecting resales or transfers of, or other transactions involving, the Shares in compliance with federal and state securities laws and acknowledges and agrees
that the Company and legal counsel to the Company (together, the “Indemnified Parties”) are relying on Holder’s representations and agreements in this letter. Holder will
indemnify and hold harmless the Indemnified Parties against any and all loss, damage, claim, liability and expense arising out of or resulting from the breach of any such representation or agreement.
|
Annex D-1
|
Very truly yours,
|
|||
|
[HOLDER]
|
|||
|
By:
|
|||
|
Name:
|
|||
|
Title:
|
|||
Annex D-2
Exhibit 10.4
Confidential
FORM OF LOCK-UP AGREEMENT
March 2, 2026
Cullgen Inc.
12730 High Bluff Drive, Suite 250
San Diego, CA 92130
Ladies and Gentlemen:
The undersigned signatory of this lock-up agreement (this “Lock-Up Agreement’’) understands that Gyre Therapeutics, Inc., a Delaware corporation (“Parent”), is entering into an Agreement
and Plan of Merger and Reorganization, dated as of March 2, 2026 (as the same may be amended from time to time, the “Merger Agreement’’) with Helix Merger Sub Corp., a Delaware corporation and a wholly owned subsidiary of Parent, and
Cullgen Inc., a Delaware corporation (the “Company”). Capitalized terms used but not otherwise defined herein shall have the respective meanings ascribed to such terms in the Merger Agreement.
As a condition and inducement to each of the parties to enter into the Merger Agreement and to consummate the transactions contemplated thereby, and for other good and valuable
consideration, the receipt and sufficiency of which is hereby acknowledged, the undersigned hereby irrevocably agrees that, subject to the exceptions set forth herein, without the prior written consent of Parent, the undersigned will not, during
the period commencing upon the Closing and (w) with respect to one-third of the Undersigned’s Shares, ending on the date that is 180 days after the Closing Date, (x) with respect to one-third of the Undersigned’s Shares, ending on the date that is twelve (12) months after the Closing Date, and (y) with respect to one-third of the Undersigned’s Shares, ending on the date that is eighteen (18)
months after the Closing Date (each such period the “Restricted Period”):
(1) offer, pledge, sell, contract to sell, sell any option or contract to purchase, purchase any option or contract to sell, grant any option, right or warrant to purchase, or otherwise transfer
or dispose of, directly or indirectly, any shares of Parent Common Stock or any securities convertible into or exercisable or exchangeable for shares of Parent Common Stock (including without limitation, shares of Parent Common Stock or such other
securities of Parent which may be deemed to be beneficially owned by the undersigned in accordance with the rules and regulations of the SEC and securities of Parent which may be issued upon exercise or vesting, as applicable, of a stock option or
warrant or settlement of a restricted stock unit or restricted stock award and Parent Common Stock or such other securities to be issued to the undersigned in connection with the Merger, in each case, that are currently or hereafter owned of record
or beneficially (collectively, the “Undersigned’s Shares”);
(2) enter into any swap, short sale, hedge or other agreement that transfers, in whole or in part, any of the economic consequences of ownership of the Undersigned’s Shares regardless of whether
any such transaction described in clause (1) above or this clause (2) is to be settled by delivery of shares of Parent Common Stock or other securities, in cash or otherwise;
(3) make any demand for, or exercise any right with respect to, the registration of any shares of Parent Common Stock or any security convertible into or exercisable or exchangeable for shares of
Parent Common Stock (other than such rights set forth in the Merger Agreement or Registration Rights Agreement);
(4) except for any support agreement entered into as of the date hereof by the undersigned with Parent and the Company, grant any proxies or powers of attorney with respect to any Parent Common
Stock, deposit any Parent Common Stock into a voting trust or enter into a voting agreement or similar arrangement or commitment with respect to any Parent Common Stock; or
(5) publicly disclose the intention to do any of the foregoing.
The restrictions and obligations contemplated by this Lock-Up Agreement shall not apply to:
(a) transfers of the Undersigned’s Shares:
(i) (A) to any person related to the undersigned (or to an ultimate beneficial owner of the undersigned) by blood or adoption who is an immediate family member of the undersigned, or by
marriage or domestic partnership (each, a “Family Member”), or to a trust formed for the benefit of the undersigned or any of the undersigned’s Family Members, (B) to the undersigned’s estate, following the death of the undersigned, by will,
intestacy or other operation of Law, (C) as a bona fide gift or a charitable contribution, as such term is described in Section 501(c)(3) of the Code, or otherwise to a trust or other entity for the direct or indirect benefit of an immediate family
member of a beneficial owner (as defined in Rule 13d-3 of the Exchange Act) of the Undersigned’s Shares (D) by operation of Law, such as pursuant to a qualified domestic order or in connection with a divorce settlement or (E) to any partnership,
corporation, limited liability company or other entity, in each case, the beneficial ownership interests of all of which are held by or otherwise under common control (via beneficial ownership, contract or otherwise) with the undersigned or a
Family Member of the undersigned;
(ii) if the undersigned is a corporation, partnership, limited liability company or other entity, (A) to another corporation, partnership, limited liability company or other entity that is a
direct or indirect affiliate (as defined under Rule 12b-2 of the Exchange Act) of the undersigned, including investment funds or other entities that controls or manages, is under common control or management with, or is controlled or managed by,
the undersigned (including, for the avoidance of doubt, where the undersigned is a partnership, to its general partner or a successor partnership or fund, or any other funds managed by such partnership), (B) as a distribution or dividend to equity
holders, current or former partners, members, stockholders or managers (or to the estates of any of the foregoing), as applicable, of the undersigned (including upon the liquidation and dissolution of the undersigned pursuant to a plan of
liquidation approved by the undersigned’s equity holders), (C) as a bona fide gift or a charitable contribution, as such term is described in Section 501(c)(3) of the Code, or otherwise to a trust or other entity for the direct or indirect benefit
of an immediate family member of a beneficial owner (as defined in Rule 13d-3 of the Exchange Act) of the Undersigned’s Shares, (D) transfers or dispositions not involving a change in beneficial ownership or (E) with prior written consent of Parent
(as constituted following the Closing); or
(iii) if the undersigned is a trust, to any grantors or beneficiaries of the trust;
provided that, in the case of any transfer or distribution pursuant to this clause (a), such transfer is not for value (other than transfers pursuant to clauses (a)(i)(A), (a)(i)(E) or
(a)(ii)(A) hereto) and each donee, heir, beneficiary or other transferee or distributee shall sign and deliver to Parent a lock-up agreement in the form of this Lock-Up Agreement with respect to the shares of Parent Common Stock or such other
securities that have been so transferred or distributed and if a filing pursuant to Section 16(a) of the Exchange Act is required, such filing shall describe the nature of the transfer or distribution;
(b) the exercise of an option to purchase shares of Parent Common Stock (including a net or cashless exercise of an option to purchase shares of Parent Common Stock ), and any related
transfer of shares of Parent Common Stock to Parent or open market sale of Parent Common Stock for the purpose of paying the exercise price of such options or for paying taxes (including estimated taxes) due as a result of the exercise of such
options, and open market sale for the purpose of paying taxes (including estimated taxes) due as a result of the receipt of Parent Common Stock (or in the case of the vesting of restricted stock units pursuant to terms of the Merger Agreement
converted into shares of Parent Common Stock pursuant to the terms of the Merger Agreement); provided that, for the avoidance of doubt, any underlying shares of Parent Common Stock not transferred to sold pursuant to this clause shall
continue to be subject to the restrictions on transfer set forth in this Lock-Up Agreement;
(c) transfers to Parent in connection with the net settlement of any other equity award that represents the right to receive in the future shares of Parent Common Stock, settled in shares
of Parent Common Stock, to pay any tax withholding obligations; provided that, for the avoidance of doubt, the underlying shares of Parent Common Stock shall continue to be subject to the restrictions on transfer set forth in this Lock-Up
Agreement;
(d) the establishment of a trading plan pursuant to Rule 10b5-l under the Exchange Act for the transfer of shares of Parent Common Stock; provided that such plan does not provide for
any transfers of shares of Parent Common Stock during the Restricted Period;
(e) the disposition (including a forfeiture or repurchase) to Parent of any shares of restricted stock granted pursuant to the terms of any employee benefit plan or restricted stock
purchase agreement;
(f) transfers, distributions, sales or other transactions by the undersigned of shares of Parent Common Stock purchased by the undersigned on the open market or in a public offering by
Parent, in each case following the date of the Closing;
(g) transfers pursuant to a bona-fide third party tender offer, merger, consolidation or other similar transaction made to all holders of Parent’s capital stock involving a change of control
of Parent, provided that in the event that such tender offer, merger, consolidation or other such transaction is not completed, the Undersigned’s Shares shall remain subject to the restrictions contained in this Lock-Up Agreement;
(h) transfers pursuant to an order of a court or regulatory agency; or
(i) transfers, distributions, sales or other transactions with the prior written consent of Parent (as constituted following the Closing);
and provided further, that, with respect to each of (b), (c), and (d) above, no filing by any party (including any donor, donee, transferor, transferee, distributor or distributee) under Section 16 of the
Exchange Act or other public announcement shall be made voluntarily reporting a reduction in beneficial ownership of shares of Parent Common Stock or any securities convertible into or exercisable or exchangeable for Parent Common Stock in
connection with such transfer or disposition during the Restricted Period (other than any exit filings) and if any filings under Section 16(a) of the Exchange Act, or other public filing, report or announcement reporting a reduction in beneficial
ownership of shares of Parent Common Stock in connection with such transfer or distribution, shall be legally required during the Restricted Period, such filing, report or announcement shall clearly indicate in the footnotes therein, in reasonable
detail, a description of the circumstances of the transfer and that the shares remain subject to this Lock-Up Agreement.
For purposes of this Lock-Up Agreement, “change of control” shall mean the transfer (whether by tender offer, merger, consolidation or other similar transaction), in one transaction or a series of related
transactions to a person or group of affiliated persons, of Parent’s voting securities if, after such transfer, Parent’s stockholders as of immediately prior to such transfer do not hold a majority of the outstanding voting securities of Parent (or
the surviving entity).
Any attempted transfer in violation of this Lock-Up Agreement will be of no effect and null and void, regardless of whether the purported transferee has any actual or constructive knowledge of the
transfer restrictions set forth in this Lock-Up Agreement, and will not be recorded on the share register of Parent. In furtherance of the foregoing, the undersigned agrees that Parent and any duly appointed transfer agent for the registration or
transfer of the securities described herein are hereby authorized to decline to make any transfer of securities if such transfer would constitute a violation or breach of this Lock-Up Agreement. Parent may cause the legend set forth below, or a
legend substantially equivalent thereto, to be placed upon any certificate(s) or other documents, ledgers or instruments evidencing the undersigned’s ownership of Parent Common Stock or any securities convertible into or exercisable or exchangeable
for Parent Common Stock:
THE SHARES REPRESENTED BY THIS CERTIFICATE ARE SUBJECT TO AND MAY ONLY BE TRANSFERRED IN COMPLIANCE WITH A LOCK-UP AGREEMENT, A COPY OF WHICH IS ON FILE AT THE PRINCIPAL OFFICE
OF THE COMPANY.
The undersigned hereby represents and warrants that the undersigned has full power and authority to enter into this Lock-Up Agreement. All authority herein conferred or agreed to be conferred and any
obligations of the undersigned shall be binding upon the successors, assigns, heirs or personal representatives of the undersigned.
The undersigned understands that if the Merger Agreement is terminated for any reason, the undersigned shall be released from all obligations under this Lock-Up Agreement. The undersigned understands
that the Board of Directors of Parent may waive any term of this Lock-Up Agreement at any time and for any reason. The undersigned understands that Parent is proceeding with the transactions contemplated by the Merger Agreement in reliance upon
this Lock-Up Agreement.
Any and all remedies herein expressly conferred upon Parent or the Company will be deemed cumulative with and not exclusive of any other remedy conferred hereby, or by Law or equity, and the exercise
by Parent or the Company of any one remedy will not preclude the exercise of any other remedy. The undersigned agrees that irreparable damage for which monetary damages, even if available, would not be an adequate remedy, would occur to Parent
and/or the Company in the event that any of the provisions of this Lock-Up Agreement were not performed in accordance with its specific terms or were otherwise breached. It is accordingly agreed that Parent and the Company shall be entitled to an
injunction or injunctions to prevent breaches of this Lock-Up Agreement and to enforce specifically the terms and provisions hereof in any court of the United States or any state having jurisdiction, this being in addition to any other remedy to
which Parent or the Company is entitled at Law or in equity, and the undersigned waives any bond, surety or other security that might be required of Parent or the Company with respect thereto. Each of the parties further agrees that it will not
oppose the granting of an injunction, specific performance or other equitable relief on the basis that any other party has an adequate remedy at Law or that any award of specific performance is not an appropriate remedy for any reason at Law or in
equity.
In the event that any holder of Parent’s securities that are subject to a substantially similar agreement entered into by such holder, other than the undersigned, is permitted by Parent to sell or
otherwise transfer or dispose of shares of Parent Common Stock or any securities convertible into or exercisable or exchangeable for Parent Common Stock for value other than as permitted by this or a substantially similar agreement entered into by
such holder (whether in one or multiple releases or waivers), the same percentage of shares of Parent Common Stock or any securities convertible into or exercisable or exchangeable for Parent Common Stock held by the undersigned on the date of such
release or waiver as the percentage of the total number of outstanding shares of such securities held by such holder on the date of such release or waiver that are the subject of such release or waiver shall be immediately and fully released on the
same terms from any remaining restrictions set forth herein (the “Pro-Rata Release”); provided, however, that such Pro-Rata Release shall not be applied unless and until permission has been granted by Parent to an equity
holder or equity holders to sell or otherwise transfer or dispose of all or a portion of such equity holders shares of Parent Common Stock in an aggregate amount in excess of 1% of the number of shares of Parent Common Stock subject to a
substantially similar agreement. In the event of any Pro-Rata Release, the Company shall promptly (and in any event within two (2) Business Days of such release) inform each relevant holder of Parent Common Stock of the terms of such Pro-Rata
Release.
Upon the release of any of the Undersigned’s Shares from this Lock-Up Agreement, Parent will promptly facilitate the timely preparation and delivery of certificates or the establishment of book-entry
positions at Parent’s transfer agent representing the Undersigned’s Shares without the restrictive legend above or the withdrawal of any stop transfer instructions by virtue of this Lock-Up Agreement.
This Lock-Up Agreement and any claim, controversy or dispute arising under or related to this Lock-Up Agreement shall be governed by and construed in accordance with the Laws of the State of Delaware,
regardless of the laws that might otherwise govern under applicable principles of conflicts of laws. In any action or proceeding between any of the parties arising out of or relating to this Lock-Up Agreement, each of the parties: (i) irrevocably
and unconditionally consents and submits to the exclusive jurisdiction and venue of the Court of Chancery of the State of Delaware or, to the extent such court does not have subject matter jurisdiction, the Superior Court of the State of Delaware
or the United States District Court for the District of Delaware, (ii) agrees that all claims in respect of such action or proceeding shall be heard and determined exclusively in accordance with foregoing clause (i) of this paragraph, (iii) waives
any objection to laying venue in any such action or proceeding in such courts, (iv) waives any objection that such courts are an inconvenient forum or do not have jurisdiction over any party and (v) irrevocably and unconditionally waives the right
to trial by jury. This Lock-Up Agreement constitutes the entire agreement between the parties to this Lock-Up Agreement and supersedes all other prior agreements, arrangements and understandings, both written and oral, among the parties with
respect to the subject matter hereof.
THE PARTIES HERETO HEREBY WAIVE ANY RIGHT TO TRIAL BY JURY WITH RESPECT TO ANY ACTION OR LEGAL PROCEEDING RELATED TO OR ARISING OUT OF THIS LOCK-UP AGREEMENT, ANY DOCUMENT
EXECUTED IN CONNECTION HEREWITH AND THE MATTERS CONTEMPLATED HEREBY AND THEREBY.
This Lock-Up Agreement may be executed in several counterparts, each of which shall be deemed an original and all of which shall constitute one and the same instrument. The exchange of a fully
executed Lock-Up Agreement (in counterparts or otherwise) by Parent, the Company and the undersigned by electronic transmission in .pdf format shall be sufficient to bind such parties to the terms and conditions of this Lock-Up Agreement.
[SIGNATURE PAGE FOLLOWS]
The undersigned understands that this Lock-Up Agreement is irrevocable and shall be binding upon the undersigned and the heirs, personal representatives, successors and assigns of the undersigned.
|
Very truly yours,
|
|||
|
Print Name of Stockholder:
|
|||
|
By:
|
|||
|
Name:
|
|||
|
Title:
|
|||
[Signature Page to Lock-Up Agreement]
Accepted and Agreed
by Gyre Therapeutics, Inc.:
|
By:
|
||
|
Name:
|
||
|
Title:
|
||
|
Accepted and Agreed
|
||
|
by Cullgen Inc.:
|
||
|
By:
|
||
|
Name:
|
||
|
Title:
|
||
[Signature Page to Lock-Up Agreement]
Exhibit 99.1
 |
 |
Gyre Therapeutics Enters into Agreement to Acquire Cullgen to Gain Targeted Protein Degradation Platform and Pipeline
| • |
Acquisition will create a U.S.- and China-based fully integrated biopharmaceutical company with revenue-producing commercial assets and a robust pipeline of degraders, targeting inflammatory diseases and
cancers.
|
| • |
Access to degrader-antibody conjugates (DACs) platform technology for future discovery engine.
|
| • |
Strengthened leadership team designed to support future global growth.
|
| • |
Transaction is anticipated to close early in the second quarter of 2026.
|
| • |
An updated corporate presentation has been posted to the Gyre and Cullgen websites.
|
San Diego, CA, March 2, 2026 – Gyre Therapeutics, Inc. (Gyre or the Company) (Nasdaq: GYRE), an innovative, commercial-stage biopharmaceutical company dedicated to
advancing fibrosis-first therapies across organ systems affected by chronic diseases, today announced its agreement to acquire Cullgen Inc. (Cullgen), a privately-held, clinical-stage biopharmaceutical company focused on the discovery and
development of targeted protein degrader (TPD) and degrader antibody conjugate (DAC) therapies, in an all-stock transaction valued at approximately $300 million. Following the closure of the acquisition, the new combined entity will be a fully
integrated biopharmaceutical company with U.S.- and China-based capabilities spanning from discovery to manufacturing and commercialization and covering multiple therapeutic areas including inflammatory diseases, cancers, and pain.
Under the terms of the definitive agreement, Cullgen will become a wholly owned subsidiary of Gyre. Upon the completion of the acquisition, the interim Chief Executive
Officer and Executive Chairman of Gyre, Ping Zhang, will remain as the Executive Chairman. The current Chief Executive Officer of Cullgen, Dr. Ying Luo, is expected to become the President and Chief Executive Officer and a member of the board of
directors of Gyre.
Dr. Luo, the expected President and Chief Executive Officer of Gyre, commented, “We are thrilled
about the synergistic coalescing of our companies. Cullgen brings strong drug discovery capabilities and a solid preclinical and clinical pipeline to complement Gyre’s existing and highly efficient China-based manufacturing capabilities and sales
team. Gyre is already a commercial-stage company with ETUARY® on the market in China for the treatment of lung fibrosis and a second product for liver fibrosis, Hydronidone (F351), nearing New Drug Application (NDA), submission in China.
Gyre is also exploring the expansion of F351’s development in ex-China territories. Following the acquisition, we will have a fully-integrated biopharmaceutical company that will be capable of leveraging emerging drug discovery capabilities in
China and strong clinical development in the United States to address unmet medical needs worldwide. I am excited for the potential of TPDs and DACs to drive this Company’s future growth globally.”
Mr. Zhang, Chairman of Gyre, commented, “Recently, Gyre, through its majority owned subsidiary, Gyre
Pharmaceuticals, had a pre-NDA meeting with the Center for Drug Evaluation (CDE) of China’s National Medical Products Administration (NMPA) which supported a conditional approval and priority review eligibility filing for Gyre Pharmaceuticals’
first-in-class anti-liver fibrosis candidate, Hydronidone, subject to formal approval. As a result, Gyre Pharmaceuticals plans to submit an NDA for Hydronidone for conditional approval in the first half of 2026 and conduct a Phase 3c confirmatory
trial to support full approval in China. The addition of Cullgen’s TPD/DAC platform and pipeline is expected to enhance our long-term growth prospects. We are excited to have Cullgen colleagues join our team in both the United States and China.”
The transaction is expected to close early in the second quarter of 2026, subject to customary closing conditions, including necessary regulatory approvals in the United
States.
Prior to entering into this transaction, Cullgen’s proposed merger with Pulmatrix was terminated.
Updated Corporate Presentation
In connection with this announcement, a new presentation has been posted on each company’s respective website. Visit http://www.cullgen.com or http://www.gyretx.com.
About Cullgen Inc.
Cullgen is a clinical-stage biopharmaceutical company focused on the discovery and development of targeted protein degrader and DAC therapies designed to improve the
lives of patients suffering from critical conditions such as pain, cancer and inflammatory diseases. Cullgen has created a portfolio of highly selective targeted protein degrader product candidates designed to potently and efficiently eliminate
therapeutically relevant proteins in patients. By leveraging its expertise in targeted protein degraders, Cullgen believes its product candidates have many distinct advantages over other therapeutic modalities, including higher selectivity,
improved therapeutic profile and avoidance of known toxicities.
Cullgen’s lead product candidate, CG001419, is an oral pan-tropomyosin receptor kinase (TRK) degrader that previously completed a Phase 1 trial for the treatment of acute post-operative pain and Cullgen released positive top-line results from the study in late 2025. Cullgen recently submitted an IND for the molecule, and pending FDA
allowance, expects to initiate a Phase 2 trial in acute pain in bunionectomy patients in the United States. The molecule is also being studied in a Phase 1 trial for the
treatment of solid tumors. Cullgen’s second product candidate, CG009301, is a GSPT1 degrader being studied in a Phase 1 trial for the treatment of blood cancers, including
relapsed/refractory acute myeloid leukemia, higher-risk myelodysplastic syndrome and acute lymphoblastic leukemia. In addition to CG001419 and CG009301, Cullgen is also progressing a number of preclinical programs including next-generation
degrader-antibody conjugates.
About Gyre Pharmaceuticals
Gyre Pharmaceuticals is a commercial-stage biopharmaceutical company committed to the research, development, manufacturing and commercialization of innovative drugs for
organ fibrosis. Its flagship product, ETUARY® (pirfenidone capsule), was the first approved treatment for IPF in the PRC in 2011 and has maintained a prominent
market share (2024 net sales of $105.8 million). In addition, Gyre Pharmaceuticals’ pipeline includes Hydronidone, a structural analogue of pirfenidone, which demonstrated statistically significant fibrosis regression after 52 weeks of treatment
in a pivotal Phase 3 clinical trial in CHB-associated liver fibrosis in the PRC. Hydronidone received Breakthrough Therapy designation by the CDE of the NMPA in March 2021. Gyre Pharmaceuticals is also developing treatments for PD, RILI with or
without immune-related pneumonitis, COPD, PAH and ALF/ACLF. As of the third quarter of 2025, Gyre Therapeutics owns a 69.7% equity interest in Gyre Pharmaceuticals.
About Gyre Therapeutics
Gyre Therapeutics is a biopharmaceutical company headquartered in San Diego, CA, primarily focused on the development and commercialization of Hydronidone for liver
fibrosis including MASH in the U.S. Gyre’s strategy builds on its experience in mechanistic studies using MASH rodent models and clinical studies in CHB-induced liver fibrosis. In the PRC, Gyre is advancing a broad pipeline through its
controlling interest in Gyre Pharmaceuticals, including therapeutic expansions of ETUARY, and development programs for F573, F528, and F230.
Advisory and Legal Counsel
Moelis & Company LLC is acting as financial advisor to the special committee to Gyre’s Board of Directors, and Gyre’s legal counsel is Gibson, Dunn & Crutcher
LLP.
Mintz, Levin, Cohn, Ferris, Glovsky & Popeo, P.C. is serving as legal counsel to Cullgen.
Forward-Looking Statements
This press release contains “forward-looking statements” within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995,
which statements are subject to substantial risks and uncertainties and are based on estimates and assumptions. All statements, other than statements of historical facts included in this press release, are forward-looking statements, including
statements concerning: the structure, timing and completion of the proposed acquisition; the anticipated timing of closing; the expected executive officers and directors of the combined entity; the future operations of the combined entity; the
nature, strategy and focus of the combined entity; the development and commercial potential and potential benefits of any product candidates of the combined entity; and anticipated clinical drug development activities and related timelines,
including the anticipated timing of the filing of Gyre’s NDA with the NMPA for the conditional approval of Hydronidone and the initiation of the confirmatory Phase 3c clinical trial of Hydronidone to support full approval in China and the
anticipated timing of Cullgen’s Phase 2 trial of its TRK degrader in acute pain. In some cases, you can identify forward-looking statements by terms such as “may,” “might,” “will,” “objective,” “intend,” “should,” “could,” “can,” “would,”
“expect,” “believe,” “design,” “estimate,” “predict,” “potential,” “plan” or the negative of these terms, and similar expressions intended to identify forward-looking statements. These statements reflect our plans, estimates, and expectations, as
of the date of this press release. These statements involve known and unknown risks, uncertainties and other factors that could cause our actual results to differ materially from the forward-looking statements expressed or implied in this press
release. Actual results and the timing of events could differ materially from those anticipated in such forward-looking statements as a result of these risks and uncertainties, which include, without limitation: the risk that the conditions to
the closing of the acquisition are not satisfied; uncertainties as to the timing of the consummation of the acquisition and the ability of each of Gyre and Cullgen to consummate the acquisition; risks related to the failure or delay in obtaining
required approvals from any governmental or quasi-governmental entity necessary to consummate the acquisition; unexpected costs, charges or expenses resulting from the acquisition; potential adverse reactions or changes to business relationships
resulting from the announcement or completion of the acquisition; the uncertainties associated with Gyre’s and Cullgen’s product candidates, as well as risks associated with the clinical development and regulatory approval of product candidates,
including potential delays in the commencement, enrollment and completion of clinical trials; risks related to the inability of the combined entity to obtain sufficient additional capital to continue to advance these product candidates and its
preclinical programs; uncertainties in obtaining successful clinical results for product candidates and unexpected costs that may result therefrom; risks related to the failure to realize any value from product candidates and preclinical programs
being developed and anticipated to be developed in light of inherent risks and difficulties involved in successfully bringing product candidates to market; risks associated with the possible failure to realize certain anticipated benefits of the
acquisition, including with respect to future financial and operating results. Additional risks and factors are identified under “Risk Factors” in Gyre’s Annual Report on Form 10-K for the year ended December 31, 2024 filed on March 17, 2025 and
in other filings with the Securities and Exchange Commission.
Gyre expressly disclaims any obligation to update any forward-looking statements whether as a result of new information, future events or otherwise, except as required
by law.
CONTACTS:
Gyre Therapeutics, Inc.
Ping Zhang, interim CEO and executive chairman
ping.zhang@gyretx.com
Cullgen Inc.
Thomas Eastling, CFO
ir@cullgen.com
Investors
Chuck Padala
Managing Director, LifeSci Advisors
chuck@lifesciadvisors.comManaging Director, LifeSci Advisors
Exhibit 99.2